2003 6 KDA Mild Cognitive Impairment bhj 1405eulji

2003. 6. KDA 월례집담회 Mild Cognitive Impairment 배희준 (bhj 1405@eulji. or. kr) 을지의과대학 노원을지병원 신경과

CONTENTS I § § § § Why MCI? Definition of amnestic MCI Epidemiology Natural Hx of MCI Rating Scales Heterogeneity of MCI Diagnostic Criteria of AD: Probable and definite AD Neuropathology of AD and MCI

CONTENTS II § § § Neuropsychologic assessment Neuroimaging Biomarker Prognosis and Risk Factors Treatment – MCI trials

Why MCI?
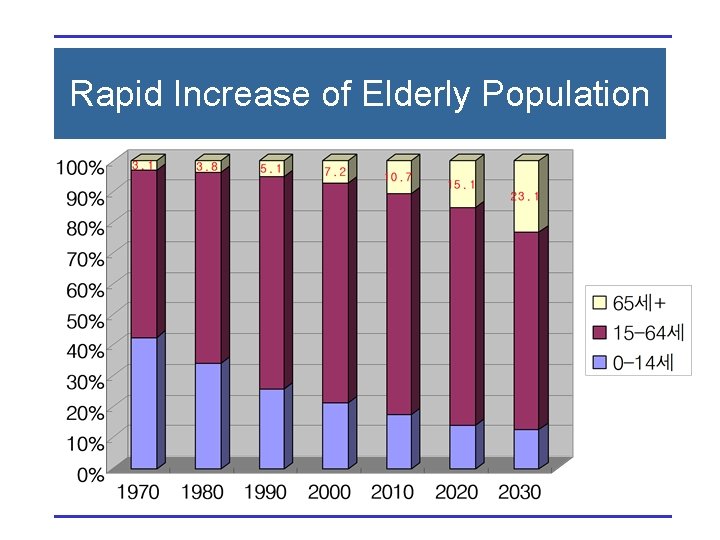
Rapid Increase of Elderly Population
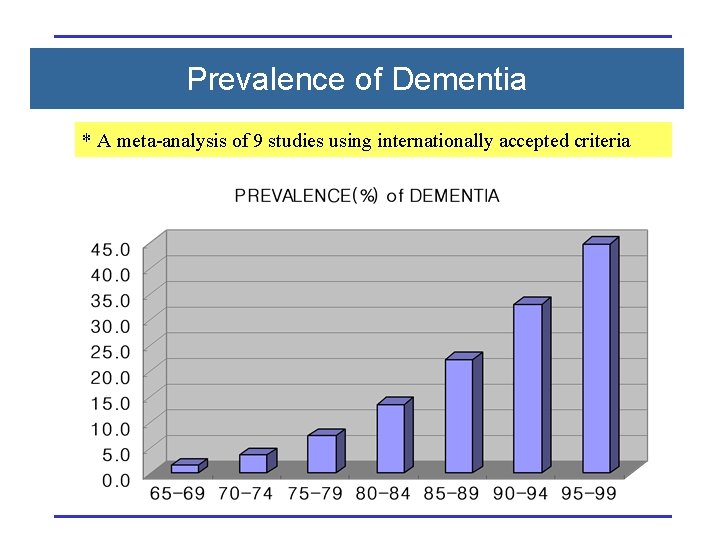
Prevalence of Dementia * A meta-analysis of 9 studies using internationally accepted criteria
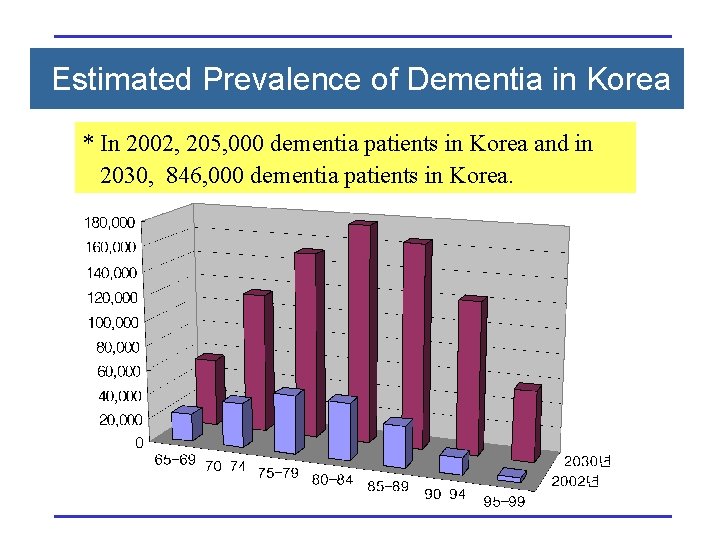
Estimated Prevalence of Dementia in Korea * In 2002, 205, 000 dementia patients in Korea and in 2030, 846, 000 dementia patients in Korea.
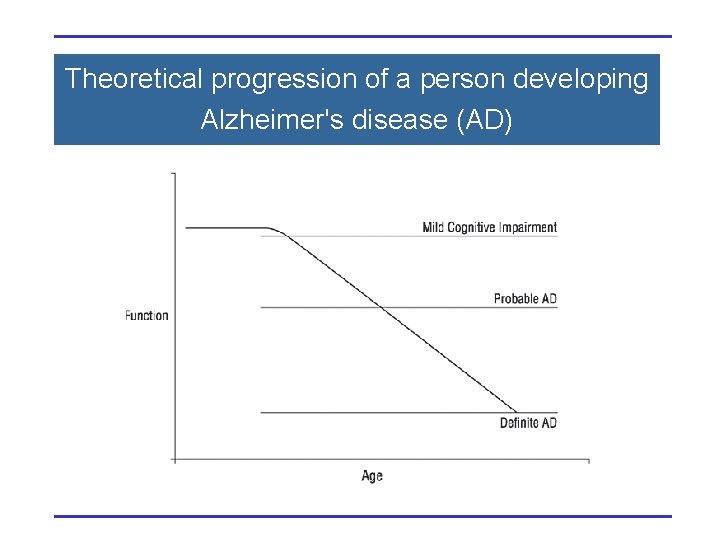
Theoretical progression of a person developing Alzheimer's disease (AD)

