2 3 CarbonBased Molecules KEY CONCEPT Carbonbased molecules

2. 3 Carbon-Based Molecules KEY CONCEPT Carbon-based molecules are the foundation of life.
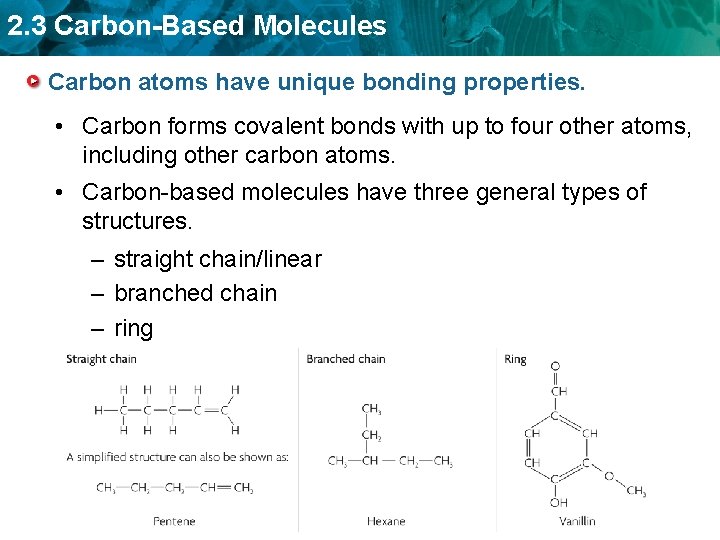
2. 3 Carbon-Based Molecules Carbon atoms have unique bonding properties. • Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. • Carbon-based molecules have three general types of structures. – straight chain/linear – branched chain – ring
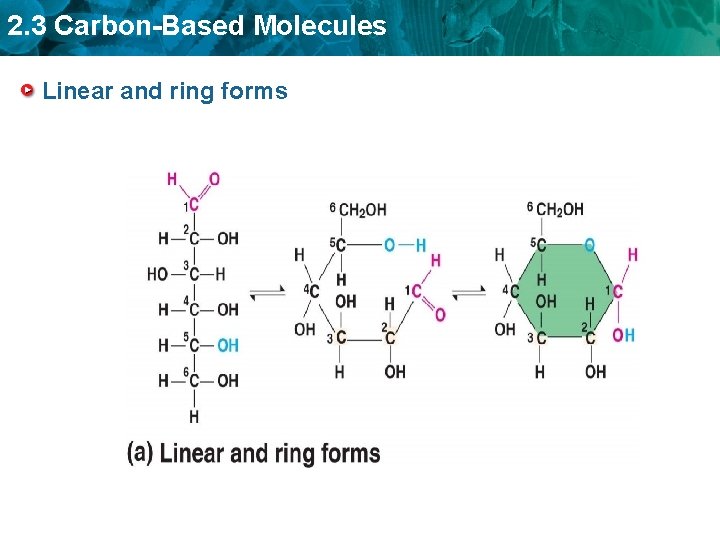
2. 3 Carbon-Based Molecules Linear and ring forms

2. 3 Carbon-Based Molecules Functional groups
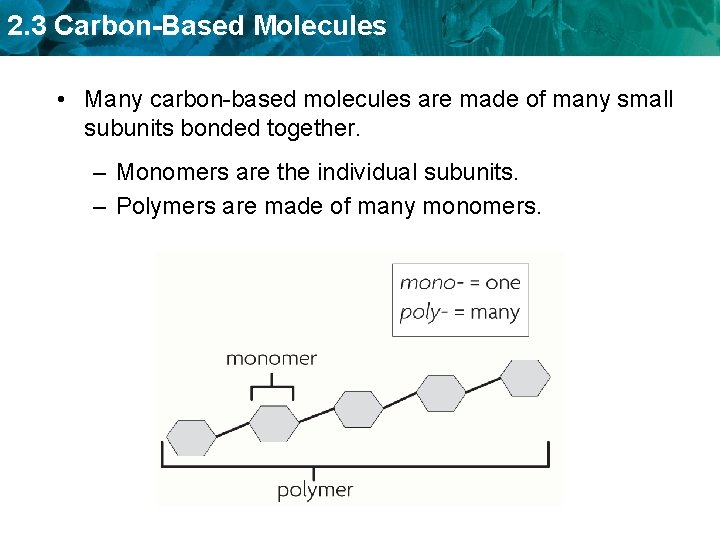
2. 3 Carbon-Based Molecules • Many carbon-based molecules are made of many small subunits bonded together. – Monomers are the individual subunits. – Polymers are made of many monomers.
