2 4 Chemical Reactions Key Concept Life depends











- Slides: 18

2. 4 Chemical Reactions Key Concept: Life depends on chemical reactions

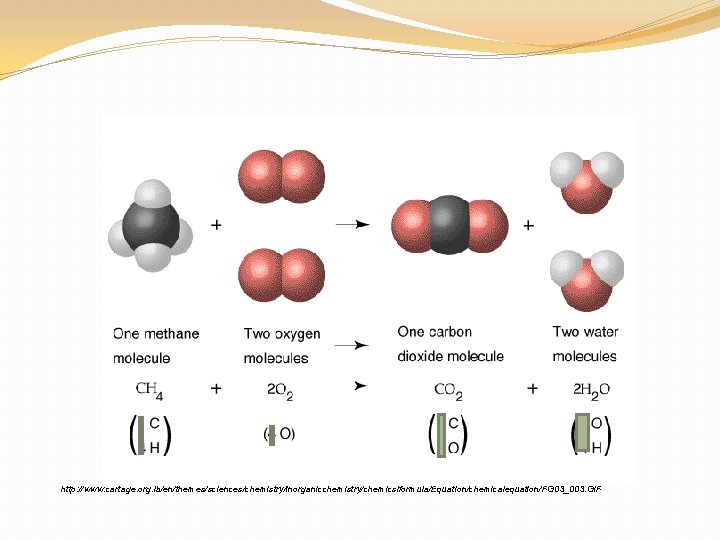
Chemical Reaction – the process of breaking the intramolecular bonds between atoms in a molecule and then reforming them so the atoms are arranged into new types of molecules.

http: //www. cartage. org. lb/en/themes/sciences/chemistry/inorganicchemistry/chemicslformula/Equation/chemicalequation/FG 03_003. GIF

Parts of a Chemical Reaction Chemical Equation – Shorthand way to describe what happens in the overall chemical reaction. Reactants – Elements or compounds that are the “starter materials” for a reaction. Products – Elements or compounds produced by a chemical reaction

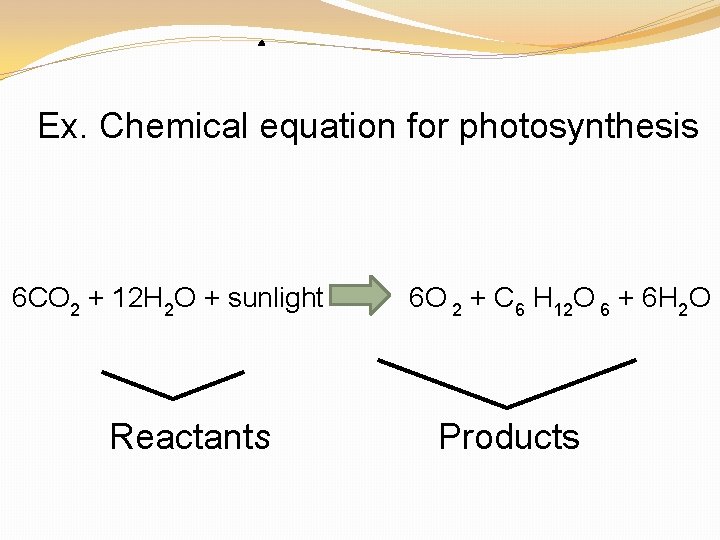
Ex. Chemical equation for photosynthesis 6 CO 2 + 12 H 2 O + sunlight 6 O 2 + C 6 H 12 O 6 + 6 H 2 O Reactants Products

Types of Chemical Reactions Synthesis Decomposition Combustion Single Displacement Double Displacement

Indicators a Reaction has Occurred

Indicators that a Rxn. occurred � 1. Gives off heat , light or sound (all forms of Energy) � 2. Gives off a gas / bubbles � 3. Forms a solid precipitate � 4. Color change is observed

Chemical reactions always involve the breaking of bonds in reactant molecules and the formation of new bonds in the product molecules.

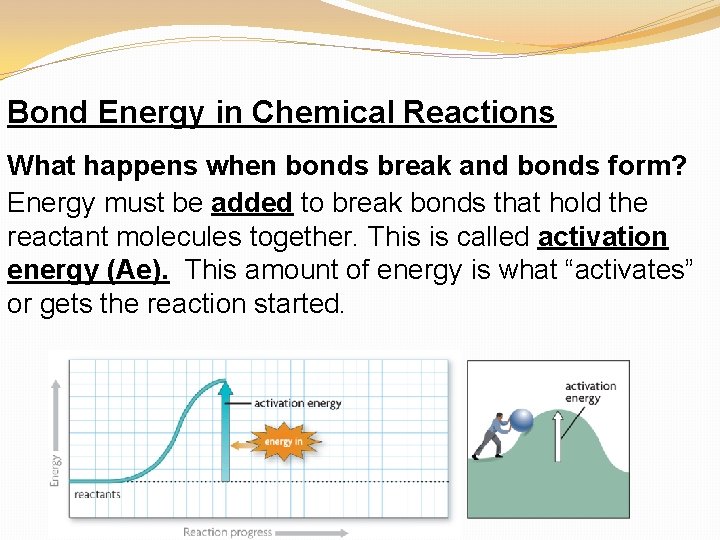
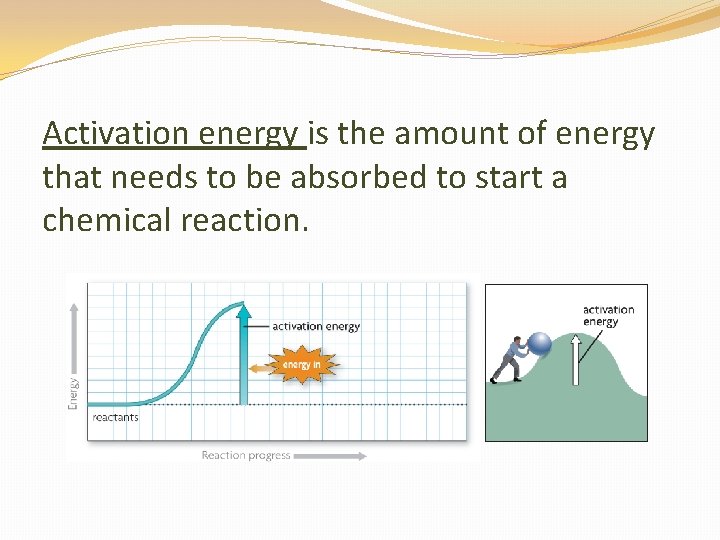
Bond Energy in Chemical Reactions What happens when bonds break and bonds form? Energy must be added to break bonds that hold the reactant molecules together. This is called activation energy (Ae). This amount of energy is what “activates” or gets the reaction started.

