20 5 Water as a nucleophile Is water



















- Slides: 38

20. 5 Water as a nucleophile • Is water generally a strong or weak nucleophile? • Show a generic mechanism for water attacking an aldehyde or ketone Predict whether the nucleophilic attack is product favored or reactant favored. WHY? • • Would the presence of an acid improve the reaction? Copyright 2012 John Wiley & Sons, Inc. 20 -1

20. 5 Water as a nucleophile • If water were to attack the carbonyl, what likely mechanism steps would follow? • Will the overall process be fast or slow? • Will the overall process be product or reactant favored? See next slide for examples Copyright 2012 John Wiley & Sons, Inc. 20 -2

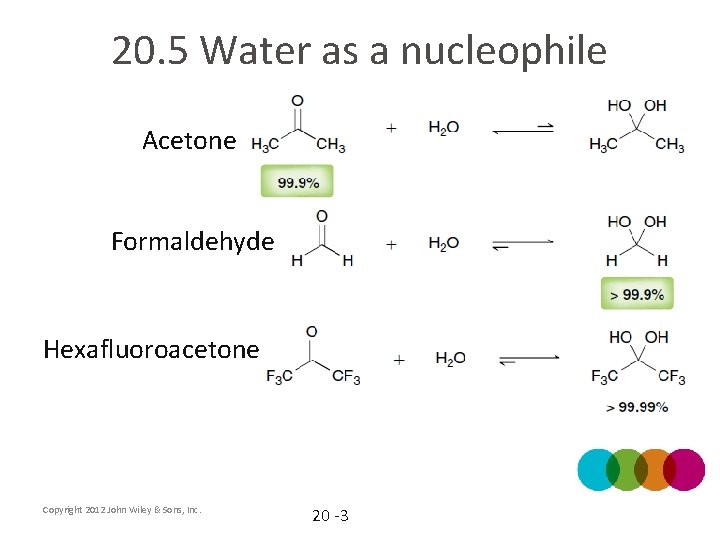
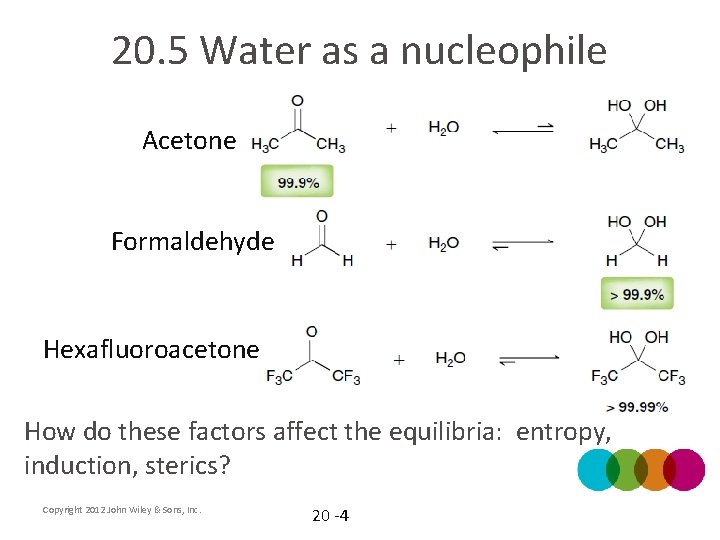
20. 5 Water as a nucleophile Acetone Formaldehyde Hexafluoroacetone Copyright 2012 John Wiley & Sons, Inc. 20 -3

20. 5 Water as a nucleophile Acetone Formaldehyde Hexafluoroacetone How do these factors affect the equilibria: entropy, induction, sterics? Copyright 2012 John Wiley & Sons, Inc. 20 -4

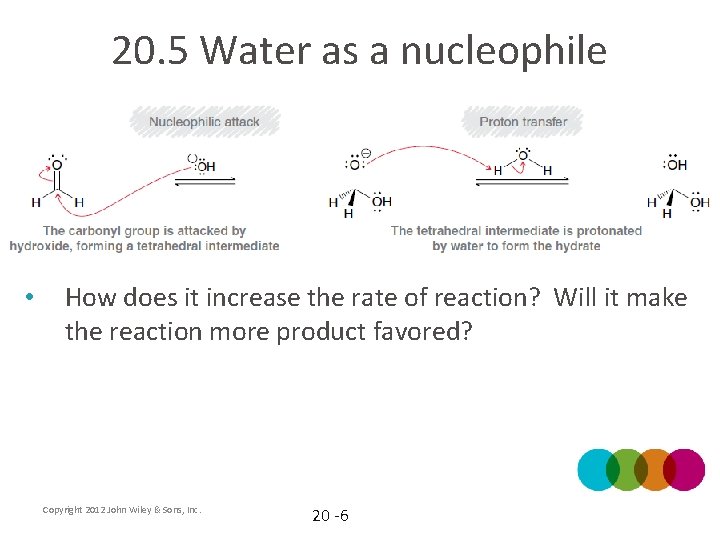
20. 5 Water as a nucleophile • To avoid the unstable intermediate with two formal charges, the reaction can be catalyzed by a base (mechanism 20. 3) Copyright 2012 John Wiley & Sons, Inc. 20 -5

