2 6 Redox Part 1 a Balancing Redox

2. 6 Redox Part 1 a. Balancing Redox Reactions (Half-equation method) b. write ionic half-equations and use them to construct full ionic equations. Connector: Updated_Crowe_2012
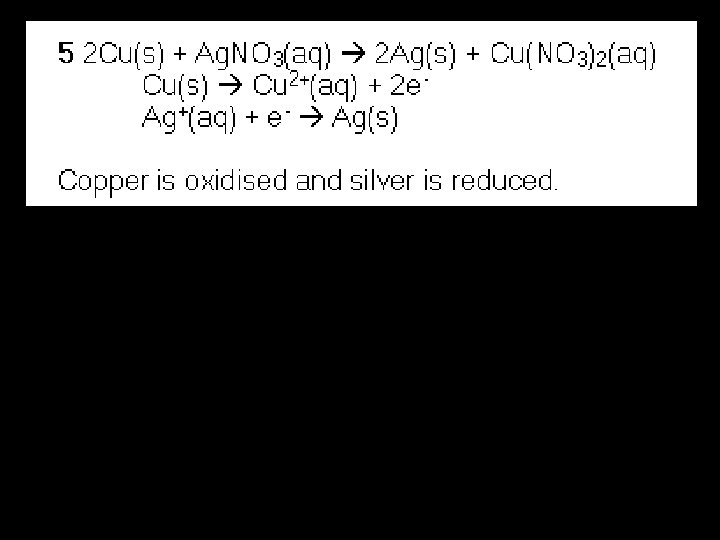

Balancing Redox Reactions (Half-equation method) The half-equation method separates a redox reaction into its oxidation and reduction half reactions. Overall scheme for the half reaction method: Step 1: Step 2: Step 3: Step 4: Step 5: Split reaction into half-reactions (reduction and oxidation) Balance the charge or oxidation number with electrons Balance O by adding H 2 O Balance H by adding H+ Multiply by some integer to make electrons (lost) = electrons (gained) Step 6: Add half equations and cancel substances on both sides

Example 1. Reaction between manganese (II) ion and the bismuthate ion in acid solution 2+ 5+ 7+ What are the oxidation numbers for Mn and Bi ? Write the half equation for Mn 2+ Balance O by adding H 2 O, then H by adding H+ Repeat the process for the bismuthate ion 3+

Balance O by adding H 2 O, then H by adding H+ We now have the two half equations: Step 5: Step 6: Multiply by some integer to make electrons (lost) = electrons (gained) Add half equations and cancel substances on both sides

8 10 Step 6: 2 30 2 5 16 5 10 15 Add half equations and cancel substances on both sides

Overview

Solve these acid solution reactions: : Mn. O 4 - Mn 2+ 2 I- I 2 H 2 O 2 H 2 O + O 2 Cr 2 O 72 - Cr 3+
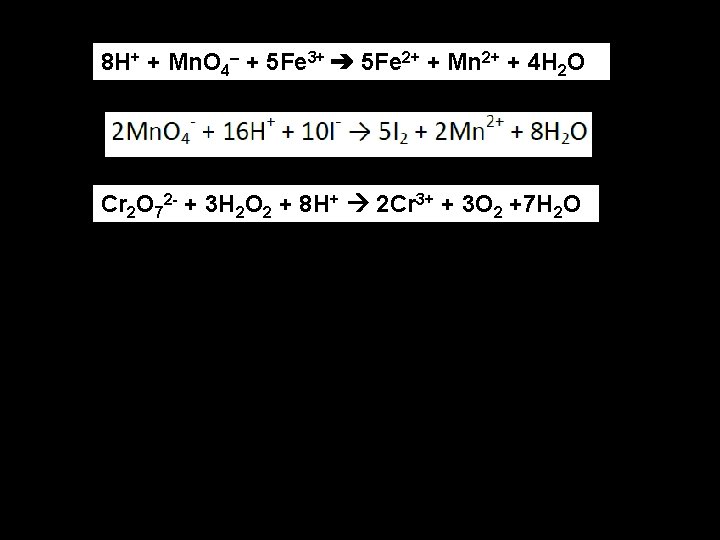
8 H+ + Mn. O 4– + 5 Fe 3+ ➔ 5 Fe 2+ + Mn 2+ + 4 H 2 O Cr 2 O 72 - + 3 H 2 O 2 + 8 H+ 2 Cr 3+ + 3 O 2 +7 H 2 O
- Slides: 9