Treatment Options in AD • Tacrine • Estrogen • Donepezil • Vitamin E • Rivastigmine tartrate • Acetyl-L-carnitine • Galantamine • Statin hydrobromide • NSAIDs • Ginkgo biloba • Selegilne

If MCI is a transitional zone between normal aging and early dementia (esp. AD), why do we not treat MCI before dementia develops?

Definition of Amnestic MCI

Petersen RC, etc (1999) Arch Neurol 56: 303 -308. Criteria for Amnestic MCI • Memory complaints, preferably corroborated by an informant • Impaired memory function for age and education • Preserved general cognitive function • Intact activities of daily living • Not demented

Chertkow H (2002) Current Opinion in Neurology 15: 401 -407. New Criteria for Amnestic MCI • • • Subjective complaint of memory loss Objective impairment of memory Generally preserved other cognitive ability Preserved basic day-to-day functioning No other obvious medical neurologic or psychiatric explanation for the memory problems • Individual does not meet criteria for dementia
Epidemiology

• There has been little work on the incidence or prevalence of MCI because of the recent characterization of this condition. • Population-based studies, especially those that ascertain cases based on psychometric test results rather than symptoms, may identify a population of individuals with mild cognitive dysfunction that differs from the amnestic MCI described clinically. • Prevalence rates for AAMI in these studies range from 17% to 34%, while age-associated cognitive decline (DSM IV) has been estimated as having a prevalence of 26%. • Other investigators have indicated that the prevalence rate for conditions such as age-associated memory impairment can range up to 85%. • In the Canadian Study of Health and Aging, the classification of CIND had a prevalence rate of 17%; this is likely a more inclusive term than MCI alone. • An important factor in considering the frequency of MCI concerns the source of subjects for a given study.


Natural History of MCI
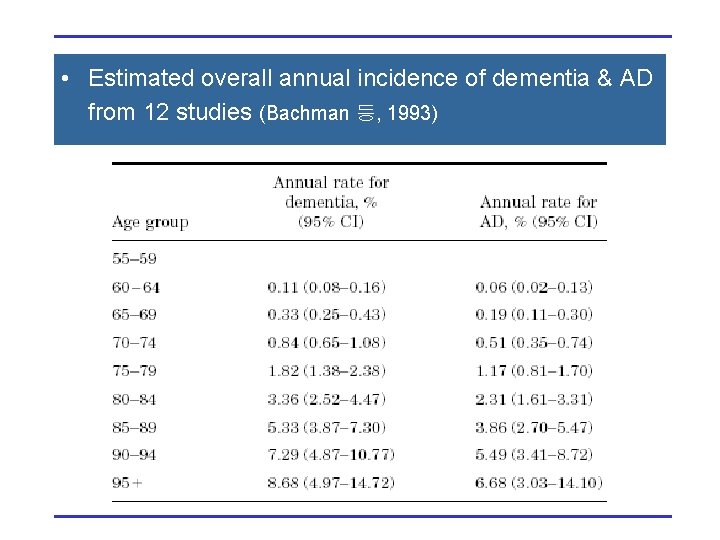
• Estimated overall annual incidence of dementia & AD from 12 studies (Bachman 등, 1993)
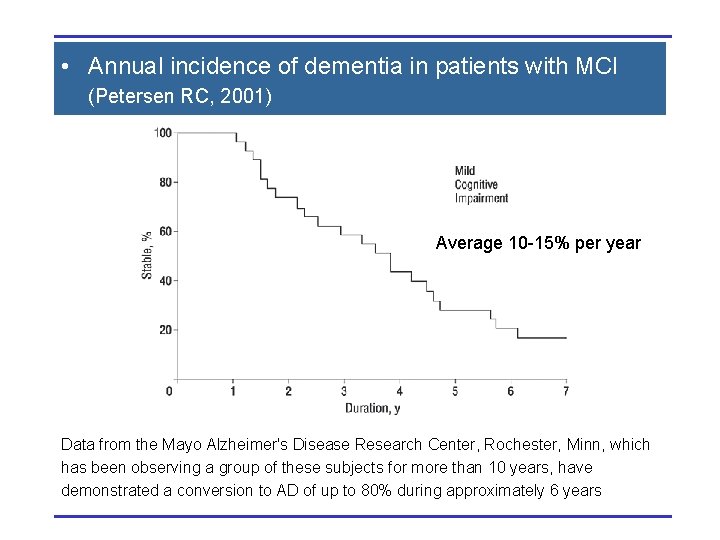
• Annual incidence of dementia in patients with MCI (Petersen RC, 2001) Average 10 -15% per year Data from the Mayo Alzheimer's Disease Research Center, Rochester, Minn, which has been observing a group of these subjects for more than 10 years, have demonstrated a conversion to AD of up to 80% during approximately 6 years