20. 5 Water as a nucleophile • How does it increase the rate of reaction? Will it make the reaction more product favored? Copyright 2012 John Wiley & Sons, Inc. 20 -6

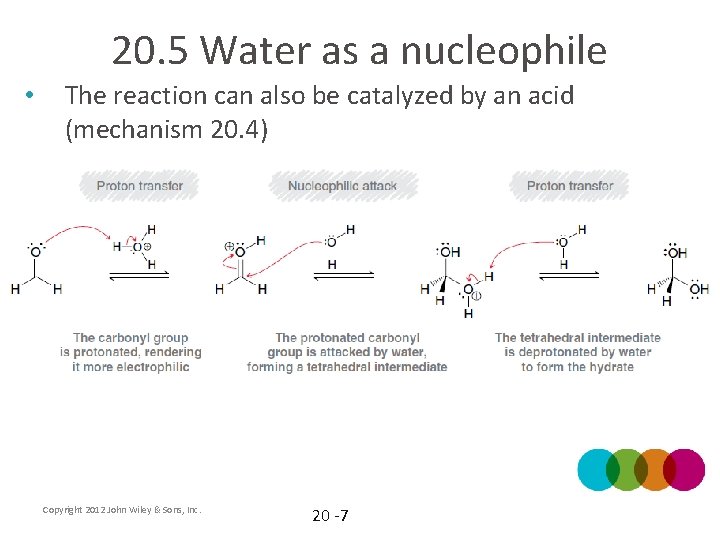
20. 5 Water as a nucleophile • The reaction can also be catalyzed by an acid (mechanism 20. 4) Copyright 2012 John Wiley & Sons, Inc. 20 -7

20. 5 Water as a nucleophile • How does the acid increase the rate of reaction? Will it make the reaction more product favored? Copyright 2012 John Wiley & Sons, Inc. 20 -8

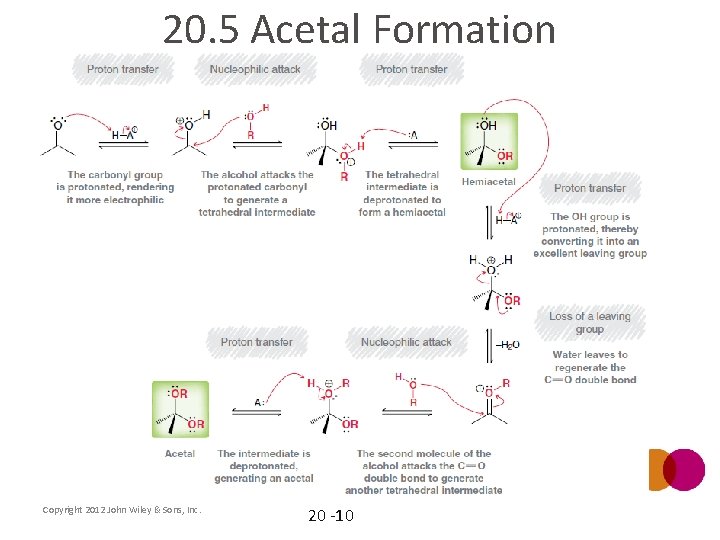
20. 5 Acetal Formation • An alcohol acts as the nucleophile instead of water • Notice that the reaction is under equilibrium and that it is acid catalyzed Analyze the complete mechanism (mechanism 20. 5) on the next slide Analyze how the acid allows the reaction to proceed through lower energy intermediates • • Copyright 2012 John Wiley & Sons, Inc. 20 -9

