2 Solubility and Molecular Weights Polymer Solubility 1

2. Solubility and Molecular Weights Polymer Solubility 1

Titles Solubility ◦ Solubility parameters ◦ Experimental determination Thermodynamics of Mixing ◦ Types of Solutions ◦ Dilute solutions ◦ Flory-Huggins parameter Polymer Solubility 2

Titles (contd. ) Molecular Weights ◦ Average Molecular weights ◦ Number average molecular weights ◦ Determination of number average MW ◦ Weight average MW ◦ Light scattering Intrinsic viscosity ◦ Mark-Houwink relationship Polymer Solubility 3

Title (contd. ) Gel permeation chromatography Solution thermodynamics and molecular weights Polymer Solubility 4

How Does a Polymer Dissolve? There are two distinguishable modes of solvent diffusion into a polymer. 1. Fickian diffusion, (T>Tg) 2. non-Fickian phenomenon known as case II swelling, (T<Tg) T is important. Why? What does swelling mean? Polymer Solubility 5
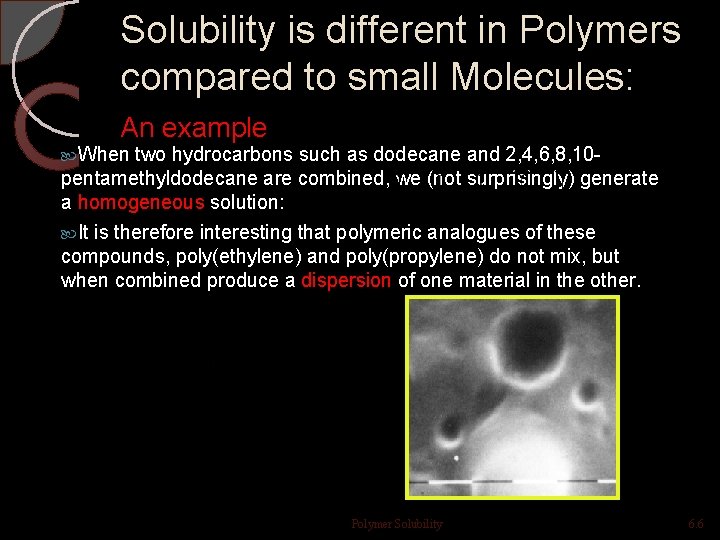
Solubility is different in Polymers compared to small Molecules: An example When two hydrocarbons such as dodecane and 2, 4, 6, 8, 10 pentamethyldodecane are combined, we (not surprisingly) generate a homogeneous solution: It is therefore interesting that polymeric analogues of these compounds, poly(ethylene) and poly(propylene) do not mix, but when combined produce a dispersion of one material in the other. Polymer Solubility 6. 6

Mixing Or Not? Whether the mixing of two compounds generates a homogeneous solution or a blend depends on the Gibbs energy change of mixing. A-B solution m. A grams polymer A m. B grams material B Gmix < 0 + Gmix > 0 Gmix (Joules/gram) is defined by: Gmix = Hmix -T Smix where immiscible blend Hmix = HAB - (w. AHA + w. BHB) Smix = SAB - (w. ASA + w. BSB) and w. A, w. B are the weight fractions of each material. Polymer Solubility 6. 8

Entropy of Mixing Consider the two-dimensional lattice representation of a solvent (open circles) and its solute (solid circles): small molecule solute polymeric solute Mixing of small molecules results in a greater number of possible molecular arrangements than the mixing of a polymeric solute with a solvent. Ø While Smix is always positive (promoting solubility), its magnitude is less for polymeric systems than for solutions of small molecules Ø When dealing with polymer solubility, the enthalpic contribution Hmix to the Gibbs energy of mixing is critical. Polymer Solubility 6. 9

Enthalpy of Mixing Hmix can be a positive or negative quantity Ø If A-A and B-B interactions are stronger than A-B interactions, then Hmix > 0 (unmixed state is lower in energy) Ø If A-B interactions are stronger than pure component interactions, then Hmix < 0 (solution state is lower in energy) An ideal solution is defined as one in which the interactions between all components are equivalent. As a result, Hmix = HAB - (w. AHA + w. BHB) = 0 for an ideal mixture In general, most polymer-solvent interactions produce Hmix > 0, the exceptional cases being those in which significant hydrogen bonding between components is possible. Ø Predicting solubility in polymer systems often amounts to considering the magnitude of Hmix > 0. Ø If the enthalpy of mixing is greater than T Smix, then we know that the lower Gibbs energy condition is the unmixed state. Polymer Solubility 6. 10

