20 d B Bell 20 Hz200 Hz Diaphragm







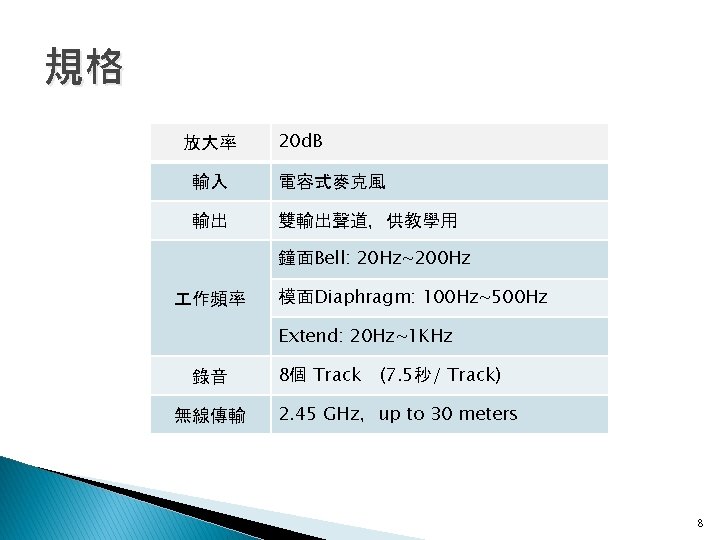
規格 放大率 20 d. B 輸入 電容式麥克風 輸出 雙輸出聲道,供教學用 鐘面Bell: 20 Hz~200 Hz 作頻率 模面Diaphragm: 100 Hz~500 Hz Extend: 20 Hz~1 KHz 錄音 無線傳輸 8個 Track (7. 5秒/ Track) 2. 45 GHz,up to 30 meters 8

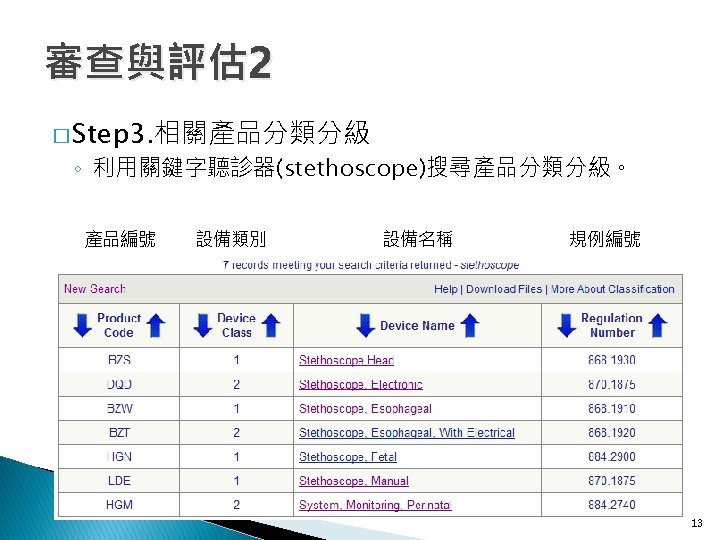
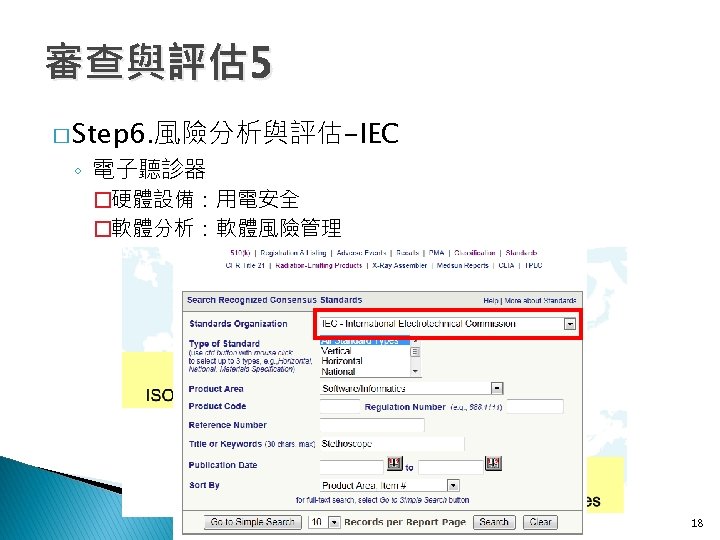
審查與評估 2 � Step 3. 相關產品分類分級 ◦ 進到FDA網站->Medical Devices->FDA Standards Database 11
審查與評估 2 � Step 3. 相關產品分類分級 ◦ FDA Recognized Consensus Standards Database ◦ Product Classification 12
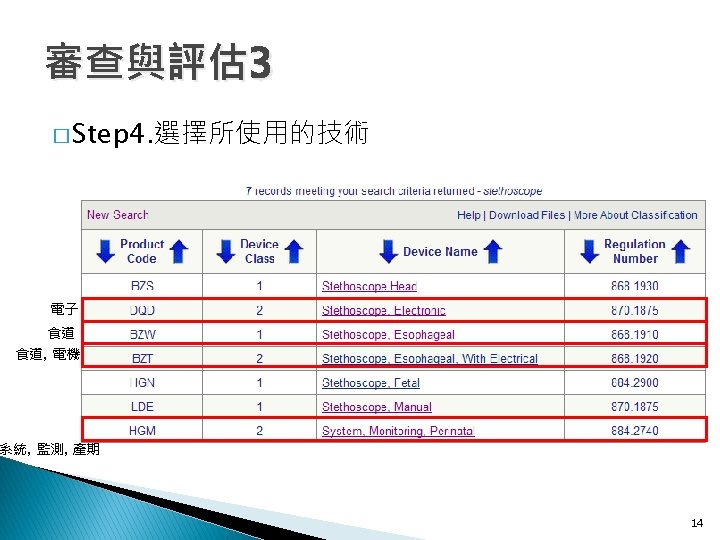
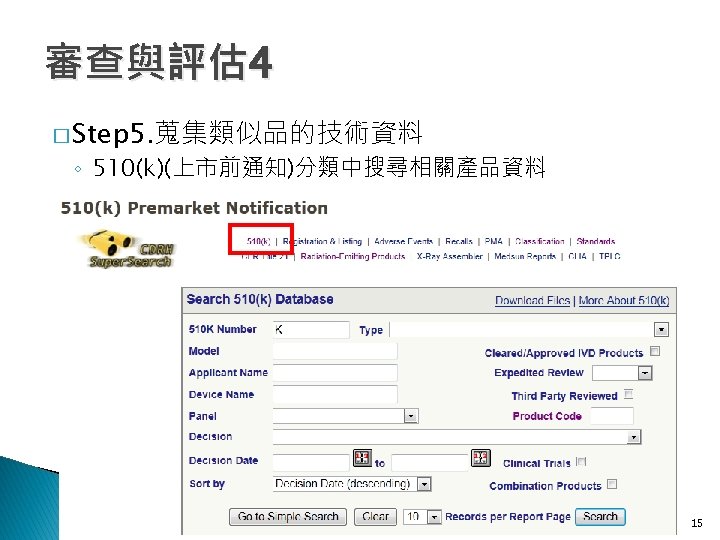

