2009 MBSW May 18 20 Quality Risk Assessment

2009 MBSW, May 18 -20 Quality Risk Assessment: a Lifecycle Approach in Evaluating Quality Attributes for Bioproducts Suntara Cahya, Ph. D

Outline • “Biomolecule Diversity” • Molecule Risk Assessments • Risk Scoring • Relation with CQA’s • Concluding Remarks Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 2
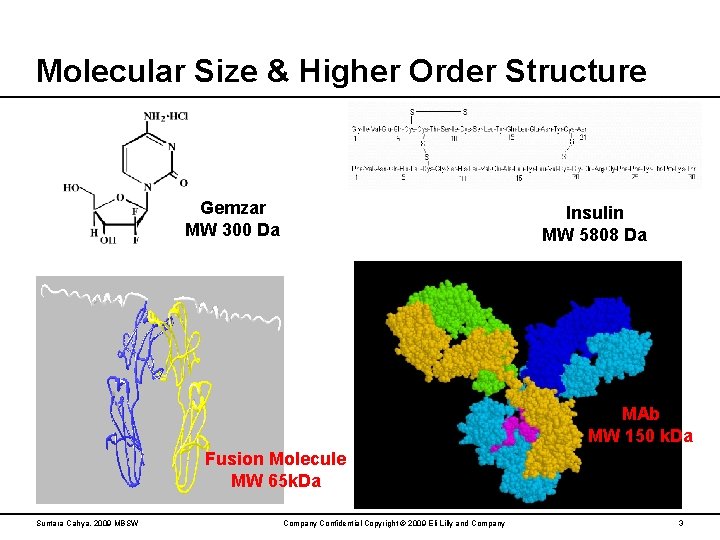
Molecular Size & Higher Order Structure Gemzar MW 300 Da Insulin MW 5808 Da MAb MW 150 k. Da Fusion Molecule MW 65 k. Da Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 3

Molecular Heterogeneity Translational Events • Splicing • Frame shifts • Intron read-through Post-Translational Modifications • Glycosylation (N- & O-linked) – Site occupancy – Glycan structures • Phosphorylation Chemical & Enzymatic Modifications • Aggregation • Proteolytic clipping • Deamidation • Isomerization • Oxidation • Glycation • Pyruvylation • Pyroglutamatation Suntara Cahya, 2009 MBSW Potential to Impact: • Manufacturing consistency (lot-tolot variability) CM&C • Stability • Toxicity Toxicology • Efficacy • Safety Clinical • Immunogenicity • PK/PD Company Confidential Copyright © 2009 Eli Lilly and Company ADME 4
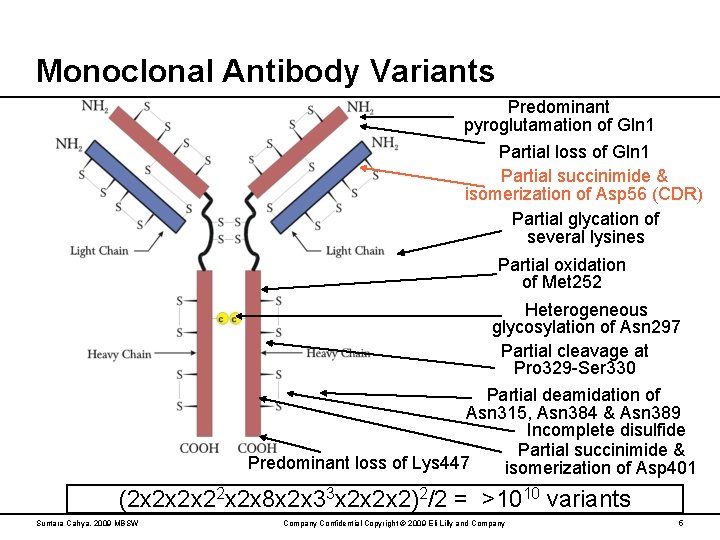
Monoclonal Antibody Variants Predominant pyroglutamation of Gln 1 Partial loss of Gln 1 Partial succinimide & isomerization of Asp 56 (CDR) Partial glycation of several lysines Partial oxidation of Met 252 Heterogeneous glycosylation of Asn 297 Partial cleavage at Pro 329 -Ser 330 Partial deamidation of Asn 315, Asn 384 & Asn 389 Incomplete disulfide Partial succinimide & Predominant loss of Lys 447 isomerization of Asp 401 (2 x 2 x 8 x 2 x 33 x 2 x 2 x 2)2/2 = >1010 variants Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 5

