2 Mass and Energy Balance Objective To introduce


����� 2 �������� (Mass and Energy Balance) • Objective Ø To introduce the principle of mass and energy conservation Ø To present the concept of process flow diagrams Ø To present the concept of system boundaries Ø To present the concept of total mass balance Ø To present the concept of component mass balance Ø To present the concept of total energy balance





The system-Flow Diagram and Boundaries • System: ������������ • Surrounding: ������������ • Boundary: ��������� system ��� surrounding • Closed system: ������������
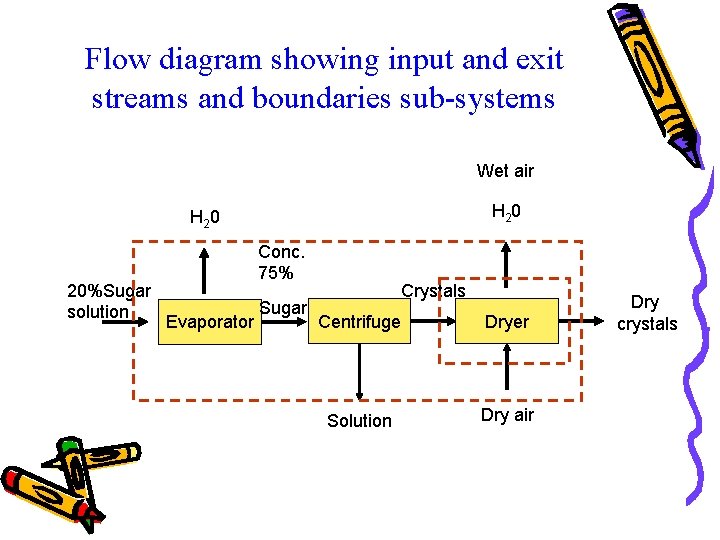
Flow diagram showing input and exit streams and boundaries sub-systems Wet air H 2 0 20%Sugar solution Conc. 75% Evaporator Sugar Crystals Centrifuge Dryer Solution Dry air Dry crystals
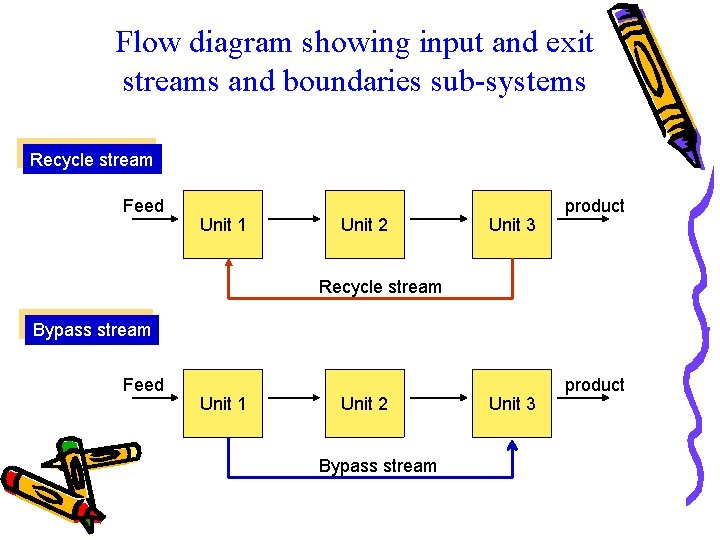
Flow diagram showing input and exit streams and boundaries sub-systems Recycle stream Feed Unit 1 Unit 2 Unit 3 product Recycle stream Bypass stream Feed Unit 1 Unit 2 Bypass stream Unit 3 product