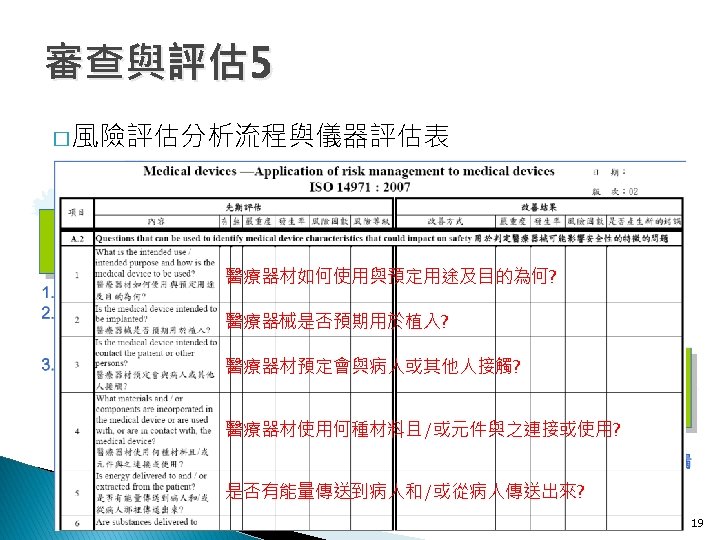
審查與評估 4 � Step 5. 蒐集類似品的技術資料 ◦ Product Code: 1. DQD 2. BZW 3. BZT 4. HGM 1. DQD 2. BZW 3. BZT 16
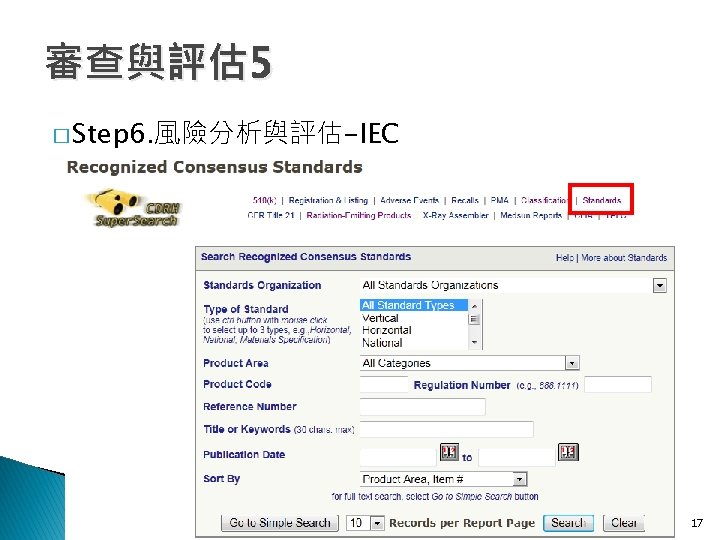
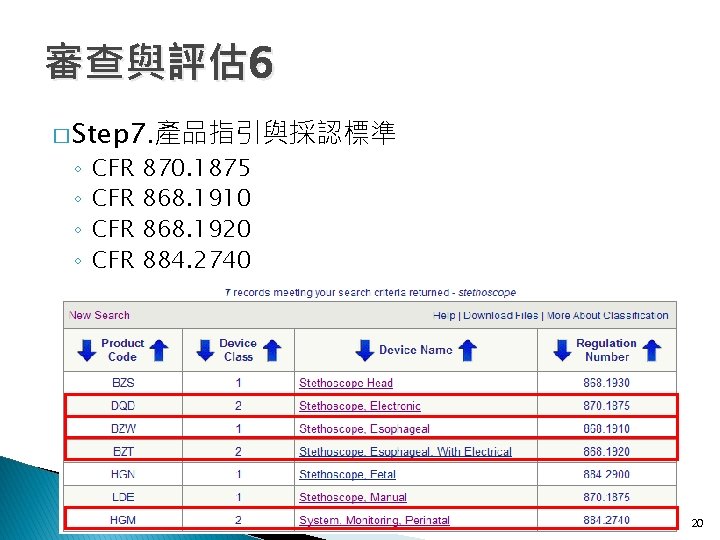
審查與評估 6 � Step 7. 產品指引與採認標準 ◦ ◦ CFR CFR 870. 1875 868. 1910 868. 1920 884. 2740 20