Rating Scales
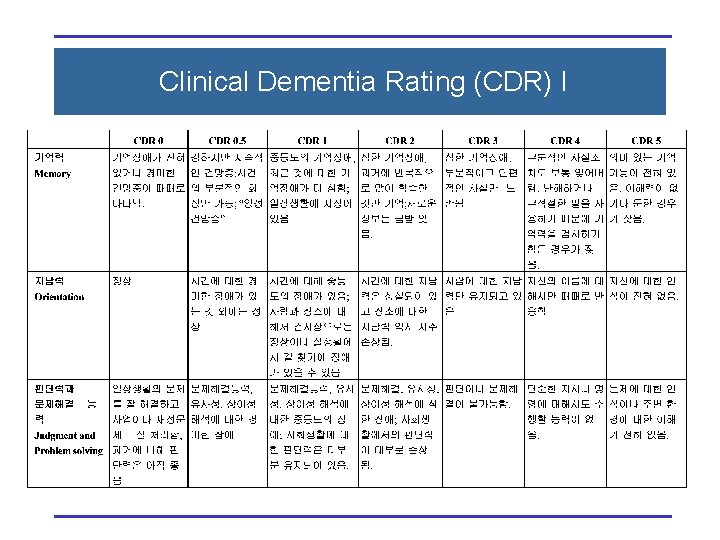
Clinical Dementia Rating (CDR) I
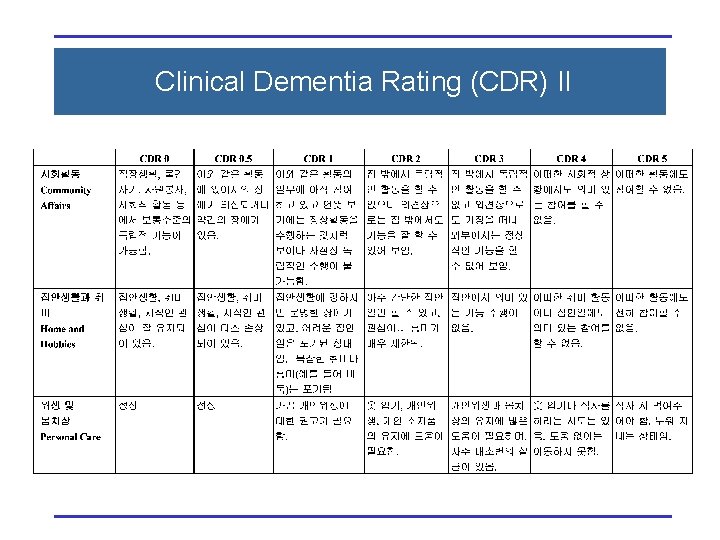
Clinical Dementia Rating (CDR) II