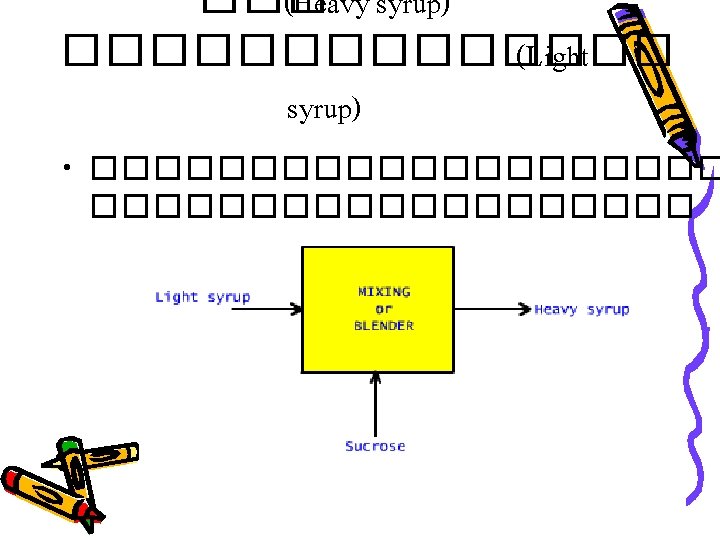



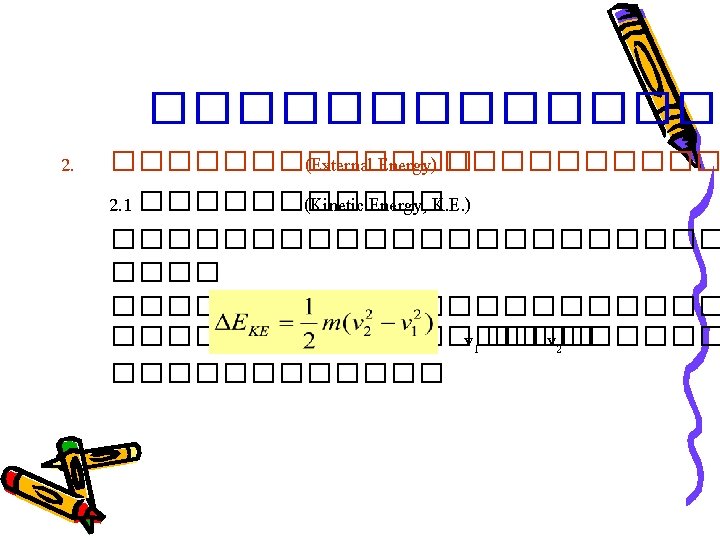
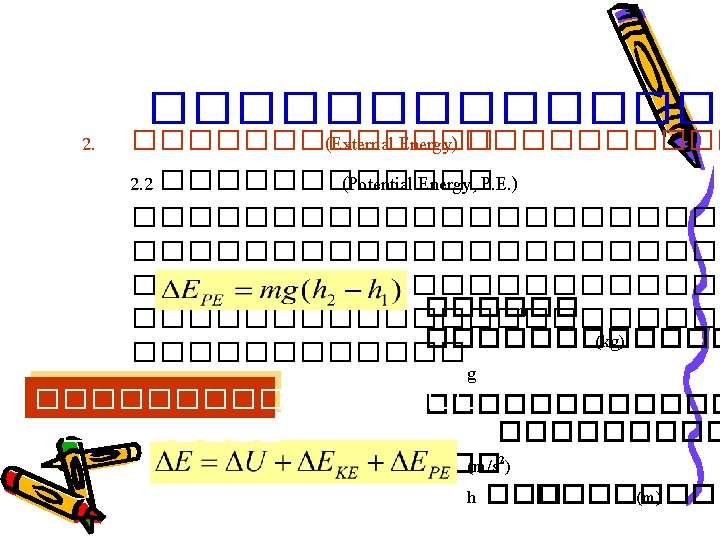
Basic Principles • Law of Conservation of Mass input – output + generation – consumption = accumulation 0 0 ������� – ����� + ���������� – ������� = • ����������������� � generation ��� consumption = 0 input – output = accumulation
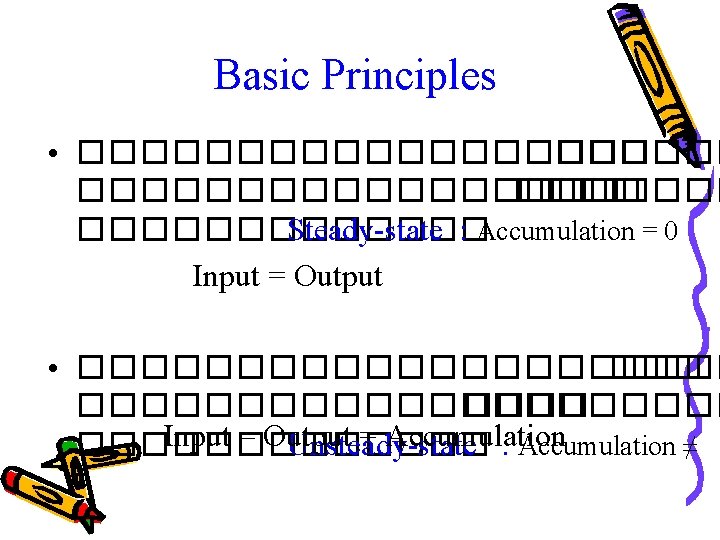
Basic Principles • ��������������������� ������� Steady-state : Accumulation = 0 Input = Output • ��������������������� Input – Output = Accumulation ������� Unsteady-state : Accumulation ≠ 0