Once the bonds are broken, the atoms are freed up and can make new molecules. When bonds form between the atoms to make new molecules, energy is released.

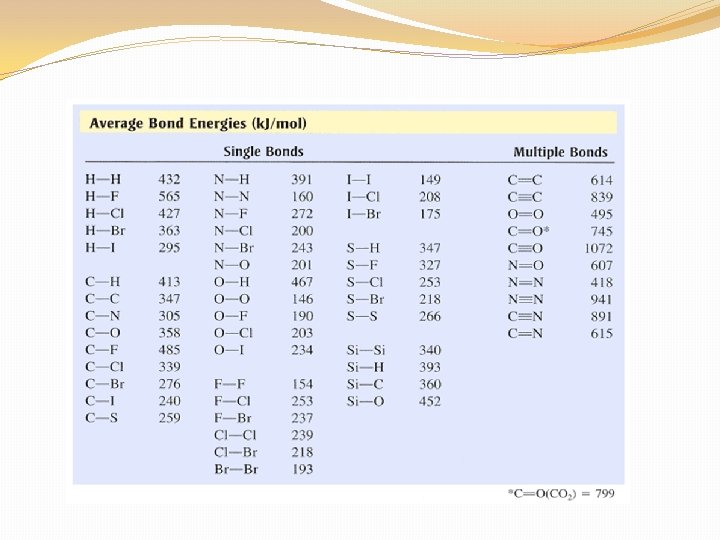
The specific amount of energy that is needed to break a bond, or is releases when that same bond forms, is called bond energy.

Two Categories of Chemical Reactions based on Energy Changes �Exothermic Reactions �Endothermic Reactions

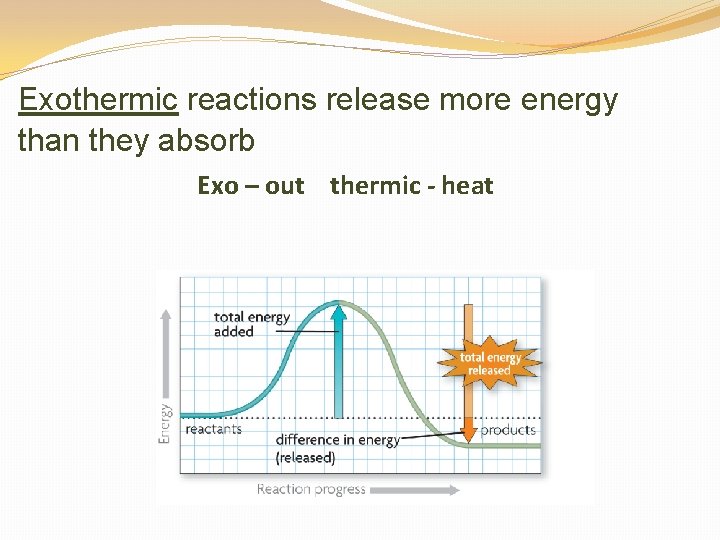
Exothermic reactions release more energy than they absorb Exo – out thermic - heat

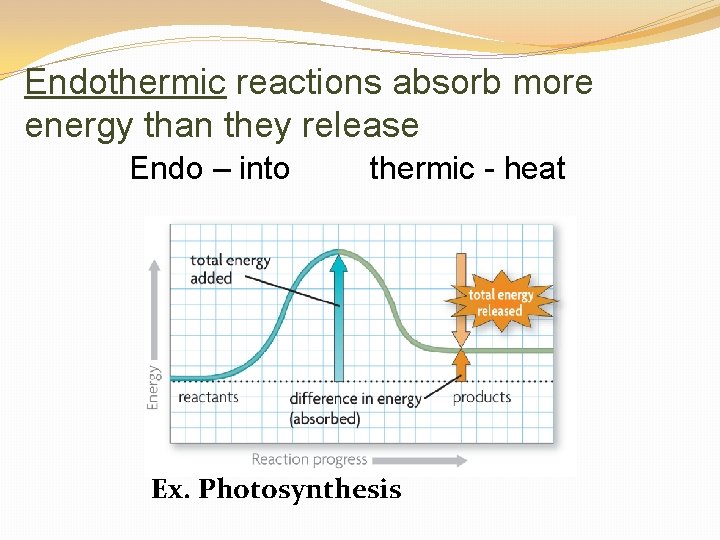
Endothermic reactions absorb more energy than they release Endo – into thermic - heat Ex. Photosynthesis


Activation energy is the amount of energy that needs to be absorbed to start a chemical reaction.

Connection to life: �In order to stay alive, organisms need to carry out chemical reactions that require energy; therefore every organism must have a source of energy to carry out reactions. �Plants trap and store energy from sunlight �Animals consume plants and other animals to produce energy