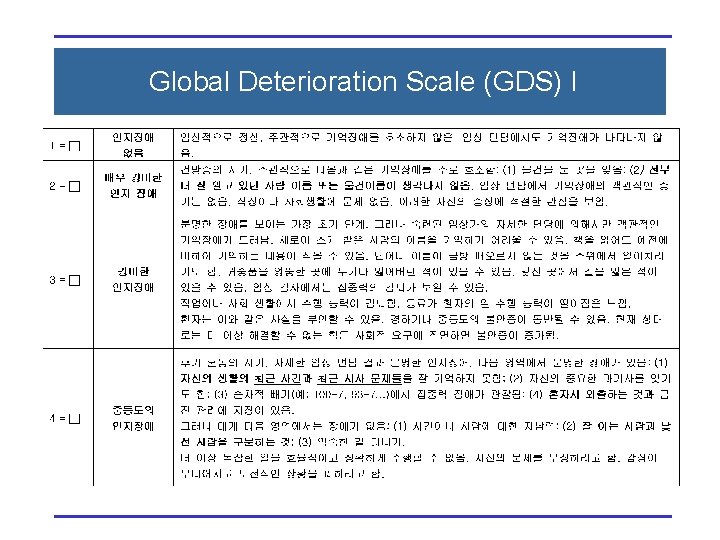
Global Deterioration Scale (GDS) I

Global Deterioration Scale (GDS) II
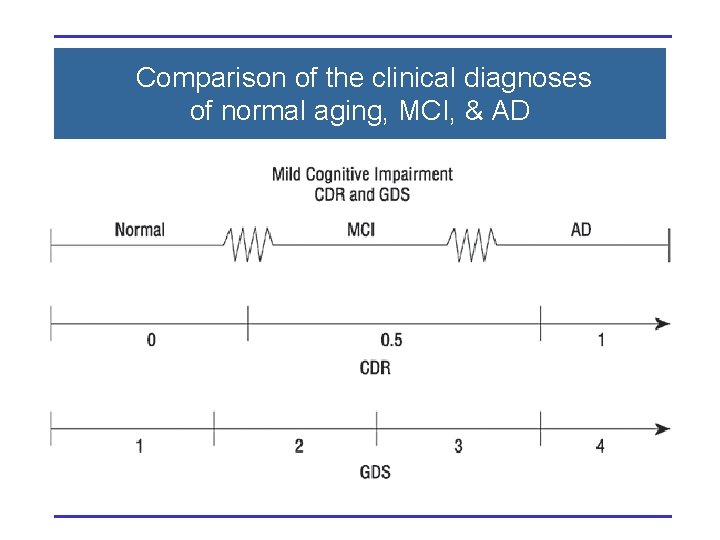
Comparison of the clinical diagnoses of normal aging, MCI, & AD

Heterogeneity of MCI
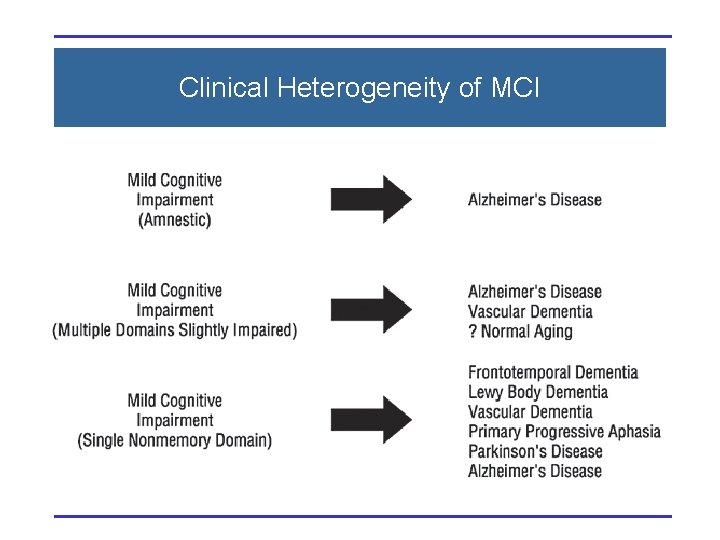
Clinical Heterogeneity of MCI
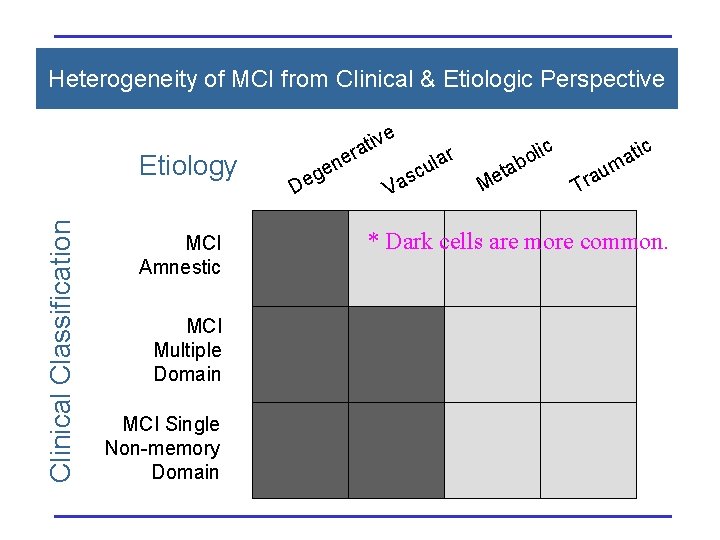
Heterogeneity of MCI from Clinical & Etiologic Perspective Clinical Classification Etiology MCI Amnestic MCI Multiple Domain MCI Single Non-memory Domain n D e eg ve i t era V c as r ula ta e M lic o b tic a m u a r T * Dark cells are more common.

Diagnostic Criteria of AD

NINDS-ADRDA Criteria Clinically Probable AD All of the followings • Dementia, established clinically • Deficits in two or more areas of cognition • Progressive worsening of memory & other cognitive function • No disturbance of consciousness • Onset between ages 40 and 90 • Absence of systemic disorders of brain diseases to account for the progressive deficits in memory & cognition Clinically Possible AD • Presence of variations in the onset, in the presentation, or in the clinical course • Presence of a second systemic or brain disorder sufficient to produce dementia, but which is not considered to be the cause of dementia

