2 4 Properties of Solids Chemistry 30 Mr



















- Slides: 27

2. 4 Properties of Solids Chemistry 30 Mr. Goller

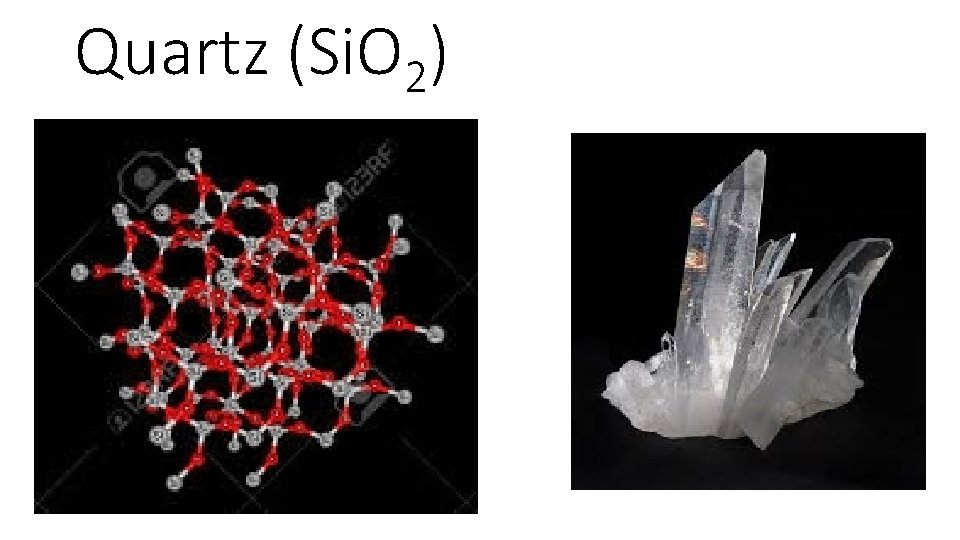
Covalent Network Solids • composed only of atoms interconnected by covalent bonds. • brittle non conductors (heat or electricity) • extremely hard • Ex Quartz and Diamond • Diamond is so hard because each carbon atom is bonded to four other carbon atoms. This tetrahedral arrangement forms a strongly bonded crystal system that is extremely hard and has a very high melting point.

The only network covalent solid that actually does conduct!

Quartz (Si. O 2)

Solubility and Polarity (Like Dissolves Like) • Solubility – The ability of a substance to dissolve in another • Polar molecules and ionic compounds are generally soluble in polar substances. • Non polar molecules dissolve only in nonpolar substances. This is why water and oil do not mix.


Ionic Compounds • Structure 3 D Crystal Lattice • Large numbers of positive and negative ions exist together in a ratio determined by the number of electrons transferred. • The ions are packed into a regular repeating pattern that balances forces of attraction and repulsion between the ions.

Ionic Compounds • Aragonite (Ca. CO 3) Barite (Ba. SO 4) Beryl )Be 3 Al 2 Si 6 O 18(

Physical Properties of Ionic Compounds • In the solid state, ions in an ionic compound are locked into place. • As a result, ionic solids do not have freely moving charged particles and therefore cannot conduct electricity. • However, when an ionic solid melts or is dissolved, the ions are free to move and conduct an electric current very well. • Electrolyte Ionic compound whose aqueous solution can conduct electricity

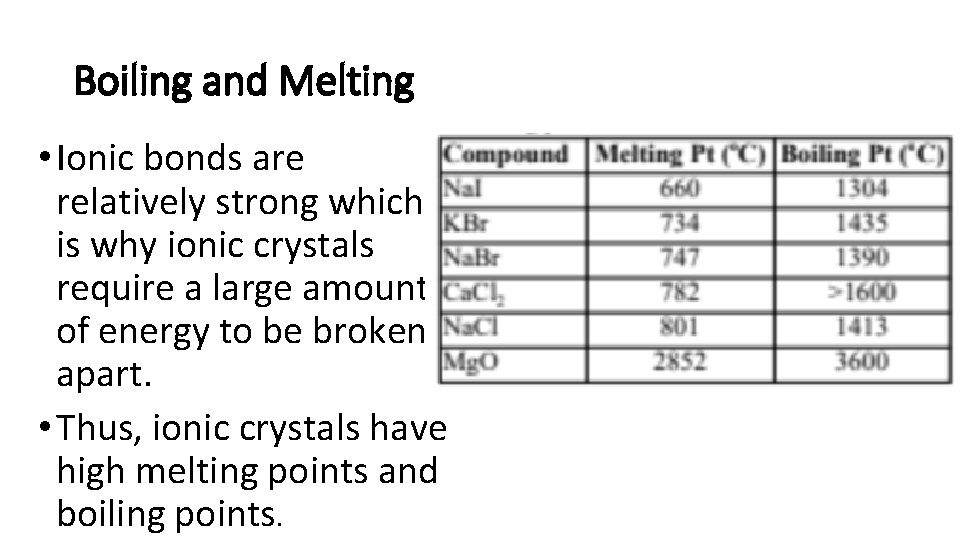
Boiling and Melting • Ionic bonds are relatively strong which is why ionic crystals require a large amount of energy to be broken apart. • Thus, ionic crystals have high melting points and boiling points.

