2 Weak acid Strong base titration eg CH

2 - Weak acid - Strong base titration : eg. CH 3 COOH (p. Ka = 4. 74) and Na. OH We have sample of 100 ml 0. 1 N HAc and tirate against 0. 1 N Na. OH a. Before the titration p. H is due to the sample i. e. HAc (weak acid) therfore p. H = 1/2 p. Ka + 1/2 p. Ca = 2. 37 + 0. 5 = 2. 87 b- During titration eg. after adding 40 ml Na. OH HAc + Na. OH Na. Ac + H 2 O HAc will be present ( not completely neutralized) and Na. Ac i. e. weak acid and its salt, therfore p. H is calculated from eq. of acidic buffer p. H = p. Ka + log salt/ acid = 4. 74 + log 40/60 = 4. 53 c- At the equivalence point (end point) all HAc is neutralized and only Na. Ac is present , therfore p. H is calculated from: p. H = 1/2 p. Kw + 1/2 p. Ka -1/2 p. Cs (salt of weak acid and strong base) =7 + 2. 37 -1/2 (- log 100 x 0. 1 /200) = 8. 72 d- After end point the p. H is calculated from excess titrant i. e. Na. OH. N. B. The end point is at the alkaline side and abrupt change in the curve is from p. H 7 to p. H 11. Therfore M. O and M. R. indicator are not suitable. Therfore use Ph. ind or any ind of p. H range on the alkaline side.

3 - Strong acid - weak base titration : eg. NH 4 OH (p. Kb = 4. 74)and HCl We have sample 100 ml 0. 1 N NH 4 OH and titrate against 0. 1 NHCl a- Before titration p. H is due to the sample NH 4 OH p. H is calculated from eq. of weak base. p. H = p. Kw - 1/2 p. Kb -1/2 p. Cb = 14 – 2. 37 0. 5 =11. 13 b-During titration eg. After adding 90 ml HCl NH 4 OH + HCl NH 4 Cl + H 2 O NH 4 OH will be present(not completely neutralized) and NH 4 Cl i. e. weak baseand its salt. therfore p. H is calculated from eq. of basic buffer p. H = p. Kw -p. Kb - log salt/ base = 14 - 4. 74 - log 90/10 = 8. 31 c- At the equivalence point (end point) all NH 4 OH is neutralized and NH 4 Cl is only present , therfore p. H is calculated from : p. H = 1/2 p. Kw- 1/2 p. Kb + 1/2 p. Cs( salt of strong acid and weak base) =7 - 2. 37 + 1/2 (- log 100 x 0. 1 /200) = 5. 13 d-After the end point the p. H is calculated from excess titrant i. e. HCl N. B. The end point is at the acidic side = 5. 13. The abrupt change is from p. H 6 to 4 , Therfore Ph. indicator cannot be used. Use M. O. or M. R. or any indicator of p. H range at acidic side.

Applications 1 - Direct titration methods : Direct titration is useful for : A- Strong acid B- Strong base C- Weak acid or base if Ka and Kb not less than 10 -7 Determination of acids : 1 - Strong acids can be titrated against strong alkali using Ph. or M. O. On titrating weak acid only Ph. is suitable. 2 - Acids like benzoic acid , salicylic acid which are not insoluble in water are dissolved in neutral ethanol then add water and titrate against Na. OH using Ph. as indicator. 3 - Boric acid: weak acid is a monobasic acid i. e. release 1 H+ , can be titrated against Na. OH only after potentiation by adding any poly hydroxy compound eg. glycerol using Ph. as indicator. Determination of bases : 1 - Strong base can be titrated against strong acids using M. O. or Ph. For weak bases we use M. O. indicator or any indicator of p. H range on the acidic side.
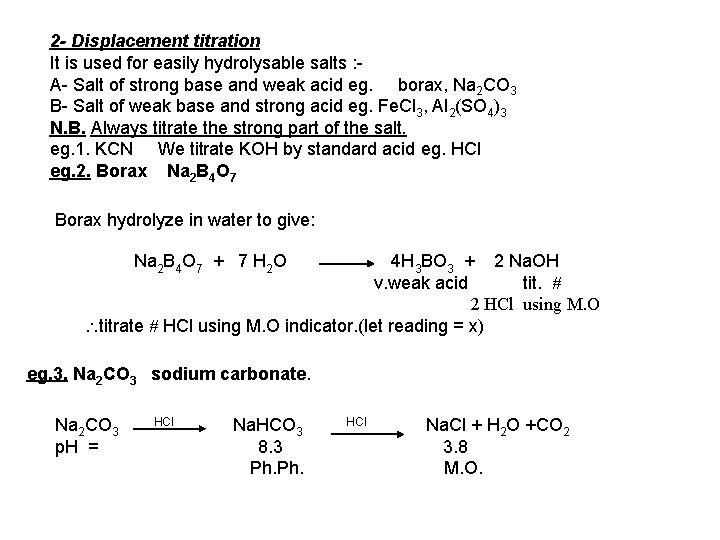
2 - Displacement titration It is used for easily hydrolysable salts : A- Salt of strong base and weak acid eg. borax, Na 2 CO 3 B- Salt of weak base and strong acid eg. Fe. Cl 3, Al 2(SO 4)3 N. B. Always titrate the strong part of the salt. eg. 1. KCN We titrate KOH by standard acid eg. HCl eg. 2. Borax Na 2 B 4 O 7 Borax hydrolyze in water to give: Na 2 B 4 O 7 + 7 H 2 O 4 H 3 BO 3 + 2 Na. OH v. weak acid tit. 2 HCl using M. O titrate HCl using M. O indicator. (let reading = x) eg. 3. Na 2 CO 3 sodium carbonate. Na 2 CO 3 p. H = HCl Na. HCO 3 8. 3 Ph. HCl Na. Cl + H 2 O +CO 2 3. 8 M. O.