Molecule Risk Assessments • Complexity in managing the risks (Safety & Efficacy) • Impact on control strategies • Had historically been part of dev process – Assessments more subjective – Opinions could vary depending on the individuals • Motivation: More standard & objective ways of performing the risk assessment Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 6

Molecule Risk Assessments • Motivation cont’d • • • Consistent approach that works for multiple molecules (Antibodies, Fusion Proteins, Peptides) Phase appropriate assessments Documentations at key points during molecule development Consistent with ICH guidance on risk assessments and Qb. D (ICH Q 9 and ICH Q 8) Focused on molecule quality attributes and analytical control strategy Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 7

Risk Components • Broken down into three risk components • Severity: The impact of the particular quality attribute on the pharmacological properties of the biomolecule, including toxicity, safety, efficacy, PK/PD profile, and immunogenicity – – Location of modification – is it in a region critical to molecule’s activity Documented evidence of impact Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 8

Risk Components • Detectability: The capability of the analytical methods to detect and quantify the particular quality attribute (i. e. , specific vs. non-specific method) as well as the use of the method in the analytical control strategy (i. e. , routine vs. non-routine method) – – – Routine method developed Non-routine developed Attribute is not currently monitored Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 9

Risk Components • Occurrence: The likelihood of the particular quality attribute to exceed a specified level of concern (“Exposure Limit”), based upon API and drug product manufacturing process variability as well as the stability of the API and drug product throughout their respective shelf-lives – – Stage of development Platform modification Data that is available Growth on stability Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 10

Risk Scoring • Each risk component has different levels of risk scoring • Overall risk scored based on Risk Priority Number (RPN) = Severity X Occurrence X Detectability • Flowcharts to facilitate scoring evaluation Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 11
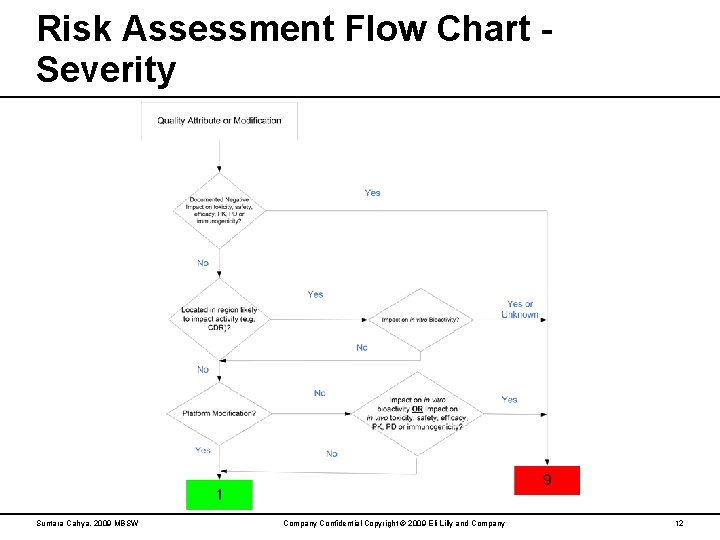
Risk Assessment Flow Chart Severity Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 12

Detectability “ 1”: Modification is detected and accurately quantified by a specific, routine test method OR Modification is detected by a non-specific, routine test method AND accurately quantified by a specific, non-routine test method, “ 3”: Modification is detected by a non-specific, routine method OR Modification is detected and accurately quantified by a specific, nonroutine test method “ 9”: Appropriate methods are not currently available or used for detection and accurate quantification of the modification. Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 13
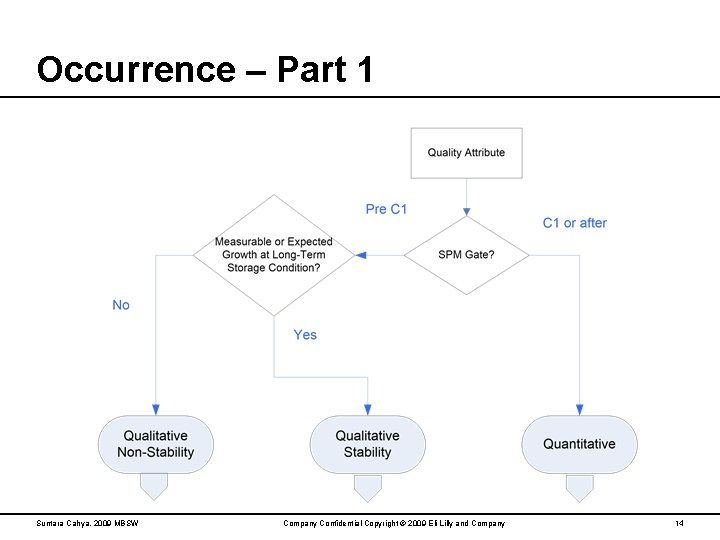
Occurrence – Part 1 Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 14