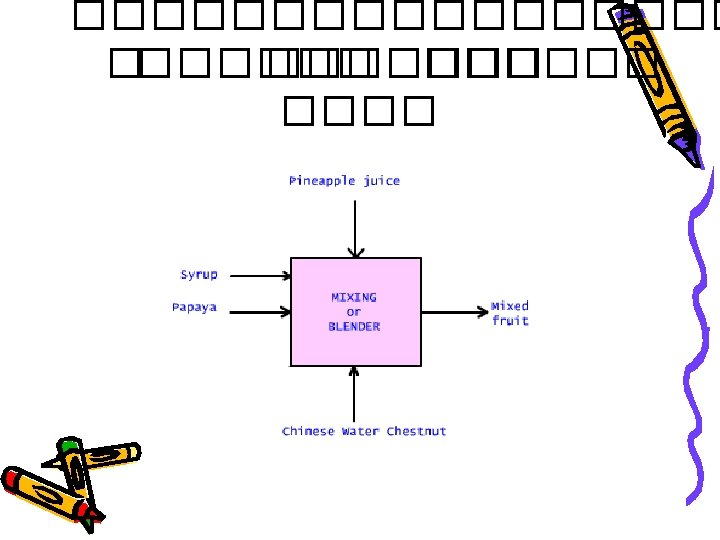
Component Mass balance • ������� total mass balance �������� (component) ���������� • ���������� component MB ������ n ��������� total MB ������ n-1 component balance equation • ������ balance ��������� mass fraction/mass percentage