Na 2 CO 3 can be determined by titration against HCl by 2 methods : a- Using M. O. as indicator it will give total CO 3 -2 b- Using Ph. as indicator it will give 1/2 CO 3 -2 and considered as half neutralization step. But care that the 1 st step to Na. HCO 3 takes place on 2 separate steps; Na 2 CO 3 + 2 HCl 2 Na. Cl + CO 2 Na 2 CO 3 +CO 2 + H 2 O 2 Na 2 CO 3 +2 HCl +H 2 O 2 Na. HCO 3 2 Na. HCO 3 + 2 Na. Cl i. e. For 1/2 neutralization, we must prevent the escape of CO 2 by : 1 - cooling 2 - dilution 3 -stirring 4 - dipping the nozzle of burrete under the surface of the soln.

3 - Indirect or back (residual titration) - In it we add Known excess of standard to the sample and titrate the excess unreacted standard. Conc. of sample A = Known excess standard - b When do we use back titration ? 1 - When sample is volatile. eg. NH 3, formic acid. 2 - When sample is insoluble eg. Zn. O, Ca. CO 3, Ba. CO 3 3 - When reaction require heat of standard solution. 4 - When reaction proceed only in presence of excess reagent eg. with lactic acid. Determination of inorganic ammonium salts : NH 4 Cl + Na. OH Na. Cl + NH 3 + H 2 O Add known excess Na. OH , then boil to remove NH 3 and titrate excess Na. OH using HCl and M. R. as indicator. Determination of nitrogen in organic compounds : (by Kjeldahl's method) : Organic cpd Conc. H 2 SO 4 Δ NH 3 (NH 4)2 SO 4 -Nitrogen of organic cpd is reduced to NH 3 by digestion with conc. H 2 SO 4 in the presence of K 2 SO 4 or Na 2 SO 4 (to raise the boiling point of acid) and Cu. SO 4 or Hg. O as a catalyst.

-The organic cpd. is oxidised to CO 2 and the acid is reduced to SO 2 and nitrogen to NH 3 which is fixed with excess acid as (NH 4)2 SO 4 then add known excess Na. OH and titrate excess unreacted alkali against HCl as before. 4 - Other indirect Titrations : Determination of esters : Esters are hydrolysed by reflux with known excess of Na. OH, cool and the excess unreacted Na. OH is titrated against standard HCl using Ph. indicator. Determination of ammonium salt and amino acids (Formol titration) Another method for determination of ammonium salts is formol titration. When formaldehyde is added to the sample , hexamethylene tetramine (hexamine) which is neutral is formed and equivalent amount of acid which can be titrated against Na. OH. 4 NH 4 Cl + 6 HCHO (CH 2)6 N 4 + 4 HCl + 6 H 2 O 4(NH 4)2 SO 4 + 6 HCHO (CH 2)6 N 4 + 2 H 2 SO 4 + 6 H 2 O N. B. HCHO must be neutralized from any formic acid due to aerial oxidation.
- Slides: 7