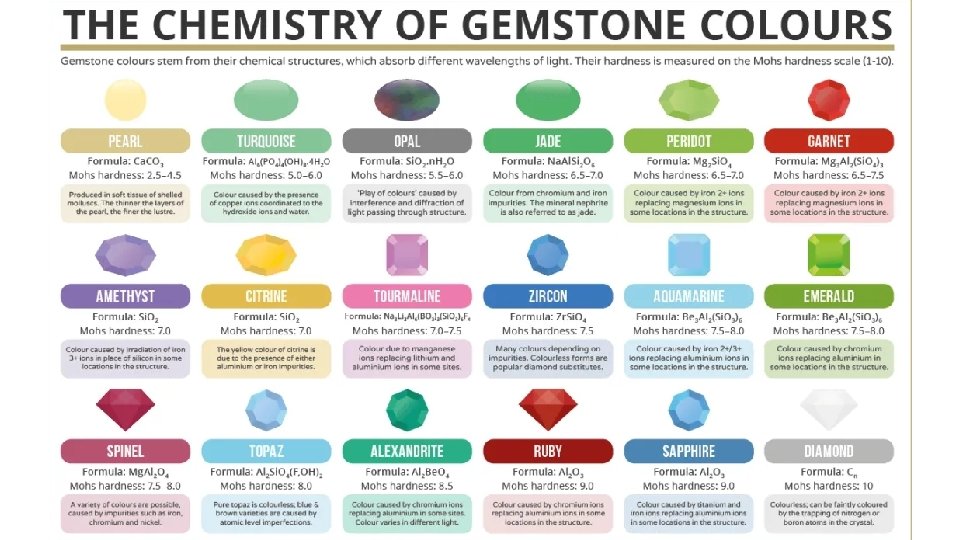
Luster • Many crystals, including gemstones, have brilliant colours due to transition metals in the crystal lattice.


Hard and Brittle • Ionic crystals are hard, rigid, brittle, and require a large amount of energy to break. When a force is strong enough to break an ionic crystal, they will generally cleave along a straight line.

Energy and the Ionic Bond • Chemical reactions can be classified into two categories, endothermic and exothermic • Endothermic Net input of energy (energy is absorbed) R + E >P • Exothermic Net output of energy (energy is give off) R > P +E • The formation of ionic compounds from their ions is always exothermic

Metallic Bonds and the Properties of Metals • Although not ionic, metals share many properties with ionic compounds. • The bonding in both metals and ionic compounds is based on the attraction of particles with unlike charges. Metals often form lattices in the solid state. • In such a lattice, 8 to 12 other metal atoms closely surround each metal atom.

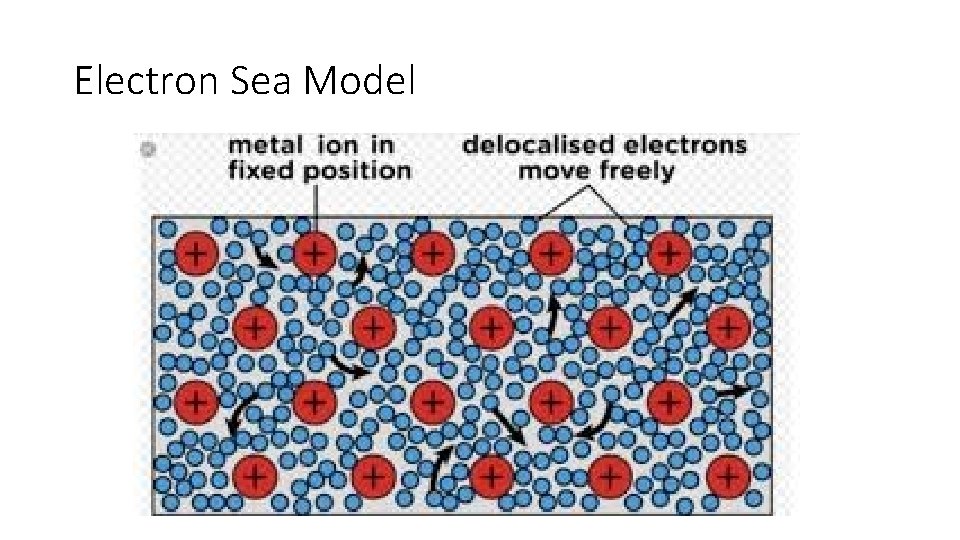
Electron Sea Model • All metal atoms in a metallic solid contribute their valence electrons to form a "sea" of electrons. • This sea of electrons surrounds the metal cations in the lattice. The outer electrons are not held by any one specific atom and can move freely from one atom to the next. • Because they are free, they are referred to as delocalized electrons.