Neuropathology of AD & MCI
Anatomy of Temporal Lobe I
Anatomy of Temporal Lobe II
Memory Circuit
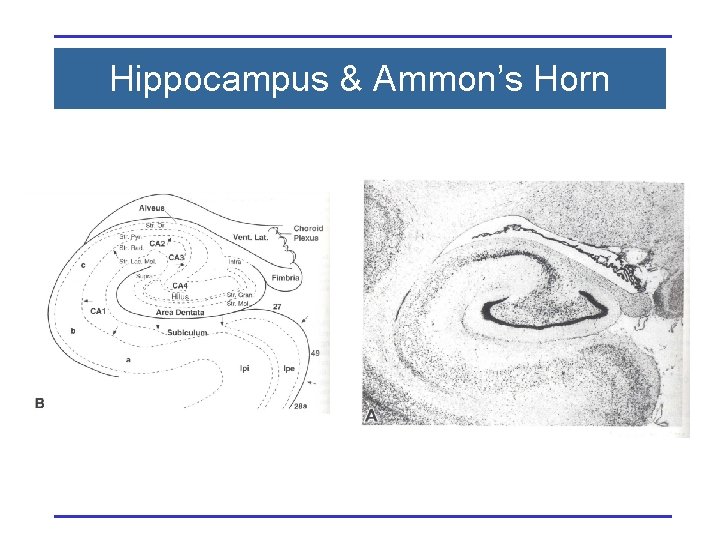
Hippocampus & Ammon’s Horn
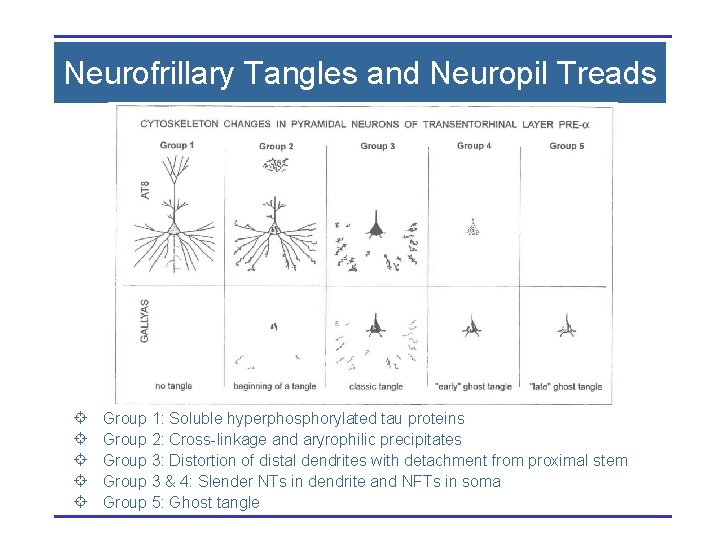
Neurofrillary Tangles and Neuropil Treads ± ± ± Group 1: Soluble hyperphosphorylated tau proteins Group 2: Cross-linkage and aryrophilic precipitates Group 3: Distortion of distal dendrites with detachment from proximal stem Group 3 & 4: Slender NTs in dendrite and NFTs in soma Group 5: Ghost tangle
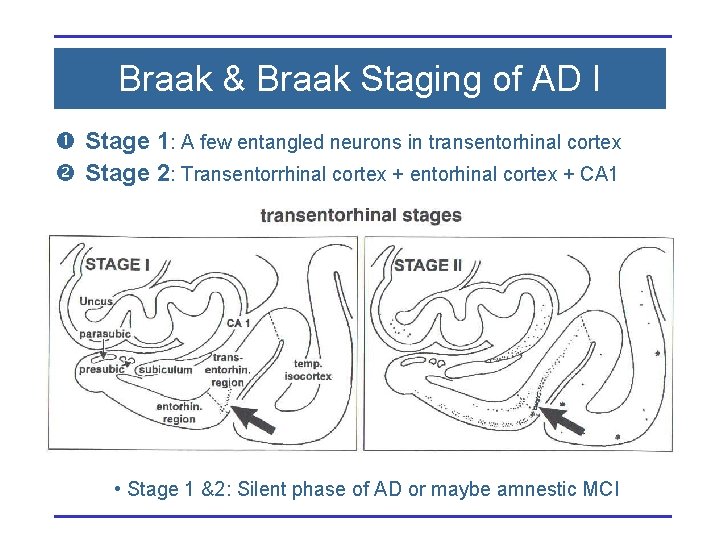
Braak & Braak Staging of AD I Stage 1: A few entangled neurons in transentorhinal cortex Stage 2: Transentorrhinal cortex + entorhinal cortex + CA 1 • Stage 1 &2: Silent phase of AD or maybe amnestic MCI
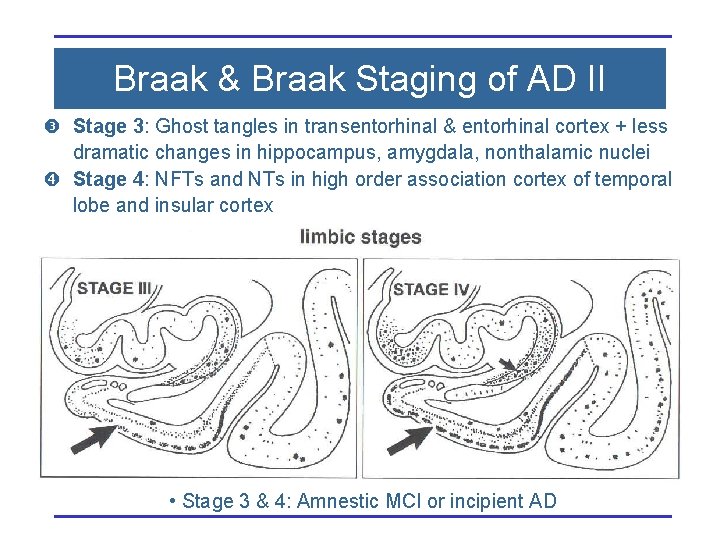
Braak & Braak Staging of AD II Stage 3: Ghost tangles in transentorhinal & entorhinal cortex + less dramatic changes in hippocampus, amygdala, nonthalamic nuclei Stage 4: NFTs and NTs in high order association cortex of temporal lobe and insular cortex • Stage 3 & 4: Amnestic MCI or incipient AD