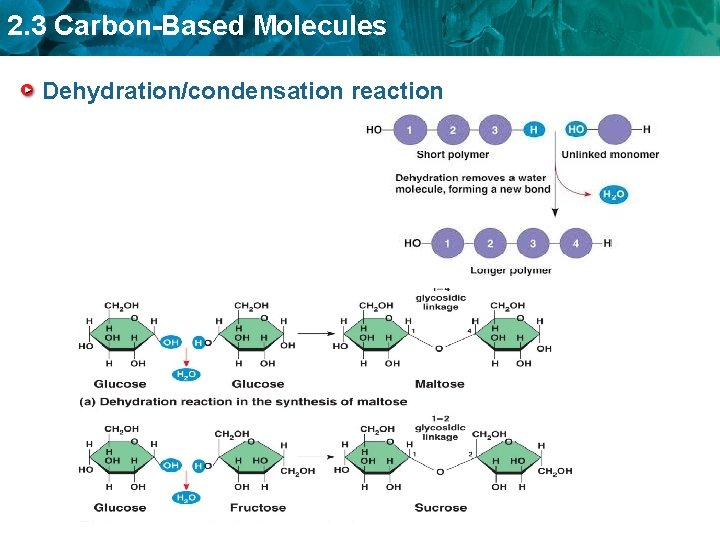
2. 3 Carbon-Based Molecules Dehydration/condensation reaction
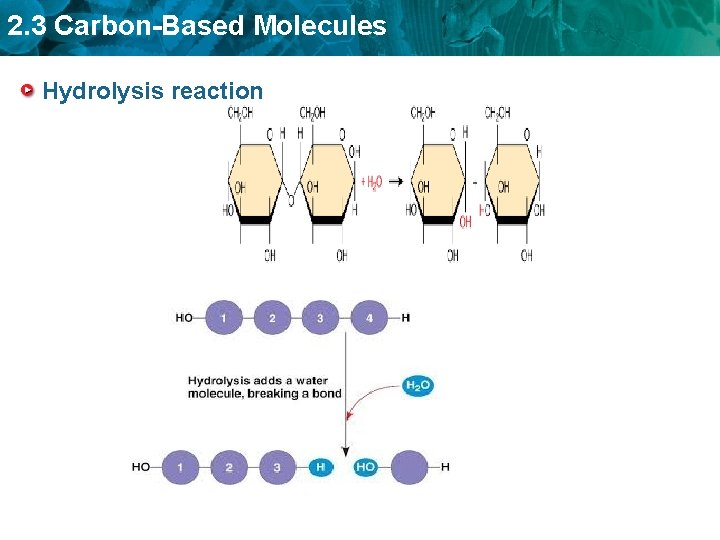
2. 3 Carbon-Based Molecules Hydrolysis reaction
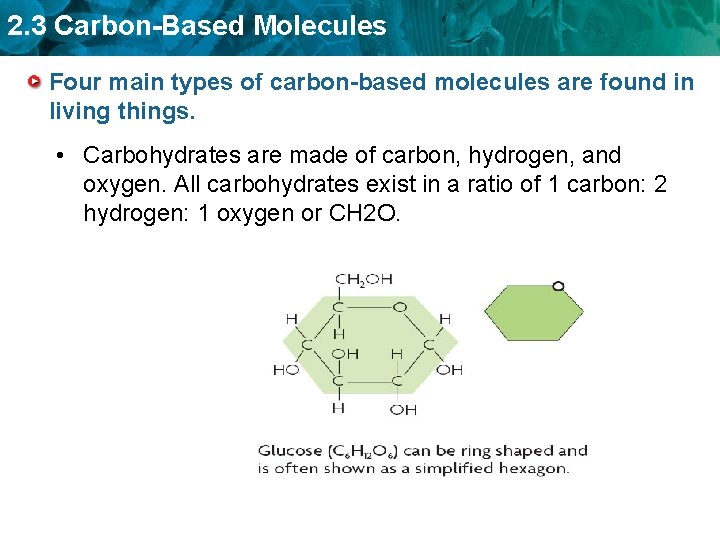
2. 3 Carbon-Based Molecules Four main types of carbon-based molecules are found in living things. • Carbohydrates are made of carbon, hydrogen, and oxygen. All carbohydrates exist in a ratio of 1 carbon: 2 hydrogen: 1 oxygen or CH 2 O.

2. 3 Carbon-Based Molecules Four main types of carbon-based molecules are found in living things. • Carbohydrates are made of carbon, hydrogen, and oxygen. – Carbohydrates include sugars and starches. – Monosaccharides are simple sugars. – Polysaccharides include starches, cellulose, and glycogen.