Occurrence – Qualitative Non. Stability Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 15
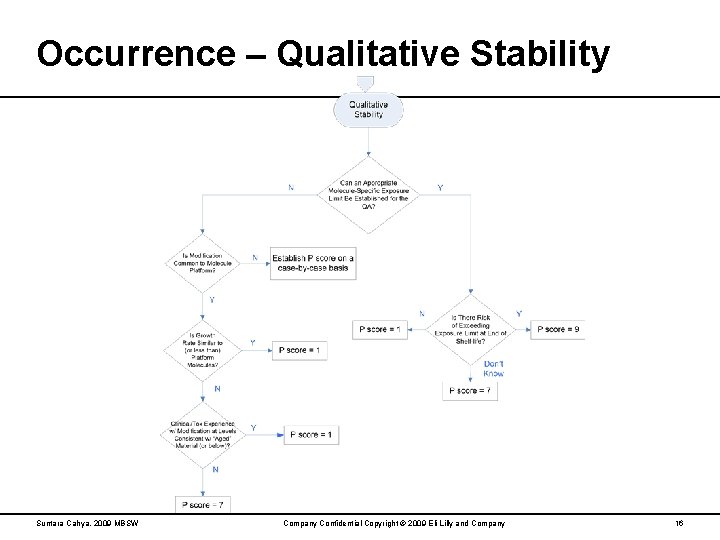
Occurrence – Qualitative Stability Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 16