20. 5 Acetal Formation Copyright 2012 John Wiley & Sons, Inc. 20 -10

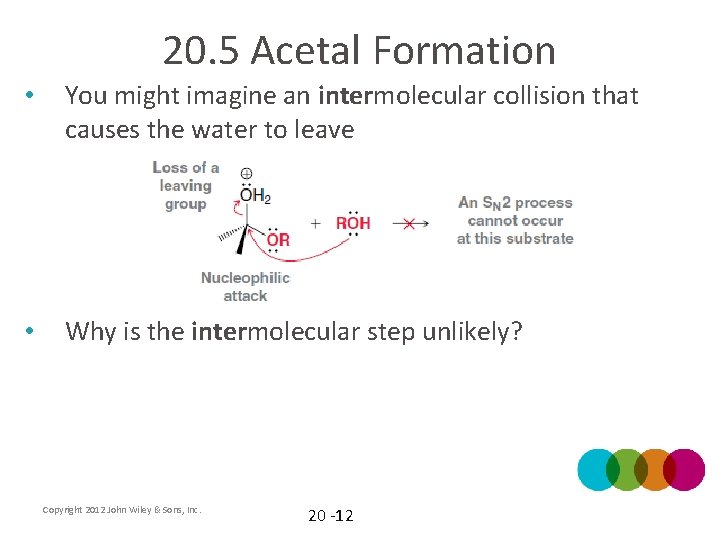
20. 5 Acetal Formation • After the hemiacetal is protonated in mechanism 20. 5, the water leaving group leaves. Why is the water leaving group pushed out intramolecularly? Copyright 2012 John Wiley & Sons, Inc. 20 -11

20. 5 Acetal Formation • You might imagine an intermolecular collision that causes the water to leave • Why is the intermolecular step unlikely? Copyright 2012 John Wiley & Sons, Inc. 20 -12

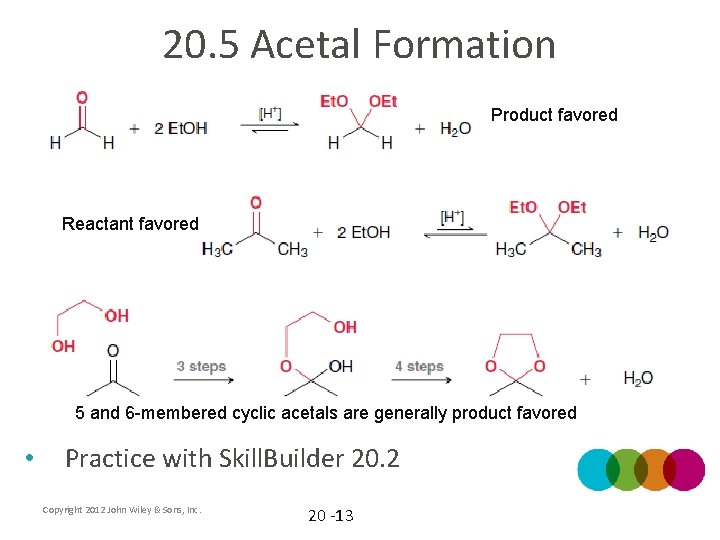
20. 5 Acetal Formation Product favored Reactant favored 5 and 6 -membered cyclic acetals are generally product favored • Practice with Skill. Builder 20. 2 Copyright 2012 John Wiley & Sons, Inc. 20 -13

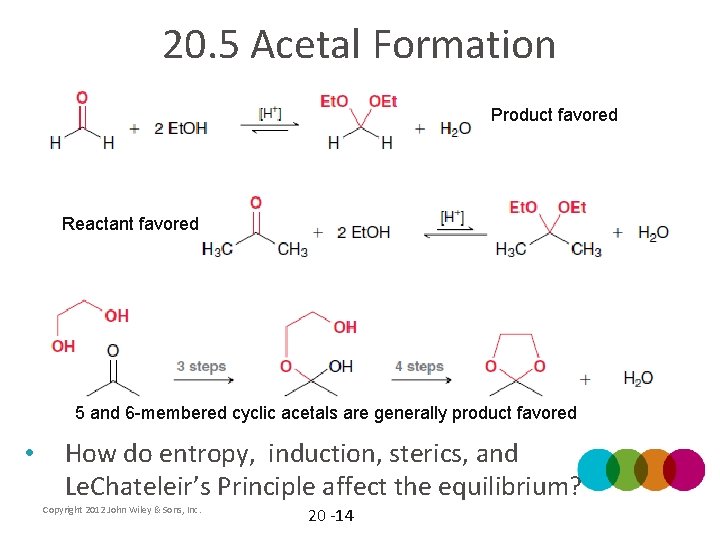
20. 5 Acetal Formation Product favored Reactant favored 5 and 6 -membered cyclic acetals are generally product favored • How do entropy, induction, sterics, and Le. Chateleir’s Principle affect the equilibrium? Copyright 2012 John Wiley & Sons, Inc. 20 -14

20. 5 Acetal Equilibrium control • • Acetals can be attached and removed fairly easily Example: • Both the forward and reverse reactions are acid catalyzed How does the presence of water affect which side the equilibrium will favor? • Copyright 2012 John Wiley & Sons, Inc. 20 -15

20. 5 Acetal Protecting Groups • • We can use an acetal to selectively protect an aldehyde or ketone from reacting in the presence of other electrophiles Fill in necessary reagents or intermediates Copyright 2012 John Wiley & Sons, Inc. 20 -16

20. 5 Acetal Protecting Groups • Fill in necessary reagents or intermediates Copyright 2012 John Wiley & Sons, Inc. 20 -17