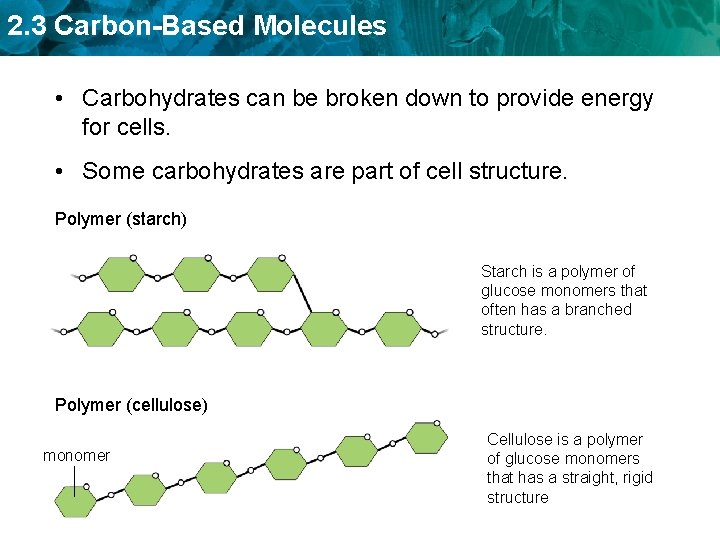
2. 3 Carbon-Based Molecules • Carbohydrates can be broken down to provide energy for cells. • Some carbohydrates are part of cell structure. Polymer (starch) Starch is a polymer of glucose monomers that often has a branched structure. Polymer (cellulose) monomer Cellulose is a polymer of glucose monomers that has a straight, rigid structure

2. 3 Carbon-Based Molecules • The polysaccharide cellulose is a major component of the tough wall of plant cells • Like starch, cellulose is a polymer of glucose, but the glycosidic linkages differ • The difference is based on two ring forms for glucose: alpha ( ) and beta ( ) • Cellulose in human food passes through the digestive tract as insoluble fiber • Some microbes use enzymes to digest cellulose • Many herbivores, from cows to termites, have symbiotic relationships with these microbes
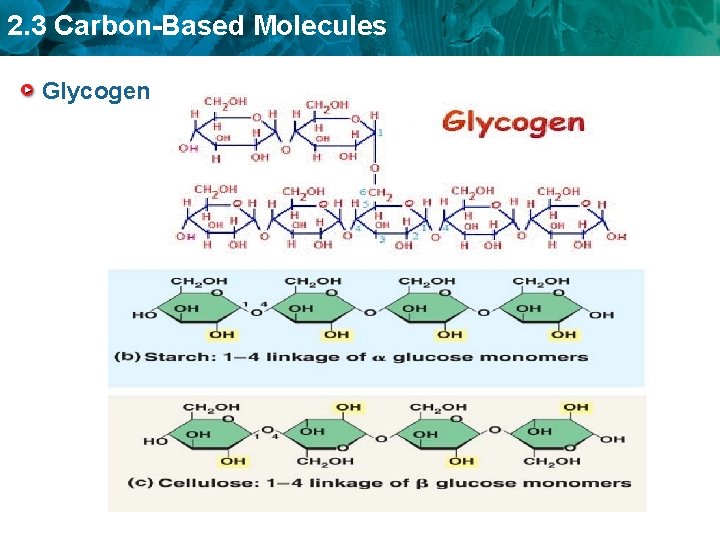
2. 3 Carbon-Based Molecules Glycogen

2. 3 Carbon-Based Molecules • Glycogen is a storage polysaccharide in animals • Humans and other vertebrates store glycogen mainly in liver and muscle cells
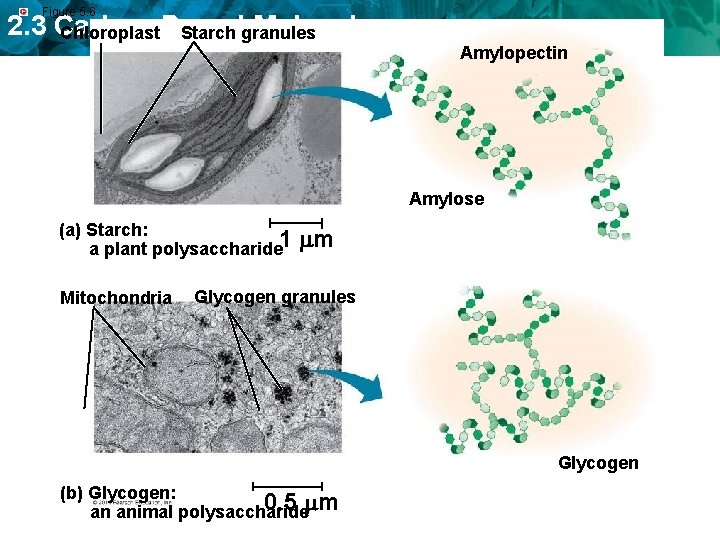
Figure 5. 6 2. 3 Carbon-Based Molecules Chloroplast Starch granules Amylopectin Amylose (a) Starch: a plant polysaccharide 1 Mitochondria m Glycogen granules Glycogen (b) Glycogen: 0. 5 m an animal polysaccharide

2. 3 Carbon-Based Molecules • Chitin, another structural polysaccharide, is found in the exoskeleton of arthropods • Chitin also provides structural support for the cell walls of many fungi

2. 3 Carbon-Based Molecules

Figure 5. 9 2. 3 Carbon-Based Molecules The structure of the chitin monomer Chitin forms the exoskeleton of arthropods. Chitin is used to make a strong and flexible surgical thread that decomposes after the wound or incision heals.