Braak & Braak Staging of AD III Stage 5: Extensive devastation of neocortex except primary motor & sensory cortex and its first order association area Stage 6: Involvement of primary areas • Stage 5 & 6: Advanced AD

Neuropsychologic Assessment

Six Definitions of Normal Gaussian: the mean ± 2 standard deviations - this one assumes a normal distribution for all tests and results in all “abnormalities” having the same frequency Percentile: within the range, say of 5 -95% - has the same defect as the Gaussian definition Culturally desirable: when “normal” is that which is preferred by society, the role of medicine gets confused. Risk factors: carrying no additional risk of disease Diagnostic: range of results beyond which target disorders become highly probable Therapeutic: range of results beyond which treatment does more good than harm

Models of Aging • Two models – “Senescence” or “Life span development” – Choices of models are not determined on an empiric basis but on philosophical decisions by researchers and clinicians • “ Successful aging” – Isolate the effect of the passage of time alone on individuals by excluding the persons with common medical illness or medications that have the potential to affect cognition – Often described as “super normals” • “Typical Aging” – Do not exclude the persons with common illness and common medications • The objective determines the choice of definition of normal aging.

Normal Aging & Successful Aging • As disabilities associated with aging become treatable, they may be considered pathologic conditions rather than inevitable features of the physiological aging process. • Previous attempts at characterizing cognitive changes intrinsic to normal aging have produced several terms, such as benign senescent forgetfulness, age-associated memory impairment, and age-associated cognitive decline. • These terms are generally meant to reflect the extremes of normal aging rather than to describe a precursor of pathologic aging. • Some investigations of these concepts demonstrate increased conversion rate in this subjects compared to healthy ones.

Criteria for AAMI Inclusion Criteria Adults at least 50 years of age Complaints of memory loss; gradual onset Memory test performance that is at least 1 SD below the mean established for young adults (20 -35 years) Evidence of adequate intellectual function Absence of dementia ( 24 on MMSE) Exclusion Criteria Evidence of delirium, confusion, or other disturbance of consciousness Evidence of any neurological or medical disorders that could produce cognitive impairment History of infective or inflammatory brain disease History of repeated minor head injury or single injury with LOC lasting more than 1 hr Current major psychiatric illness or psychiatric drug use during the month before psychiatric testing Current Dx or Hx of alcohol or drug dependence

Cognitive Impairment No Dementia (CIND) • The Canadian Study of Health and Aging (Lancet 1997; 349: 1793 -1796) • Dx of CIND is based on exclusion of dementia and the presence of various categories of impairment identified in the clinical examination and in a battery of neuropsychological tests. • Subcategories of CIND – delirium – chronic alcohol and drug use – depression – psychiatric illness – mental retardation – Circumscribed memory impairment referred to as AAMI – "other" cognitive impairment by physicians

Typical Aging • Neuropsychologic characteristics of typical aging – Loss in efficiency in the acquisition of new information – Loss of processing speed – Loss of cognitive flexibility – Loss of working memory efficiency • Identification of cognitive domains that are relatively spared, even in typical aging, but may be associated with neurological diseases – Access to remotely learned information – Integrity of semantic network – Retention of well-encoded new information

Subjective memory complaints & objective memory function • Subjective memory complaint is included in the criteria for AAMI, & MCI. • Subjective memory complaints – Poorly related to the presence of poor memory performance and longitudinal memory score changes – Well related to affective status or general self-efficacy assessment • Informant-based reports of memory decline is well related to objective memory impairment. – 1/3 ~ 1/2 of dementia patients live alone at onset or diagnosis of cognitive dementia (Smith GE et al. , 2000) – Therefore, objective assessment of cognitive function, esp. memory function is a crucial step in the dx of AAMI & MCI.

MOANS (Mayo Older American Normative Studies) • Establish neuropsychological norms for assessment of older adults • Typical aging models, most useful for routine clinical practice • The definition of “normal” persons aged 55 and over – No active psychiatric or neurological disease – No complaint of cognitive difficulty during hx taking & S/R, & no finding on P/E suggesting disorders to affect cognition – No psychotropic medication use in amounts to compromise cognition or suggest neuropsychiatric disorders – Independent community dwelling status – Prior hx of disorders (e. g. , alcohol abuse) with potential to affect cognition were not automatically excluded as long as the disorder was not active & there was recovery without apparent cognitive residual – Chronic medical illness not exclusionary as long as condition was not reported by physician to compromise cognition

Lessons from MOANS • Co-norming • Comparing cognitive domains – A statistically significant difference between two different domains • Longitudinal assessment – Practice effect only between the first and second assessment – Different cognitive domains have different stabilities over time – Rather large change-score size • Group versus individual statistics • Disease prevalence, sensitivity, specificity, PPV, & NPV • If normative cohort has a cumulative incidence of dementia that is equal to or less than that in general population, the biasing of norms by the presence of incident dementia cases is minimal.