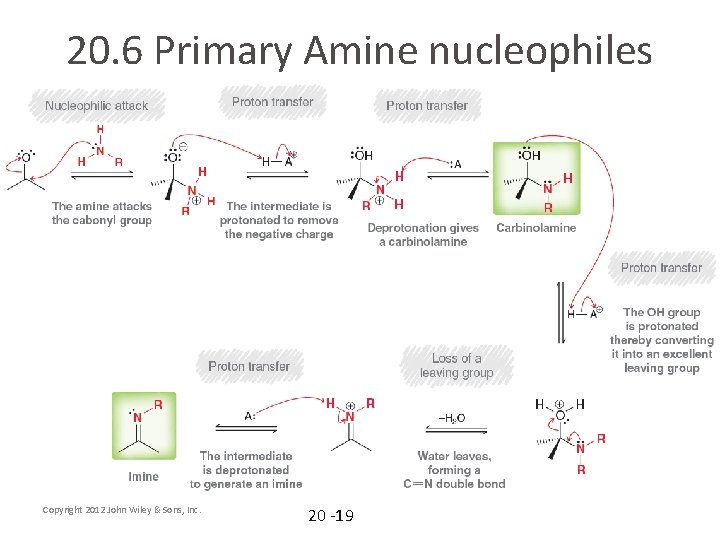
20. 6 Primary Amine nucleophiles • • • As a nucleophile, are amines stronger or weaker than water? If you want an amine to attack a carbonyl carbon, will a catalyst be necessary? Will an acid (H+) or a base (OH-) catalyst be most likely to work? WHY? What will the product most likely look like? Keep in mind that entropy disfavors processes in which two molecules combine to form one Analyze the complete mechanism (mechanism 20. 6) on the next slide Copyright 2012 John Wiley & Sons, Inc. 20 -18

20. 6 Primary Amine nucleophiles Copyright 2012 John Wiley & Sons, Inc. 20 -19

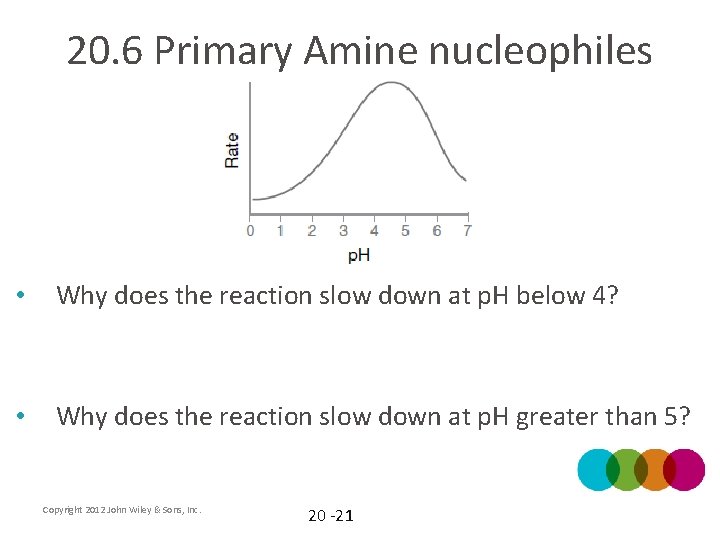
20. 6 Primary Amine nucleophiles • The mechanism requires an acid catalyst. Note that the optimal p. H to achieve a fast reaction is around 4 or 5 • Practice with Skill. Builder 20. 3 Copyright 2012 John Wiley & Sons, Inc. 20 -20

20. 6 Primary Amine nucleophiles • Why does the reaction slow down at p. H below 4? • Why does the reaction slow down at p. H greater than 5? Copyright 2012 John Wiley & Sons, Inc. 20 -21

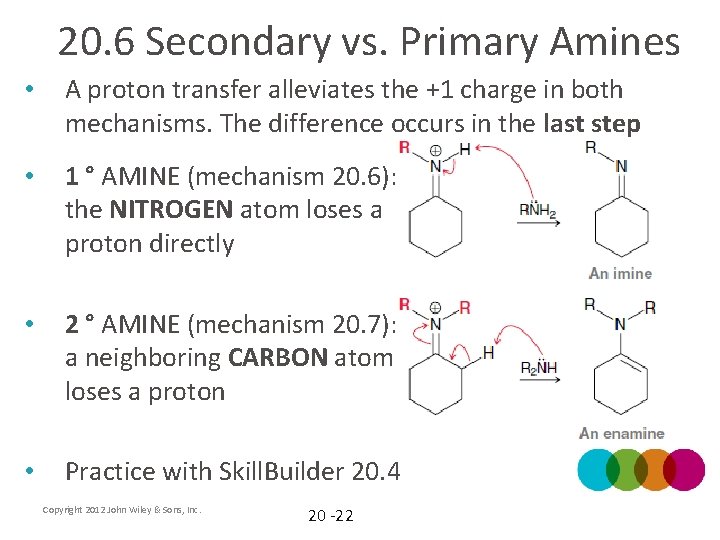
20. 6 Secondary vs. Primary Amines • A proton transfer alleviates the +1 charge in both mechanisms. The difference occurs in the last step • 1 ° AMINE (mechanism 20. 6): the NITROGEN atom loses a proton directly • 2 ° AMINE (mechanism 20. 7): a neighboring CARBON atom loses a proton • Practice with Skill. Builder 20. 4 Copyright 2012 John Wiley & Sons, Inc. 20 -22