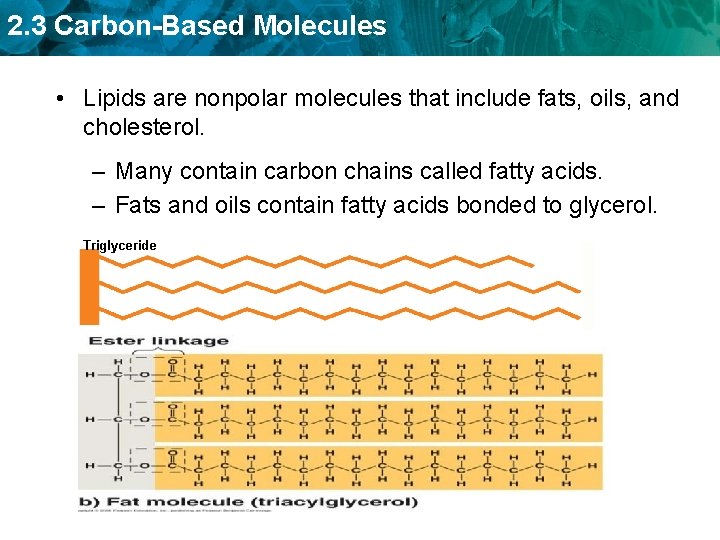
2. 3 Carbon-Based Molecules • Lipids are nonpolar molecules that include fats, oils, and cholesterol. – Many contain carbon chains called fatty acids. – Fats and oils contain fatty acids bonded to glycerol. Triglyceride
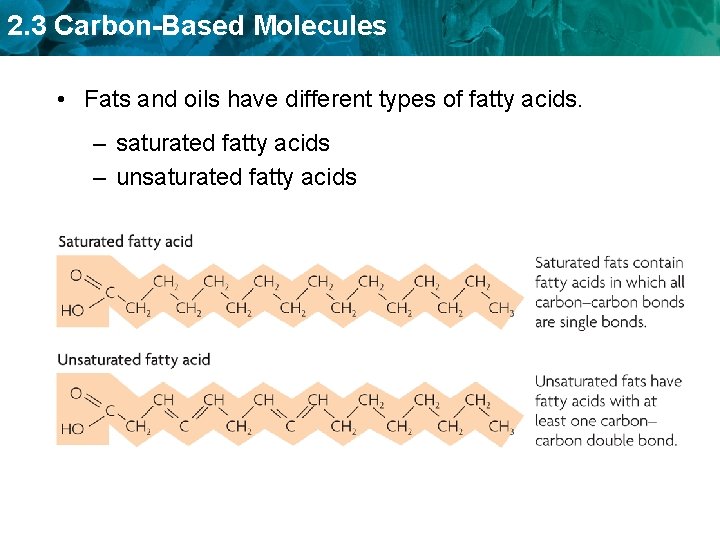
2. 3 Carbon-Based Molecules • Fats and oils have different types of fatty acids. – saturated fatty acids – unsaturated fatty acids

2. 3 Carbon-Based Molecules Saturated fatty acids
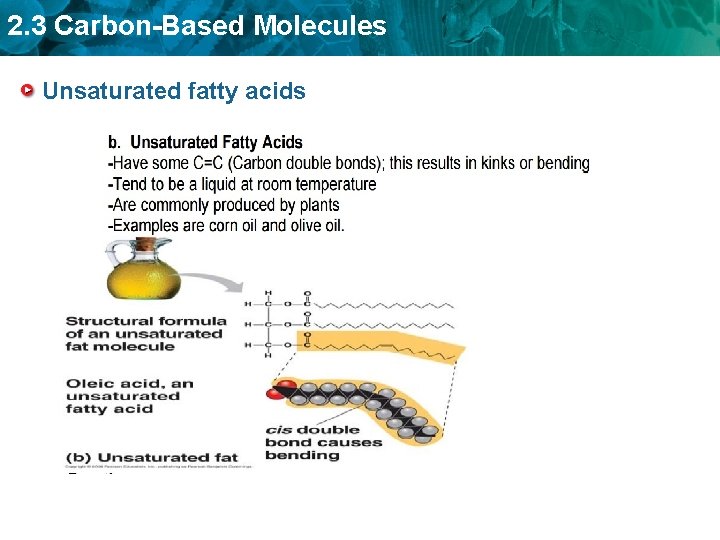
2. 3 Carbon-Based Molecules Unsaturated fatty acids