The solubility parameters Parameters Affecting the Solubility: GM = HM - T SM VM represents the total volume of the mixture, E represents the energy of vaporization to a gas at zero pressure (i. e. , at infinite separation of the molecules), and V is the molar volume of the components, for both species 1 and 2. The quantity v represents the volume fraction of component 1 or 2 in the mixture. Polymer Solubility 11

HM Based on Solubility Parameters Thus the heat of mixing of two substances dependens on 2 ( 1 - 2) Polymer Solubility 12
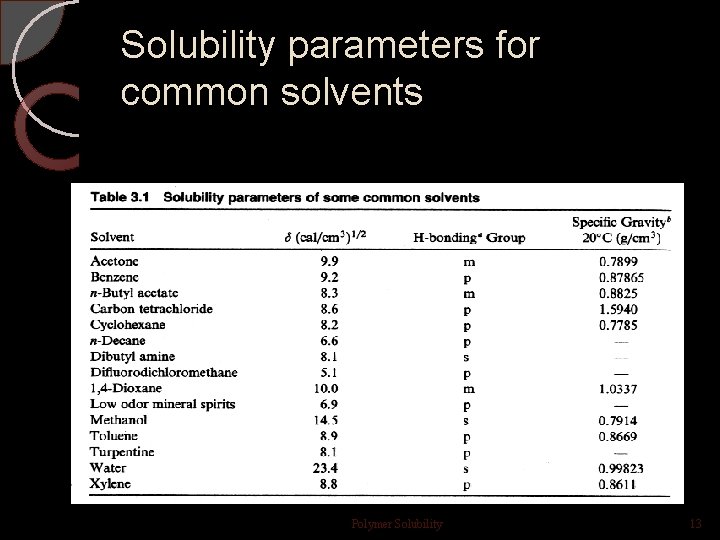
Solubility parameters for common solvents Polymer Solubility 13

Solubility parameters for common polymers Polymer Solubility 14

Determining The Solubility Parameter δ Polymer Solubility 15

Theoretical Calculations G = group molar attraction constant Polymer Solubility 16
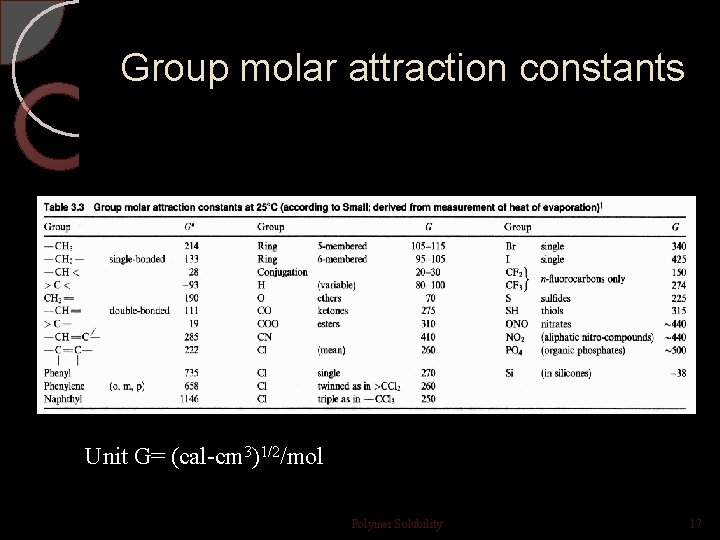
Group molar attraction constants Unit G= (cal-cm 3)1/2/mol Polymer Solubility 17

—CH 2— , G = 133, -CH- , G=28, phenyl group, G = 735. The density of polystyrene is 1. 05 g/cm 3, and the mer molecular weight is 104 g/mol. Then: Polymer Solubility 18
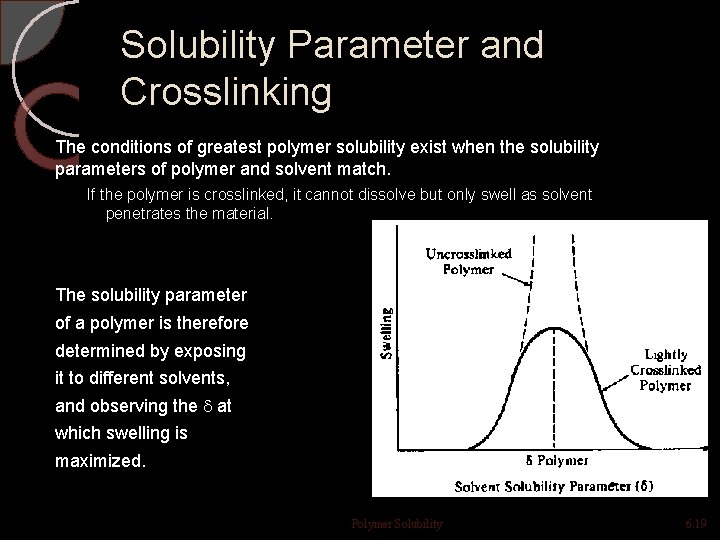
Solubility Parameter and Crosslinking The conditions of greatest polymer solubility exist when the solubility parameters of polymer and solvent match. If the polymer is crosslinked, it cannot dissolve but only swell as solvent penetrates the material. The solubility parameter of a polymer is therefore determined by exposing it to different solvents, and observing the at which swelling is maximized. Polymer Solubility 6. 19