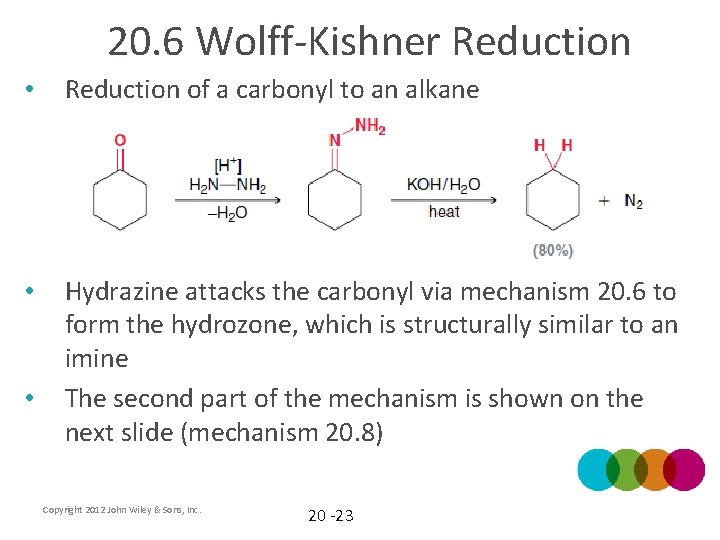
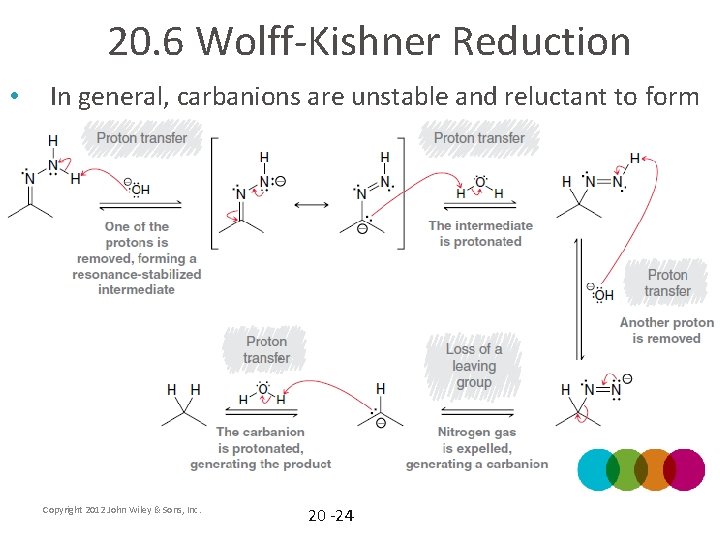
20. 6 Wolff-Kishner Reduction • Reduction of a carbonyl to an alkane • Hydrazine attacks the carbonyl via mechanism 20. 6 to form the hydrozone, which is structurally similar to an imine The second part of the mechanism is shown on the next slide (mechanism 20. 8) • Copyright 2012 John Wiley & Sons, Inc. 20 -23

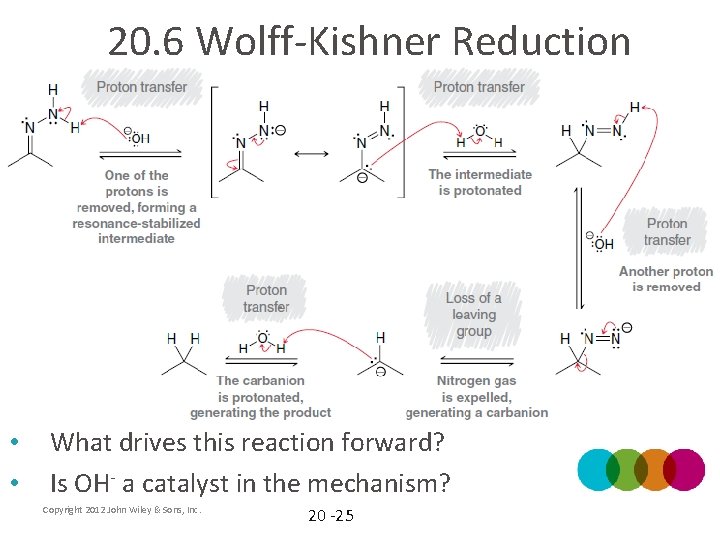
20. 6 Wolff-Kishner Reduction • In general, carbanions are unstable and reluctant to form Copyright 2012 John Wiley & Sons, Inc. 20 -24