Comparative Norms vs. Diagnostic Norms • Norms should provide information about the likelihood, or probability, that an individual has MCI. • Comparative norms & Diagnostic norms • Information used by comparative & diagnostic norms • Age-corrected vs. age-weighted – Meiran N, et al. (1996): 25 on MMSE, 11% probability of AD in ages between 65 & 74, but 61% in ages between 74 & 85

Cognitive aging and MCI • Einstein Aging Study (Sliwinski M, et al. , 1997) – Memory impairment in AD differs not only in severity but also in kind from age-related memory changes. – After control for attentional deficits, inefficient strategy use, and production deficits, many of the memory problems on controlled learning procedures in healthy older adults disappeared but those in demented adults remained. • A growing body of research suggests that age-related memory differences may be a result of inefficient information processing, caused by generalized cognitive slowing. • Memory decline in healthy aging may be “secondary” to general declines in processing speed and efficiency, but memory impairment in dementia in primary.
Neuroimaging

Diagnosis and characterization • Jack CR, et al. (1997) – Hippocampal volume measurements can discriminate between control subjects and AD patients with very mild disease. – 97. 2% of all CDR 0. 5 AD patients had hippocampal volumes below the 50 th percentile of normal. • Xu Y, et al. (2000) – Changes in the entorhinal cortex may enhance the sensitivity of an early diagnosis of MCI or AD. – However, a recent study comparing entorhinal measures with hippocampal volumes indicates that the entorhinal measurement may not always be superior.
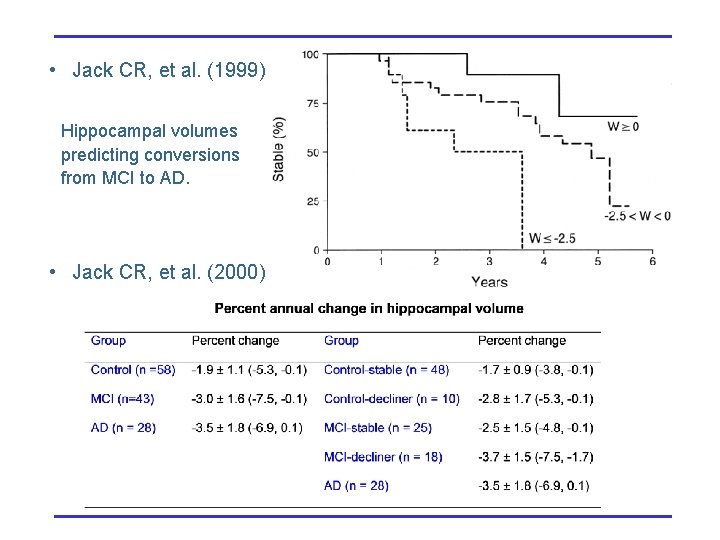
• Jack CR, et al. (1999) Hippocampal volumes predicting conversions from MCI to AD. • Jack CR, et al. (2000)

• MRS study (Kantarci K, et al. , 2000) – NAA/creatine ratios were significantly lower in AD than both MCI and control in left superior temporal and posterior cingluate VOI. – MI(myoinositol)/Cr ratios were significantly higher in both MCI and AD than in control in posterior cingulate VOI. • DWI study (Kantarci K, et al. 2001) – Higher levels of hippocampal ADC in both MCI and AD than in control • Functional MRI – A few studies in AD – Decreased functional activation in medial temporal lobe in AD compared to control • Typical aging versus successful aging
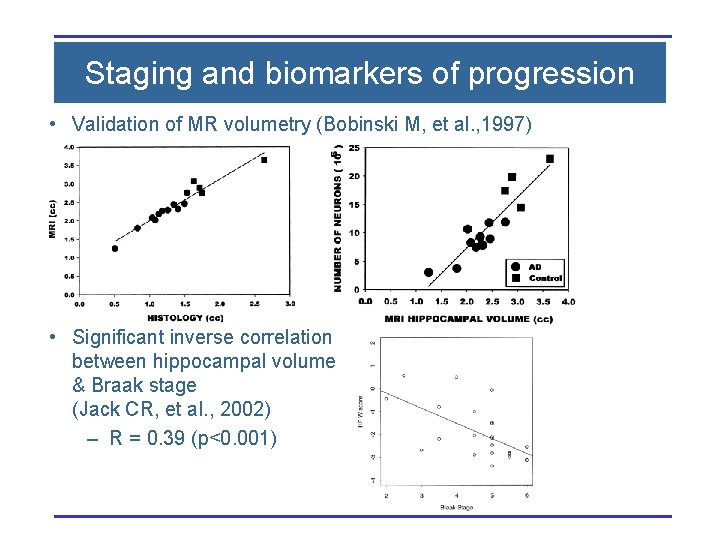
Staging and biomarkers of progression • Validation of MR volumetry (Bobinski M, et al. , 1997) • Significant inverse correlation between hippocampal volume & Braak stage (Jack CR, et al. , 2002) – R = 0. 39 (p<0. 001)

Biomarkers

• Evidences in AD – CSF tau concentrations are significantly elevated in AD – A -42 levels are significantly decreased in AD – Tau levels are also significantly increased in mildly demented subjects with AD (Mini-Mental State Examination score of 25), yielding sensitivity and specificity values approaching 0. 90. • Sunderland T, et al. (1999) – Nearly all subjects with MCI who convert to AD have high CSF tau values, but in nonprogressive MCI, tau levels remain low. – CSF tau levels do not seem to increase during the course of AD. --> This suggests that the measurement of CSF tau might be used effectively for identifying incipient AD among patients diagnosed clinically as having MCI.