2. 3 Carbon-Based Molecules • Phospholipids make up all cell membranes. – Polar phosphate “head” – Nonpolar fatty acid “tails”/two fatty acid tails – Arranged in bilayer in forming the cell membrane, with the hydrophilic heads pointing toward the watery cytosol or extra-cellular environment, and they hydrophobic tails sandwiched in between. Phospholipid
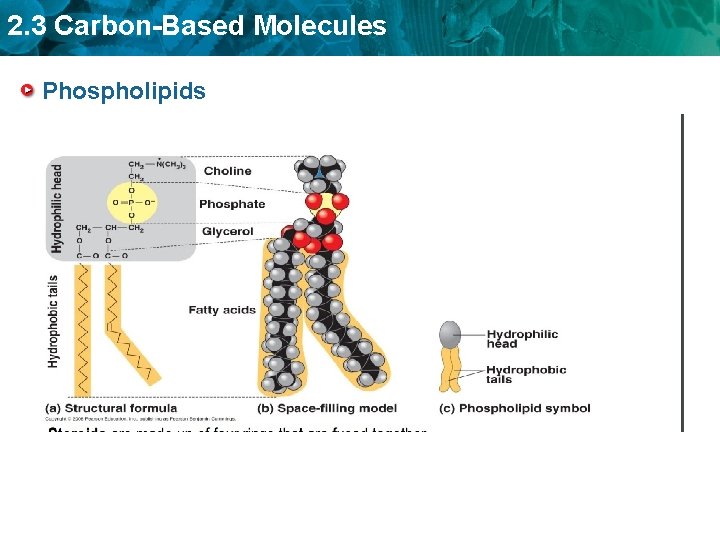
2. 3 Carbon-Based Molecules Phospholipids
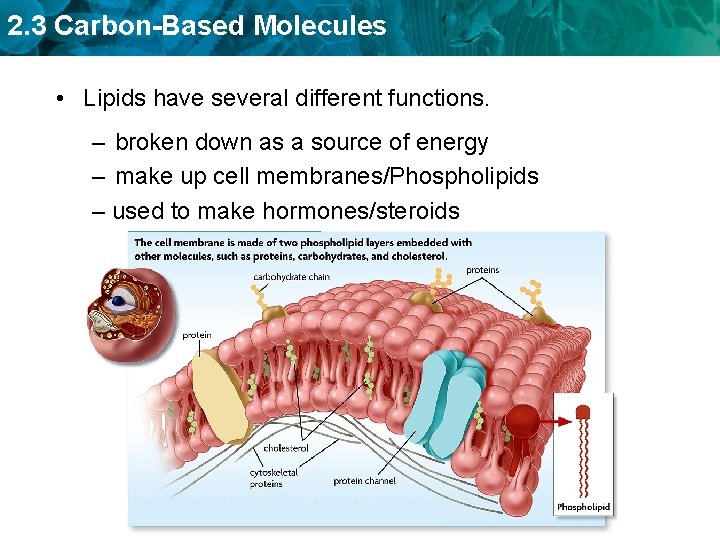
2. 3 Carbon-Based Molecules • Lipids have several different functions. – broken down as a source of energy – make up cell membranes/Phospholipids – used to make hormones/steroids
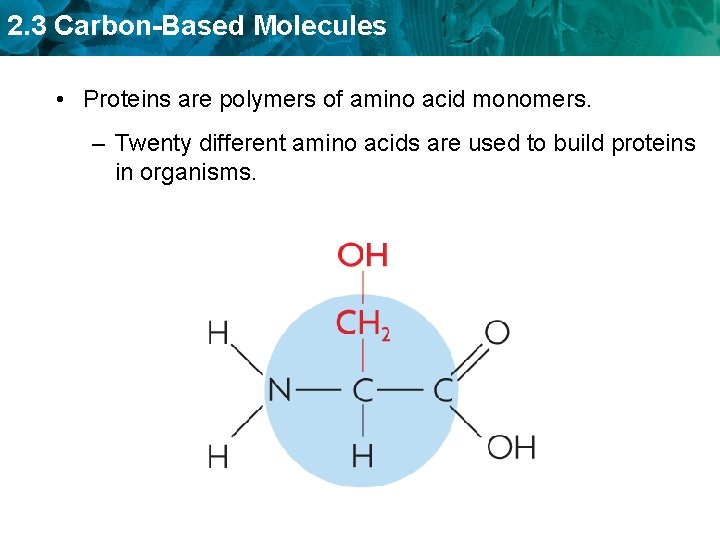