Electron Sea Model

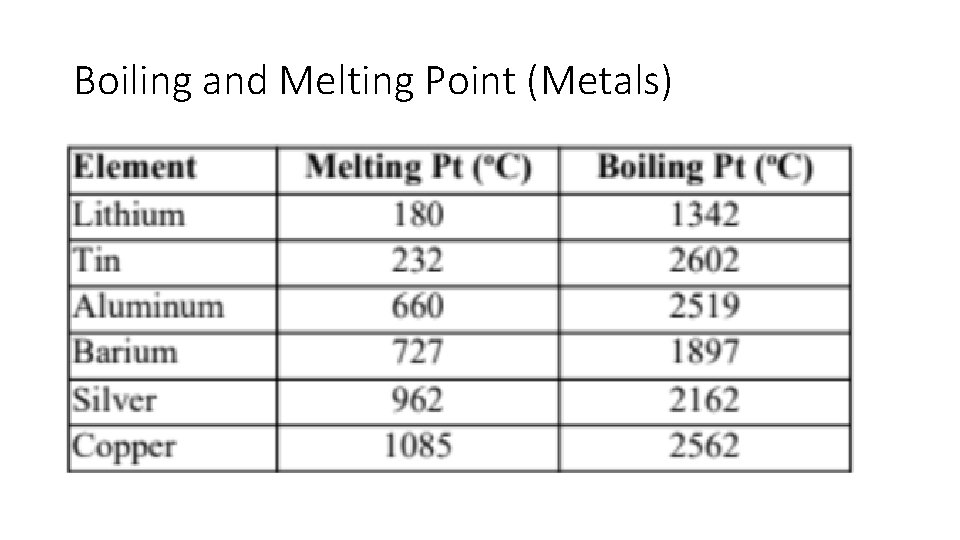
Melting and Boiling Points - Metals • Melting points vary greatly. . . Mercury is a liquid at room temperature. Tungsten (lightbub filament) has a melting point of 3422 C. • Due to the mobility of electrons and cations in a metal, it does not take a great deal of energy to move them past one another. • Boiling requires separation of cations and electrons which requires a great amount of energy.

Boiling and Melting Point (Metals)

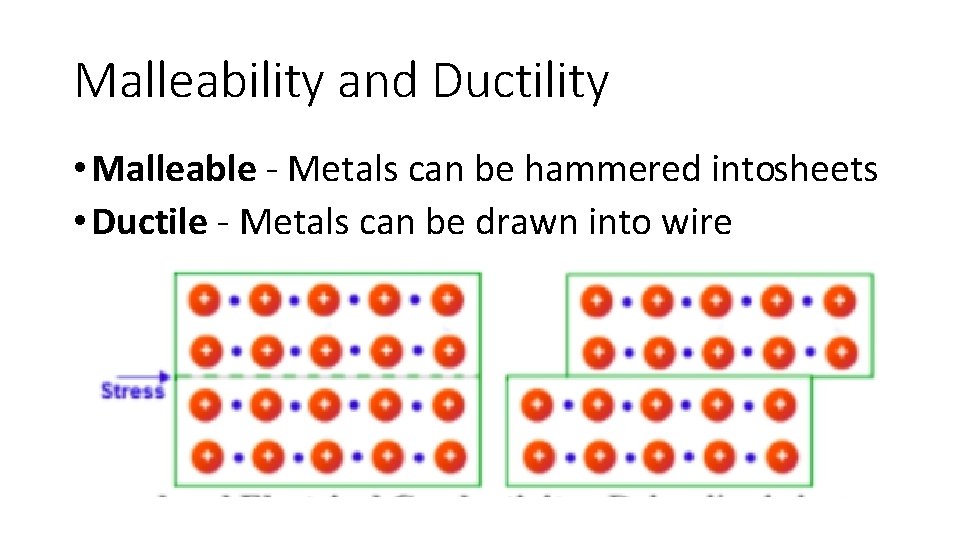
Malleability and Ductility • Malleable Metals can be hammered intosheets • Ductile Metals can be drawn into wire

Conductivity and Luster • Thermal (heat) and Electrical Conductivity Delocalized electrons are mobile and can transfer both heat and electricity. • The electrons can also absorb and release photons of light, making metals lustrous.