Occurrence – Quantitative Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 17
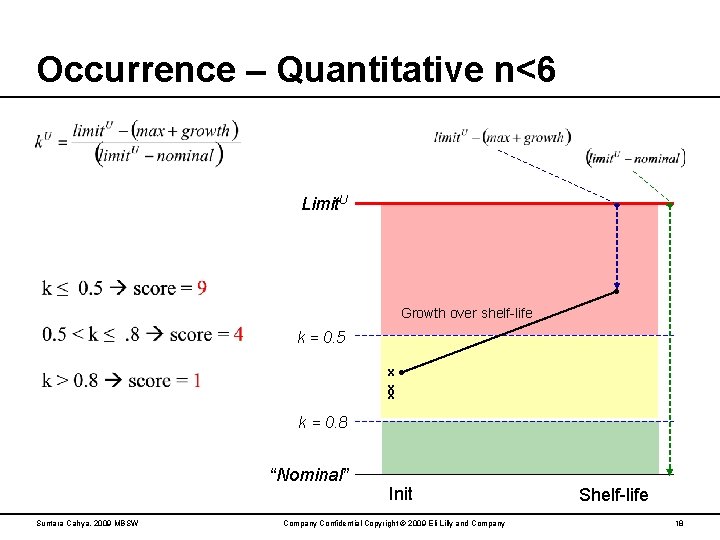
Occurrence – Quantitative n<6 Limit. U Growth over shelf-life k = 0. 5 x x x k = 0. 8 “Nominal” Suntara Cahya, 2009 MBSW Init Company Confidential Copyright © 2009 Eli Lilly and Company Shelf-life 18
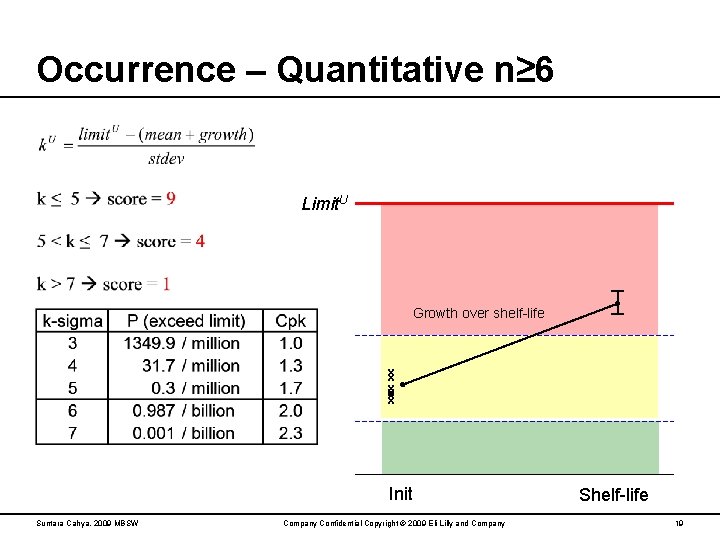
Occurrence – Quantitative n≥ 6 Limit. U Growth over shelf-life x x x Init Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company Shelf-life 19
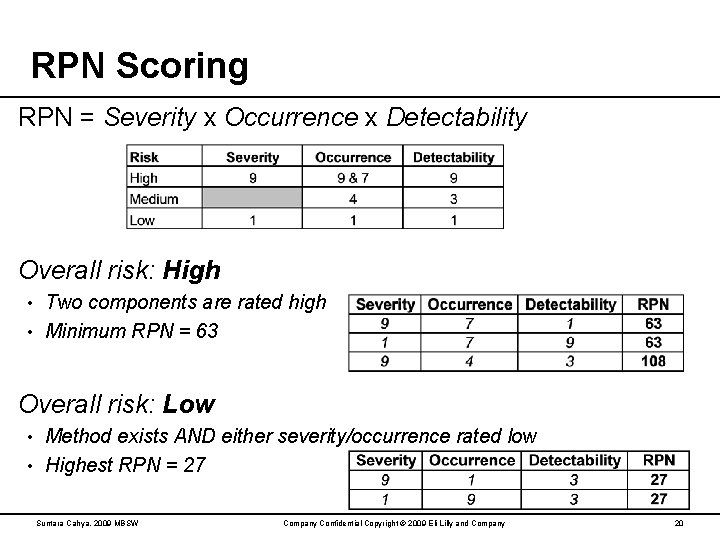
RPN Scoring RPN = Severity x Occurrence x Detectability Overall risk: High Two components are rated high • Minimum RPN = 63 • Overall risk: Low Method exists AND either severity/occurrence rated low • Highest RPN = 27 • Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 20
RPN Scoring Chance of “Harm” Degree of “Harm” Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 21

Relation with CQA’s • Tool to assess criticality of molecule attributes and identify potential CQA’s • List of potential CQA’s are broader than just molecule modifications • Some quality attributes are likely to always be “critical” due to their known potential to impact patient safety and product efficacy. • Examples Include: Host Cell Proteins, DNA, Endotoxin, Bioburden, Sterility, Viruses, Content/Protein Concentration, etc Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 22

Concluding Remarks • Risk Mitigations • Gaining additional information (more data: literature, in-vivo, additional lots, etc) • Better detection methods • Improved mfg process (level and/or variability) • Improved analytical method precision • “Exposure limit” vs Specification limit • Commercial specifications has been historically set based on process capability, may be tighter than the exposure limit • May re-assess the occurrence risk of OOS Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 23

Concluding Remarks • “Lifecycle” approach • Deliverables at gate reviews throughout development cycle • Documentations at key gate reviews • Guidance on roles and responsibilities: RACI diagram Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 24

Summary • The approach provides the basic framework to perform risk assessment of biomolecules • Consistency and objectivity across molecules • Phase appropriate evaluations • Continual revisions are expected as more experience is gained overtime Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 25

Risk Assessment Team Mike De. Felippis Bryan Harmon Cathy Srebalus Barnes Sarah Demmon Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 26

Acknowledgments Susan Janes Matt Hilton Melody Gossage Bruce Meiklejohn Juliana Kretsinger Jill Olinger Mandy Dorsey Owen Van Cauwenberghe Jeff Schwartzenhauer Kristi Griffiths Kamal Egodage Gary Sullivan Sacha Storms Chi Nguyen Agatha Feltus Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 27

Questions? Suntara Cahya, 2009 MBSW Company Confidential Copyright © 2009 Eli Lilly and Company 28
- Slides: 28