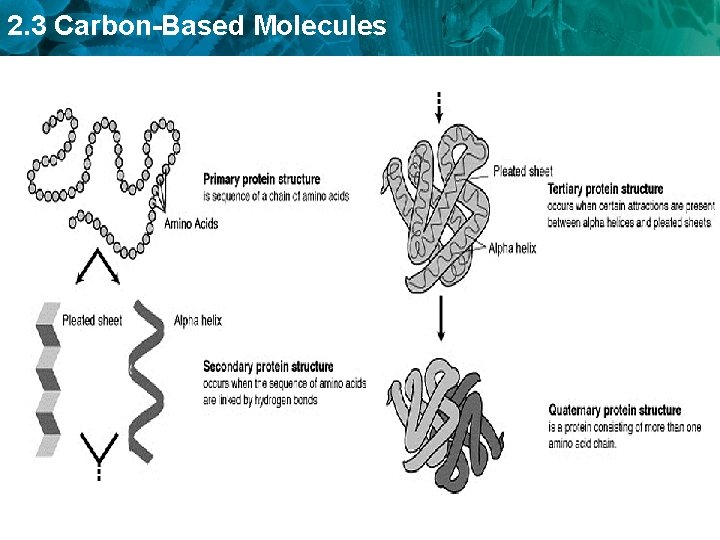
2. 3 Carbon-Based Molecules • Proteins are polymers of amino acid monomers. – Twenty different amino acids are used to build proteins in organisms.
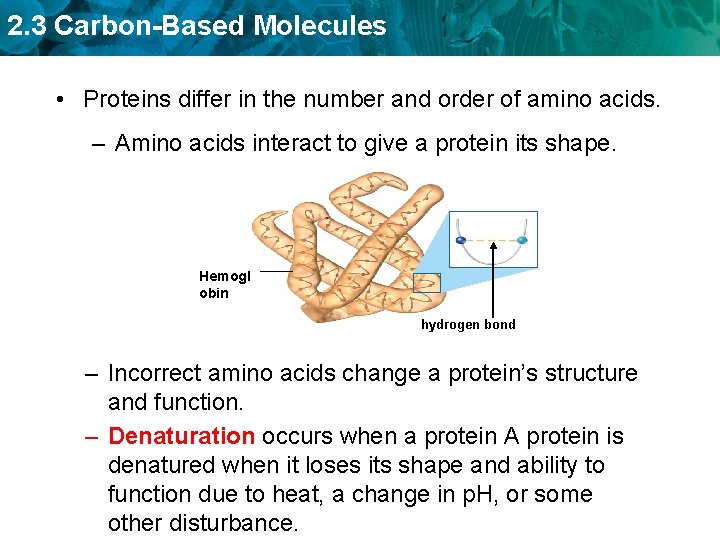
2. 3 Carbon-Based Molecules • Proteins differ in the number and order of amino acids. – Amino acids interact to give a protein its shape. Hemogl obin hydrogen bond – Incorrect amino acids change a protein’s structure and function. – Denaturation occurs when a protein A protein is denatured when it loses its shape and ability to function due to heat, a change in p. H, or some other disturbance.