The swelling coefficient, Q, is defined by, where m is the weight of the swollen sample, m 0 is the dry weight, and s is the density of the swelling agent. Polymer Solubility 20
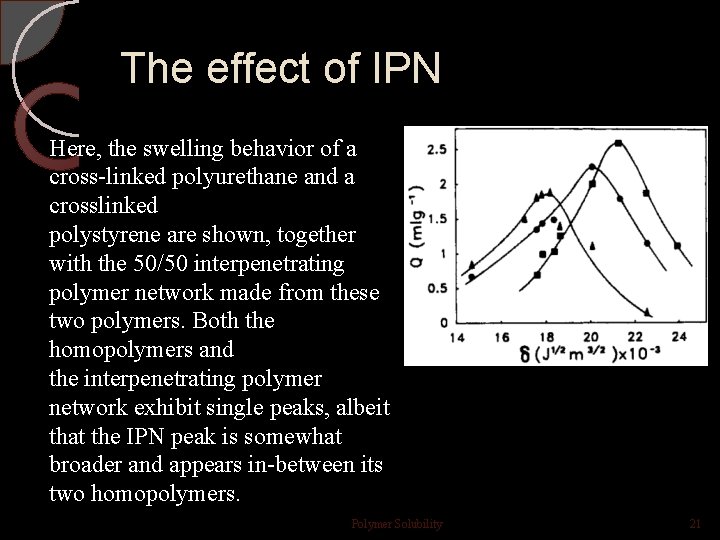
The effect of IPN Here, the swelling behavior of a cross-linked polyurethane and a crosslinked polystyrene are shown, together with the 50/50 interpenetrating polymer network made from these two polymers. Both the homopolymers and the interpenetrating polymer network exhibit single peaks, albeit that the IPN peak is somewhat broader and appears in-between its two homopolymers. Polymer Solubility 21
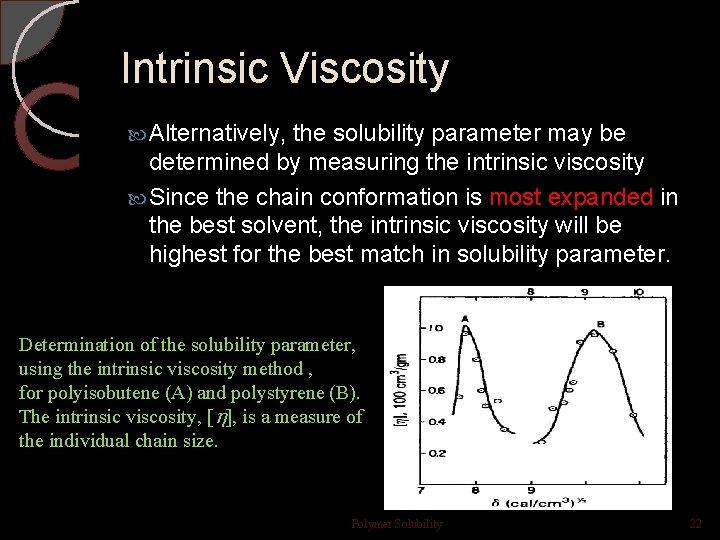
Intrinsic Viscosity Alternatively, the solubility parameter may be determined by measuring the intrinsic viscosity Since the chain conformation is most expanded in the best solvent, the intrinsic viscosity will be highest for the best match in solubility parameter. Determination of the solubility parameter, using the intrinsic viscosity method , for polyisobutene (A) and polystyrene (B). The intrinsic viscosity, [ ], is a measure of the individual chain size. Polymer Solubility 22

Thermodynamics of mixing Polymer Solubility 23

Entropy Of Mixing ΔS: Statistical thermodynamics Boltzman Equation: Polymer Solubility 24

= number of possible arrangements that the molecule may assume Polymer Solubility 25

Sterling Approx. Volume fraction of solvent and polymer Polymer Solubility 26

Mixing Enthalpy ΔH Polymer Solubility 27

1 Polymer Solubility 28

Polymer Solubility 29

Polymer Solubility 30

Chemical Potential and Energy of Mixing Polymer Solubility 32
- Slides: 30