Mass fraction A = Mass of component A Total mass of mixture containing A Mass of component A = Mass fraction of A x Total mass of mixture containing A
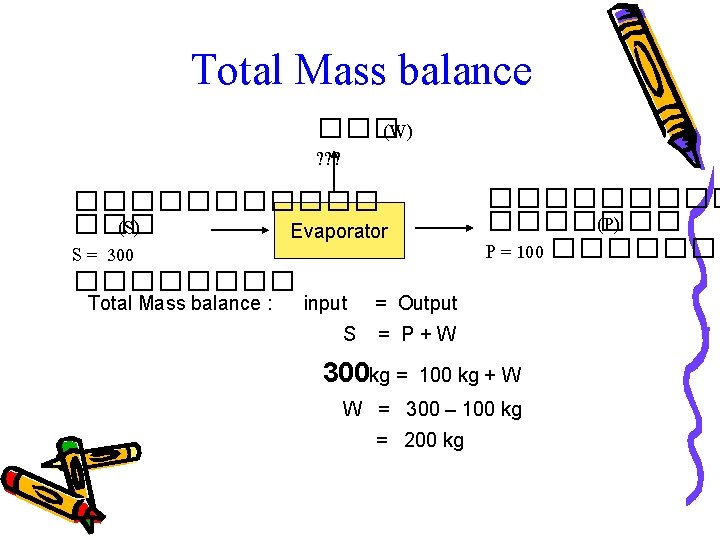
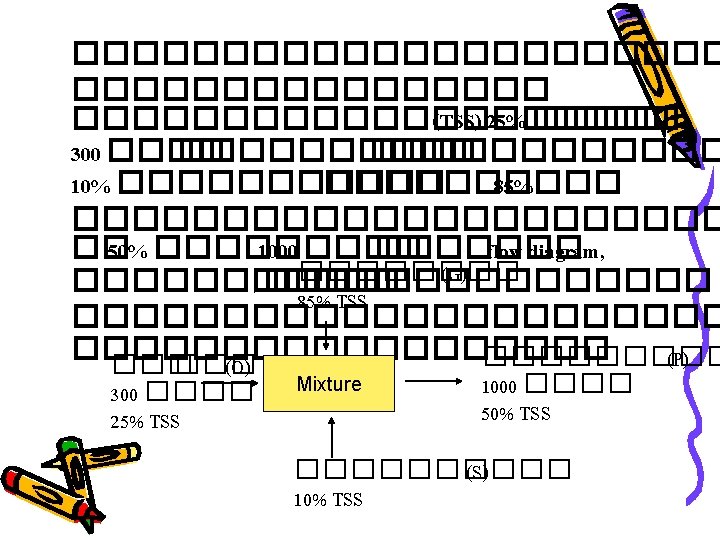
Total Mass balance ��� (W) ? ? ? ������� (P) P = 100 ����������� (S) Evaporator S = 300 ���� Total Mass balance : input S = Output = P+W 300 kg = 100 kg + W W = 300 – 100 kg = 200 kg
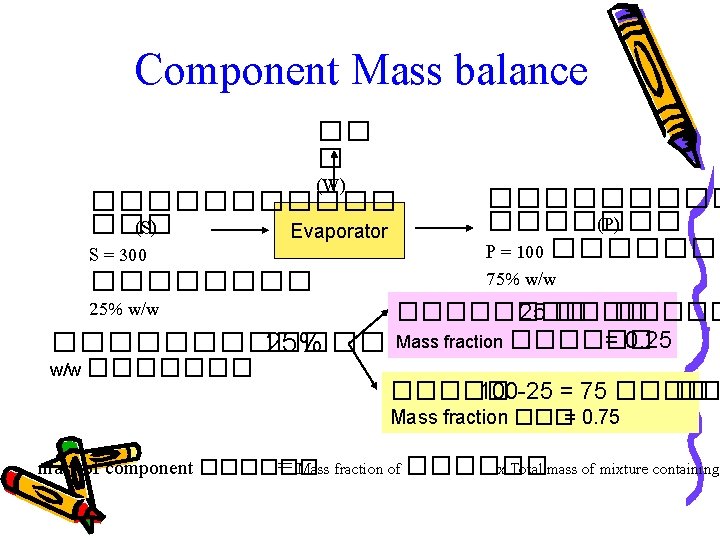
Component Mass balance �� � (W) ����������� (P) ��� (S) Evaporator P = 100 ������� S = 300 75% w/w �������� 25 ����� Mass fraction ������ = 0. 25 ������� 25% w/w ������� 100 -25 = 75 ���� �� Mass fraction ���= 0. 75 mass of component ������ = Mass fraction of ������ x Total mass of mixture containing









���������� Where m is the mass fraction ; The subscript : c = carbohydrate p = protein f = fat a = ash m = moisture



������ 3 5 kg of ice at -10 o. C is heated to melt it into water at 0 o. C; then additional heat is added to vaporize the water into stream. The saturated vapors exit at the 100 o. C. Calculate different enthalpy values involved in the process. Specific heat of ice and water are 2. 05 and 4. 182 k. J/kg K respectively, latent heat of fusion is 333. 2 k. J/kg, and latent heat of vaporization at 100 o. C is 2257. 06 k. J/Kg.


������ 4 A tubular water blancher is being used to process lima beans. The product mass flow rate is 860 kg/hr. It is found that theoretical energy consumed for the blanching process amounts to 1. 19 GJ/hr. The energy lost due to lack of insulation around the blancher is estimate to be 0. 24 GJ/hr. If the total energy input to the blancher is 2. 71 GJ/hr. – Caculate the energy required to reheat water – Determine the percent energy associated with each stream.

������ 5 Steam is used for peeling of potatoes in a semi-continuous operation. Steam is supplied at the rate of 4 kg/100 kg of unpeeled potatoes. The unpeeled potatoes enter the system with a temperature of 17 o. C, and the peeled potatoes leave at 35 o. C. A waste stream from the system leave at 60 o. C. The Cp of unpeeled potatoes, waste, peel potatoes are 3. 7, 4. 2 and 3. 5 k. J/kg o. C respectively. If the heat content (assuming 0 o. C ref. temp) of the steam is 2750 k. J/kg, determine the quantities of the waste stream and the peeled potatoes from the process.
- Slides: 41