2. 3 Carbon-Based Molecules
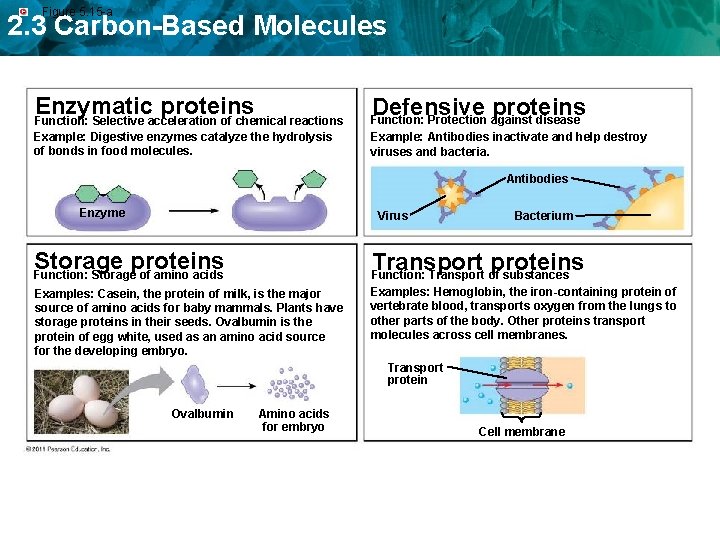
Figure 5. 15 -a 2. 3 Carbon-Based Molecules Enzymatic proteins Function: Selective acceleration of chemical reactions Example: Digestive enzymes catalyze the hydrolysis of bonds in food molecules. Defensive proteins Function: Protection against disease Example: Antibodies inactivate and help destroy viruses and bacteria. Antibodies Enzyme Virus Bacterium Storage proteins Transport proteins Function: Transport of substances Examples: Casein, the protein of milk, is the major source of amino acids for baby mammals. Plants have storage proteins in their seeds. Ovalbumin is the protein of egg white, used as an amino acid source for the developing embryo. Examples: Hemoglobin, the iron-containing protein of vertebrate blood, transports oxygen from the lungs to other parts of the body. Other proteins transport molecules across cell membranes. Function: Storage of amino acids Transport protein Ovalbumin Amino acids for embryo Cell membrane
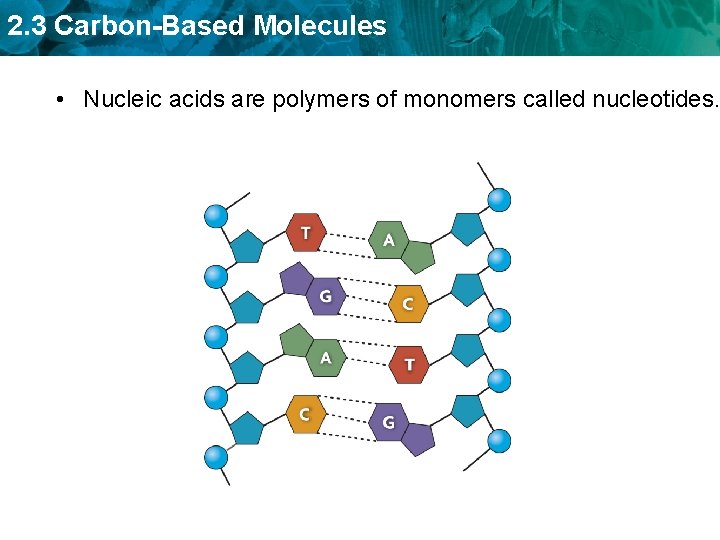
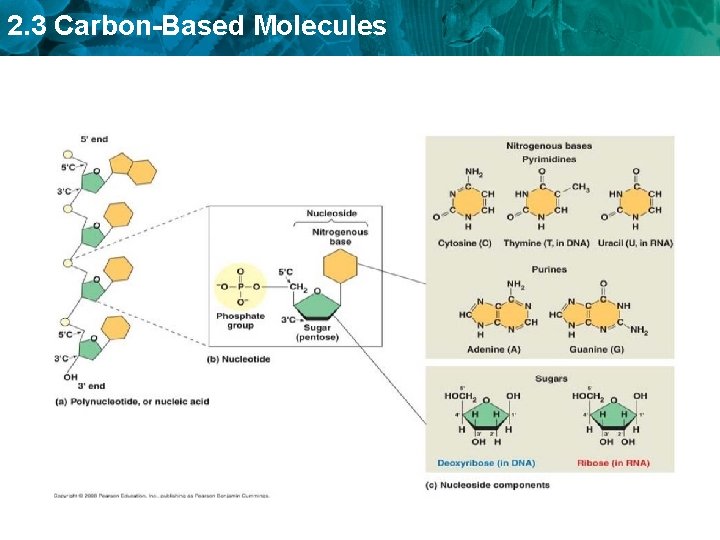
2. 3 Carbon-Based Molecules • Nucleic acids are polymers of monomers called nucleotides.