20. 6 Wolff-Kishner Reduction • • What drives this reaction forward? Is OH- a catalyst in the mechanism? Copyright 2012 John Wiley & Sons, Inc. 20 -25

20. 6 Wolff-Kishner Reduction • Note the many similarities between the acid catalyzed mechanisms we have discussed Carbonyl is protonated first – • • – – – • Makes the carbonyl more electrophilic Avoids negative formal charge on the intermediate Avoid high energy intermediate with two formal charges. Acid protonates leaving group so that it is stable and neutral upon leaving Last step of mechanism involves a proton transfer forming a neutral product Overall: Under acidic condition, reaction species should either be neutral or have a +1 formal charge Copyright 2012 John Wiley & Sons, Inc. 20 -26

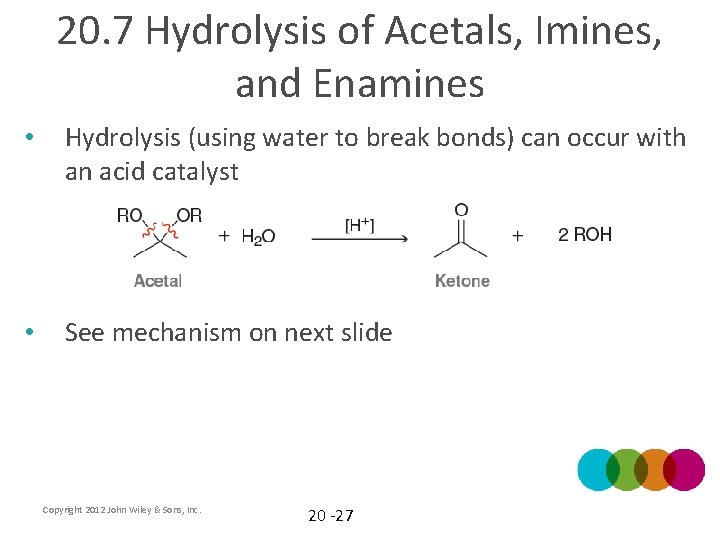
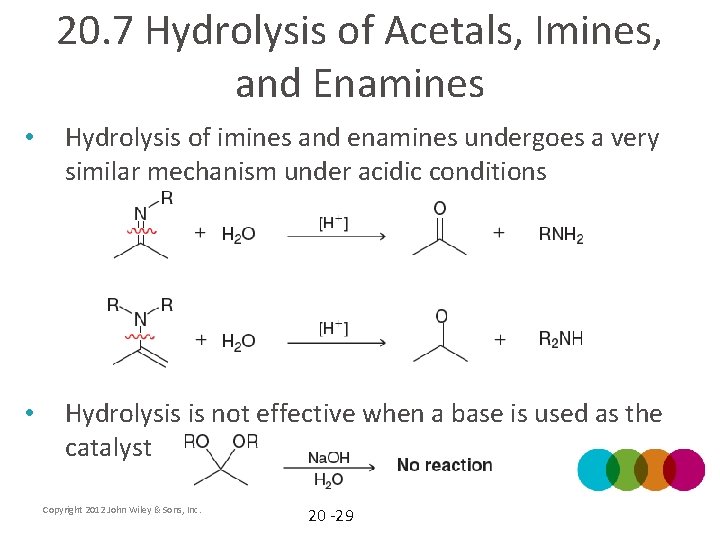
20. 7 Hydrolysis of Acetals, Imines, and Enamines • Hydrolysis (using water to break bonds) can occur with an acid catalyst • See mechanism on next slide Copyright 2012 John Wiley & Sons, Inc. 20 -27

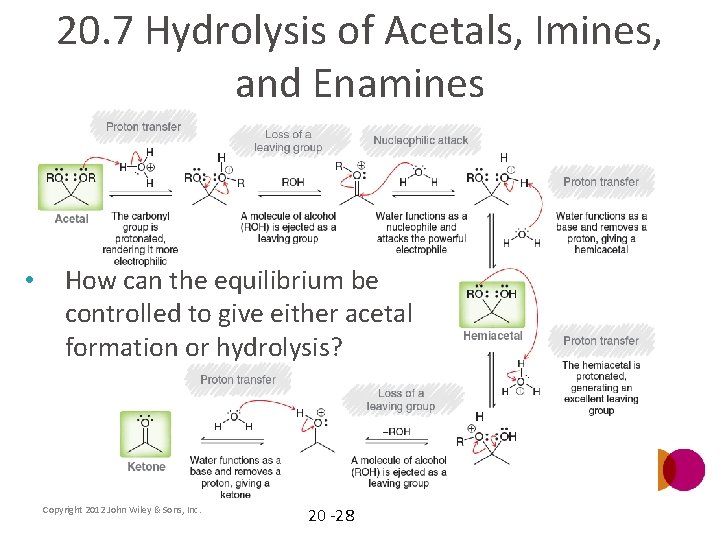
20. 7 Hydrolysis of Acetals, Imines, and Enamines • How can the equilibrium be controlled to give either acetal formation or hydrolysis? Copyright 2012 John Wiley & Sons, Inc. 20 -28