Hardness • Metals with many delocalized electrons form strong metallic bonds like chromium, iron, and nickel, while metals such as alkali metals (who only have one) are very soft and cut with ease.

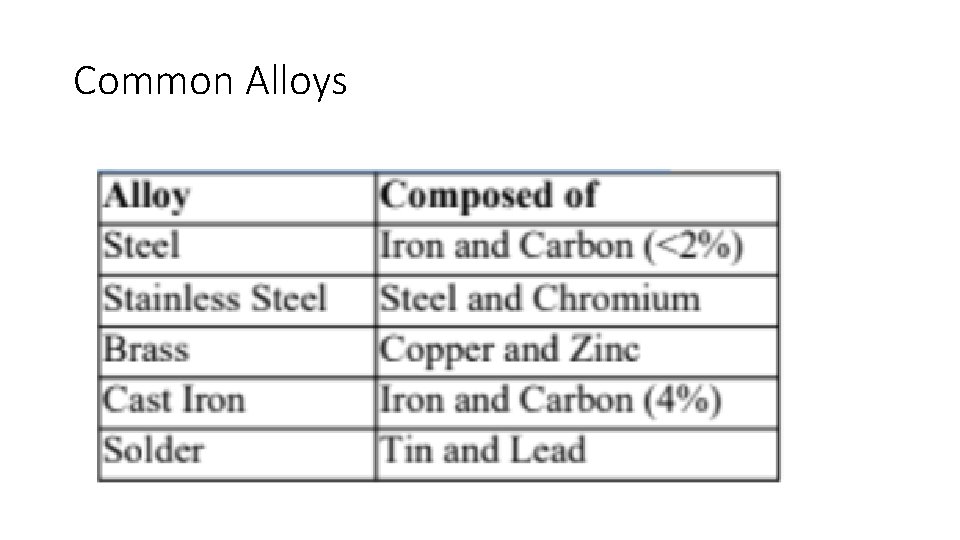
Metal Alloys • Due to the nature of metallic bonds, it is relatively easy to introduce other elements into the metallic crystal, forming an alloy, a mixture of elements that has metallic properties. • The properties of alloys differ somewhat from the properties of the elements they contain.

Common Alloys

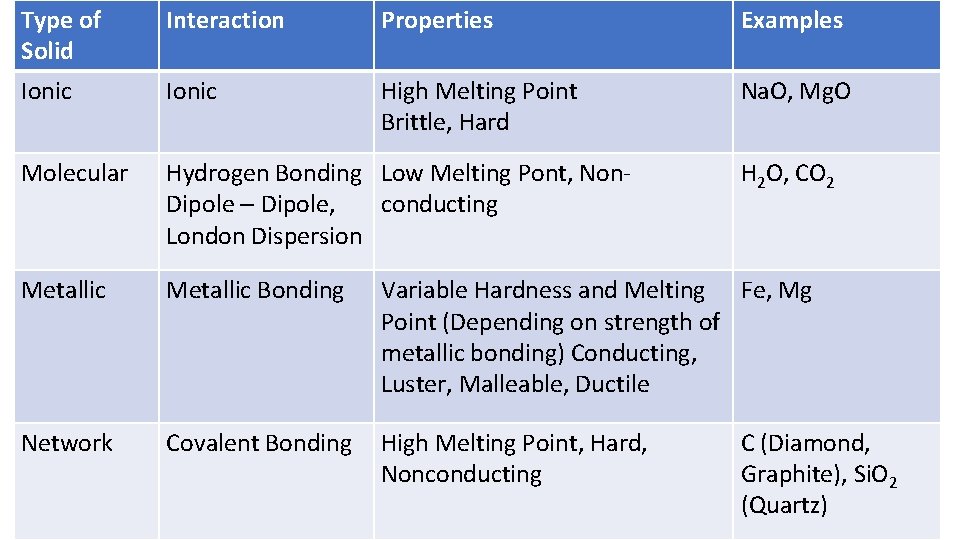
Type of Solid Interaction Properties Examples Ionic High Melting Point Brittle, Hard Na. O, Mg. O Molecular Hydrogen Bonding Low Melting Pont, Non Dipole – Dipole, conducting London Dispersion Metallic Bonding Variable Hardness and Melting Fe, Mg Point (Depending on strength of metallic bonding) Conducting, Luster, Malleable, Ductile Network Covalent Bonding High Melting Point, Hard, Nonconducting H 2 O, CO 2 C (Diamond, Graphite), Si. O 2 (Quartz)

Example 3 • Which would you expect to have a higher boiling point? Why? • Mg. O • CO 2

Example 4 • Which material would you choose if you needed something that could be stretched into wires to conduct electricity? Why? • Mg. O • Cu