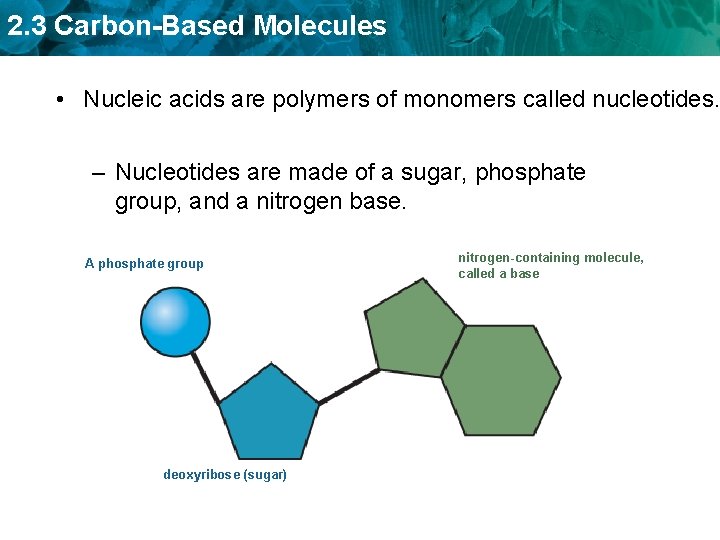
2. 3 Carbon-Based Molecules • Nucleic acids are polymers of monomers called nucleotides. – Nucleotides are made of a sugar, phosphate group, and a nitrogen base. A phosphate group deoxyribose (sugar) nitrogen-containing molecule, called a base
2. 3 Carbon-Based Molecules
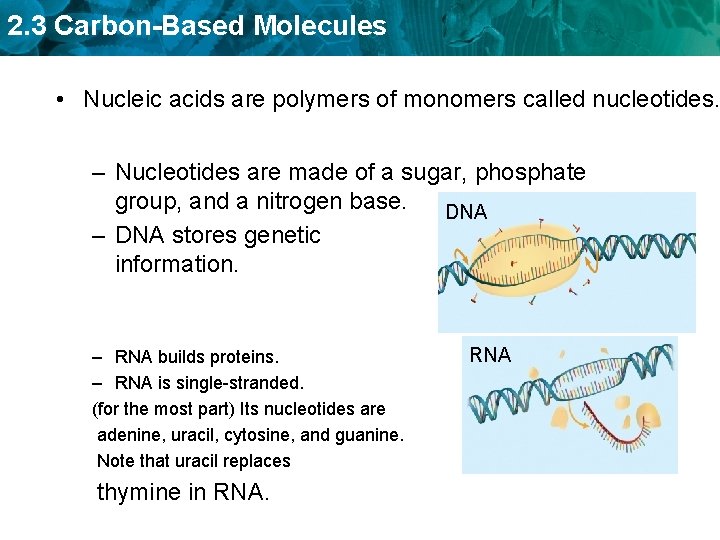
2. 3 Carbon-Based Molecules • Nucleic acids are polymers of monomers called nucleotides. – Nucleotides are made of a sugar, phosphate group, and a nitrogen base. DNA – DNA stores genetic information. – RNA builds proteins. – RNA is single-stranded. (for the most part) Its nucleotides are adenine, uracil, cytosine, and guanine. Note that uracil replaces thymine in RNA
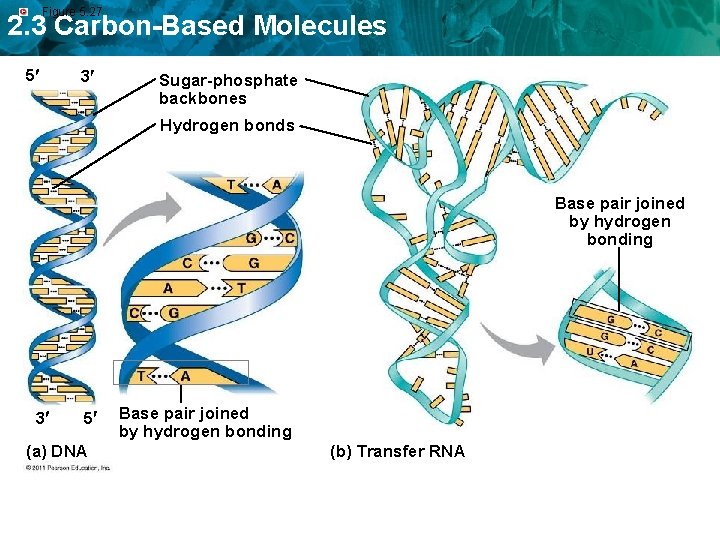
Figure 5. 27 2. 3 Carbon-Based Molecules 5 3 Sugar-phosphate backbones Hydrogen bonds Base pair joined by hydrogen bonding 3 5 (a) DNA Base pair joined by hydrogen bonding (b) Transfer RNA

2. 3 Carbon-Based Molecules DNA and Proteins as Tape Measures of Evolution • The linear sequences of nucleotides in DNA molecules are passed from parents to offspring • Two closely related species are more similar in DNA than are more distantly related species • Molecular biology can be used to assess evolutionary kinship © 2011 Pearson Education, Inc.
- Slides: 34