20. 7 Hydrolysis of Acetals, Imines, and Enamines • Hydrolysis of imines and enamines undergoes a very similar mechanism under acidic conditions • Hydrolysis is not effective when a base is used as the catalyst Copyright 2012 John Wiley & Sons, Inc. 20 -29

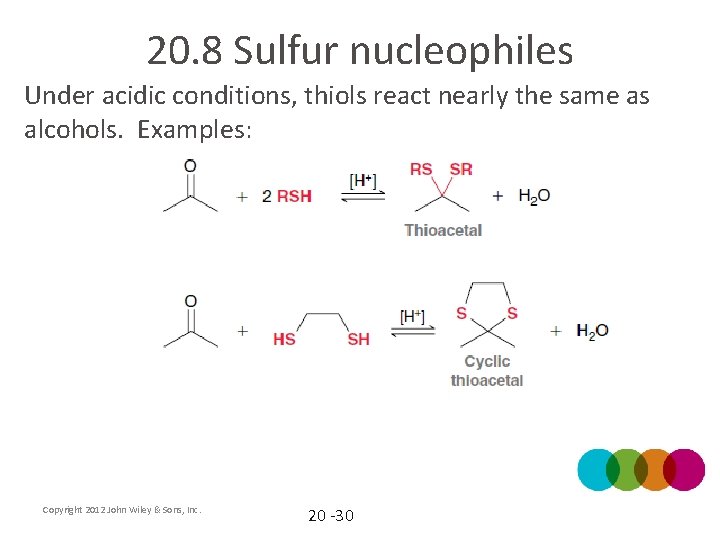
20. 8 Sulfur nucleophiles Under acidic conditions, thiols react nearly the same as alcohols. Examples: Copyright 2012 John Wiley & Sons, Inc. 20 -30

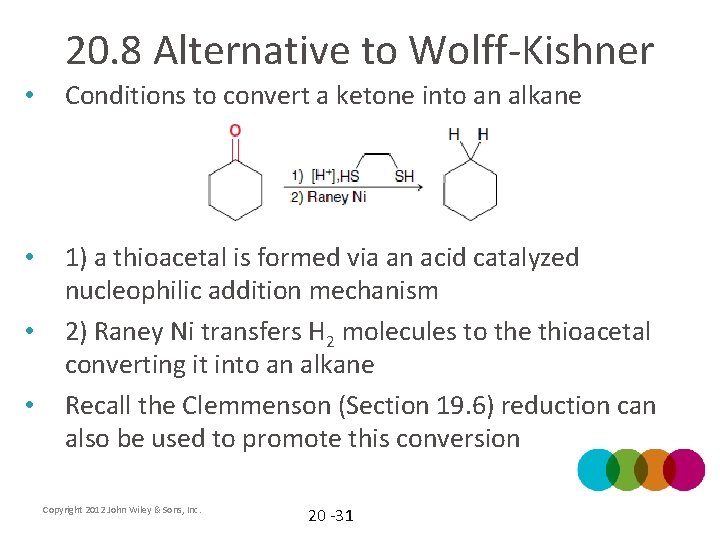
20. 8 Alternative to Wolff-Kishner • Conditions to convert a ketone into an alkane • 1) a thioacetal is formed via an acid catalyzed nucleophilic addition mechanism 2) Raney Ni transfers H 2 molecules to the thioacetal converting it into an alkane Recall the Clemmenson (Section 19. 6) reduction can also be used to promote this conversion • • Copyright 2012 John Wiley & Sons, Inc. 20 -31

20. 9 Hydrogen nucleophiles • • • We rarely see hydrogen acting as a nucleophile. WHY? What role does hydrogen normally play in mechanisms? To be a nucleophile, hydrogen must have a pair of electrons. H: 1 - is called hydride Reagents that produce hydride ions include Li. Al. H 4 and Na. BH 4. Hydride will react readily with carbonyls Copyright 2012 John Wiley & Sons, Inc. 20 -32