審查與評估 6 � Step 7. 產品指引與採認標準 CFR 870. 1875: 884. 2740: 分類 2級 (a)Manual stethoscope (a)Identification. (1)Identification. manual stethoscope is used a CFR : 分類A 1級 A 868. 1910 perinatal monitoring system is a device to mechanical devicethe used to projectbetween the sounds (a)Identification. show graphically relationship maternal CFR 868. 1920 : 分類 2級 associated with theheart, rate arteries, and veins and An esophageal stethoscope isby a nonpowered device labor and the fetal means of combining (a)Identification. other that isinternal insertedorgans. into a patient's esophagus to enable and coordinating uterine contraction and fetal heart An esophageal stethoscope with electrical conductors (2)Classification. Class I (general controls). The device the user to listen to heart and breath monitors with appropriate displays ofsounds. the well-being is a device from that is inserted into notification the esophagus to listen is the premarket procedures (b)Classification. of exempt the fetus during pregnancy, labor, and delivery. to a patient's heart and breath sounds and to monitor in subpart E of part of. The this chapter subject to the Class I (general controls). device is exempt from This generic type of 807 device may include any of the electrophysiological signals. The device may also limitations in 870. 9. the premarket notification procedures in 884. 2660, subpart E of devices subject to 884. 2600, 884. 2640, incorporate a thermistor for temperature (b)Electronic part 807 ofstethoscope this chapter subject to 868. 9. 884. 2675, 884. 2700, and 884. 2720. This generic measurement. (1)Identification. Aninclude electronic is an type of device may the stethoscope following accessories: (b)Classification. electrically amplifiedsystem deviceand used to project the Central monitoring remote repeaters, Class II (performance standards). sounds associated the equipment, heart, arteries, and veins signal analysis and with display patient and other internal organs. equipment supports, and component parts. (2)Classification. Class II (performance standards). (b)Classification. Class II (performance standards). 21

參考文獻 � � � 業技術研究院 - 電子聽診器 http: //hi. ctust. edu. tw/mediawiki/index. php/%E 9%9 B%BB%E 5%AD%90%E 8%81%BD%E 8%A 8 %BA%E 5%99%A 8#. E 9. 9 B. BB. E 5. AD. 90. E 8. 81. BD. E 8. A 8. BA. E 5. 99. A 8. E 5. 84. 80. E 5. 99. A 8. E 4. BB. 8 B. E 7. B 4. B 9 U. S. G. P. O http: //www. gpo. gov/fdsys/search/pagedetails. action; jsessionid=80 e 40 f 629599 ddc 7 df 4 e 5 a 696 a 49446 a 962 c 338 c 713 f 69 eb 0 fef 97931 be 7 e 796. e 38 Nbxu. Qa. Ni. Kb. O 0 Ma 3 y. Qb. Nm. L bxy. Me 0? browse. Path=Title+21%2 FChapter+I%2 FPart+868%2 FSubpart+B%2 FSec. +868. 19 20&granule. Id=CFR-1996 -title 21 -vol 8 -sec 868 -1920&package. Id=CFR-1996 -title 21 vol 8&collapse=true&from. Browse=true IEC http: //www. iec. ch/ 理 科技顧問公司 – 風險分析與管理介紹 業技術研究院量測技術發展中心 李子偉 - 醫療器材上市前審查 22
- Slides: 22