Treatment - MCI trials
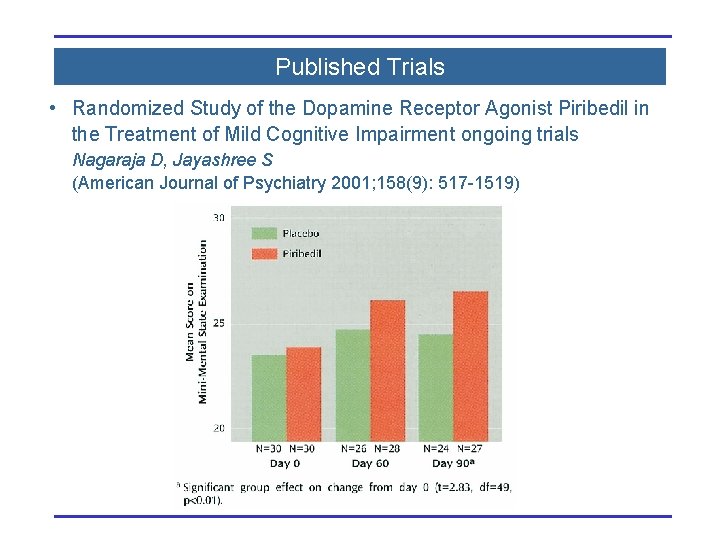
Published Trials • Randomized Study of the Dopamine Receptor Agonist Piribedil in the Treatment of Mild Cognitive Impairment ongoing trials Nagaraja D, Jayashree S (American Journal of Psychiatry 2001; 158(9): 517 -1519)
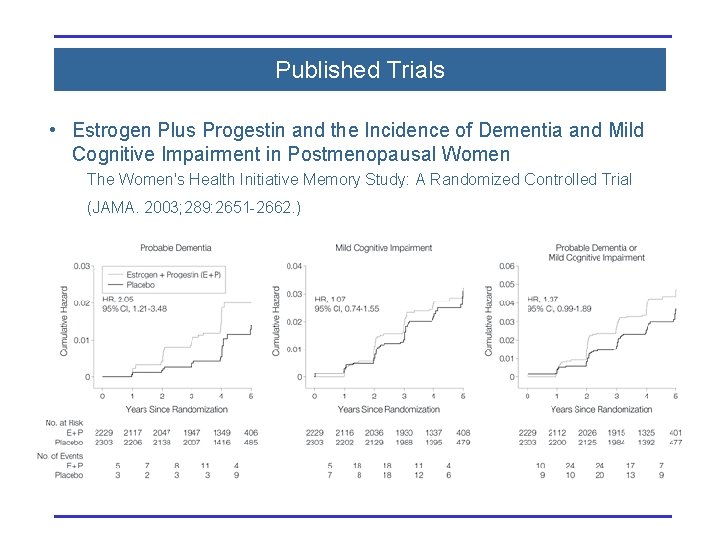
Published Trials • Estrogen Plus Progestin and the Incidence of Dementia and Mild Cognitive Impairment in Postmenopausal Women The Women's Health Initiative Memory Study: A Randomized Controlled Trial (JAMA. 2003; 289: 2651 -2662. )
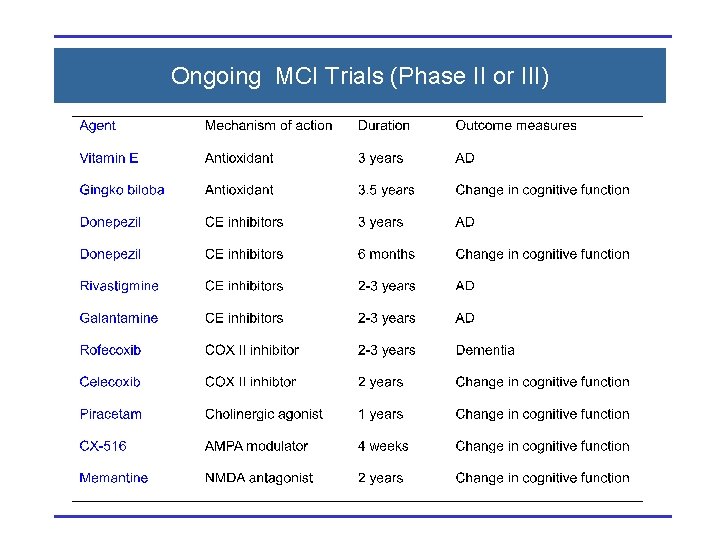
Ongoing MCI Trials (Phase II or III)

It’s never to late to study.
- Slides: 64