20. 9 Hydrogen nucleophiles • • • Identify the nucleophile Will the reaction be more effective under acidic or under basic conditions? WHY? Show a complete mechanism (mechanism 20. 9) Analyze the reversibility or irreversibility of each step Describe necessary experimental conditions (why are there two steps in the reaction? ) Copyright 2012 John Wiley & Sons, Inc. 20 -33

20. 10 Carbon nucleophiles • • • Carbon doesn’t often act as a nucleophile. WHY? What role does carbon most often play in mechanisms? To be a nucleophile, carbon must have a pair of electrons it can use to attract an electrophile Examples: 1. A carbanion with a -1 charge and available pair of electrons. However, carbanions are relatively unstable and reluctant to form 2. A carbon attached to a very low electronegativity atom such as a Grignard. Analyze the electrostatics of the Grignard reagent Copyright 2012 John Wiley & Sons, Inc. 20 -34

20. 10 Grignard Example • • • Identify the nucleophile Will the reaction be more effective under acidic or under basic conditions? WHY? Show a complete mechanism (mechanism 20. 10). Three equivalents of the Grignard are necessary Analyze the reversibility or irreversibility of each step Describe necessary experimental conditions (why are there two steps in the reaction? ) Copyright 2012 John Wiley & Sons, Inc. 20 -35

Study Guide for Sections 20. 5 -20. 10 DAY 18, Terms to know: Sections 20. 5 -20. 10 hydrate, acetal, amine, imine, enamine, Wolff-Kishner, hydrolysis DAY 18, Specific outcomes and skills that may be tested on exam 3: Sections 20. 5 -20. 10 • Be able to recognize which nucleophiles are likely to react at carbonyl sites in a product favored equilibrium and which will give a reactant favored equilibrium • Be able to fully explain how adding an acid makes carbonyls more electrophilic • Be able to predict products and give a complete mechanism for both the base-catalyzed and acid-catalyzed hydration of ketones or aldehydes • Be able to predict products and give a complete mechanism for the acid-catalyzed formation of an acetal from ketones or aldehydes and be able to predict and explain why the reaction is either product or reactant favored using entropy, induction, sterics, and Le. Chateleir’s Principle arguments • Be able to describe ideal reaction conditions for the formation of an imine or for an enamine • Given an amine and a carbonyl, be able to give a complete mechanism and predict products from their reaction under acidic conditions • Be able to describe ideal reaction conditions a Wolff-Kishner reaction • Given a carbonyl and Wolff-Kishner conditions, be able to give a complete mechanism and predict products from their reaction • Be able to describe ideal reaction conditions for the hydrolysis of a hydrate, acetal, hemiacetal, imine, or enamine • Given a hydrate, acetal, hemiacetal, imine, or enamine, be able to give a complete mechanism and predict products from their hydrolysis reaction under dilute acidic conditions • Be able to describe ideal reaction conditions for the reduction of a carbonyl with hydride or Grignard reagents • Given a carbonyl and hydride or Grignard conditions, be able to give a complete mechanism and predict products from their reaction • Be able to solve synthesis problems using the reactions described in this section

Extra Practice Problems for Sections 20. 5 -20. 10 Complete these problems outside of class until you are confident you have learned the SKILLS in this section outlined on the study guide and we will review some of them next class period. 19. 35 19. 36 19. 37 19. 38 19. 40 19. 42 19. 55 19. 71 20. 2 20. 3 20. 44 20. 45 20. 47 20. 5 20. 6 20. 7 20. 8 20. 9 20. 10 20. 11 20. 12 20. 13 20. 14 20. 15 20. 16 20. 17 20. 18 20. 21 20. 22 20. 23 20. 24 20. 25 20. 26 20. 27 b 20. 29 20. 31 20. 33 20. 34

Prep for Day 19 Must Watch videos: https: //www. youtube. com/watch? v=Hl 9 -WQSq. Io. I (reactions of aldehydes and ketones, FLC) https: //www. youtube. com/watch? v=QTLtxo. Vmg. Ug (Wittig, Moore) https: //www. youtube. com/watch? v=7 s. Btelh. Lyvk (carboxylic acids, FLC) Other helpful videos: https: //www. youtube. com/watch? v=8 m. A 9 y 3 l 4 u. K 4 (reactions of carbonyls, UC-Irvine) start at 47 minutes https: //www. youtube. com/watch? v=Jbcmxu 5 VYY 8 (Baeyer-Villiger, AKlectrues) https: //www. youtube. com/watch? v=xg. Duzj 51 t. Ac (carboxylic acid naming and properties, Khan) https: //www. youtube. com/watch? v=i. LPo. Yn. SXGEI (carboxylic acid preparation, Roxi) https: //www. youtube. com/watch? v=h. Zq 7 v. Dvm. TZg (carboxylic acids, UC-Irvine) start at 25 minutes Read Sections 20. 10 -20. 13, 21. 1 -21. 4