2 3 CarbonBased Molecules KEY CONCEPT Carbonbased molecules

2. 3 Carbon-Based Molecules KEY CONCEPT Carbon-based molecules are the foundation of life.

2. 3 Carbon-Based Molecules Draw a mark at the midpoint of a sheet of paper along the side edge. Then fold the top and bottom edges in to touch the midpoint.

2. 3 Carbon-Based Molecules Fold in half from side to side.
2. 3 Carbon-Based Molecules Open and cut along the inside fold lines to form four tabs.
2. 3 Carbon-Based Molecules Label each tab.

2. 3 Carbon-Based Molecules • As you read Section 2. 3, draw the structure and list the characteristics of carbohydrates, lipids, proteins, and nucleic acids under the appropriate tabs.
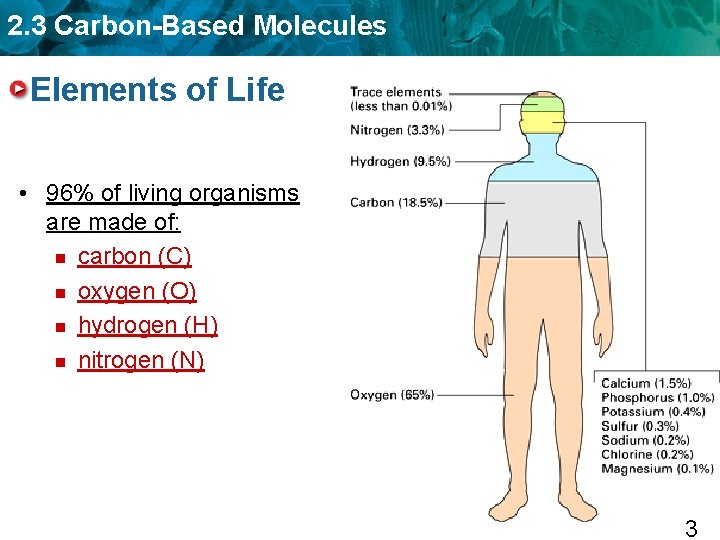
2. 3 Carbon-Based Molecules Elements of Life • 96% of living organisms are made of: n carbon (C) n oxygen (O) n hydrogen (H) n nitrogen (N) 3

2. 3 Carbon-Based Molecules of Life • Put C, H, O, N together in different ways to build living organisms • What are bodies made of? – carbohydrates - sugars & starches – proteins – fats (lipids) – nucleic acids - DNA, RNA 4

2. 3 Carbon-Based Molecules Don’t forget water • Water – 65% of your body is H 2 O – water is inorganic - doesn’t contain carbon • Rest of you is made of carbon molecules – organic molecules - carbohydrates proteins fats nucleic acids 5
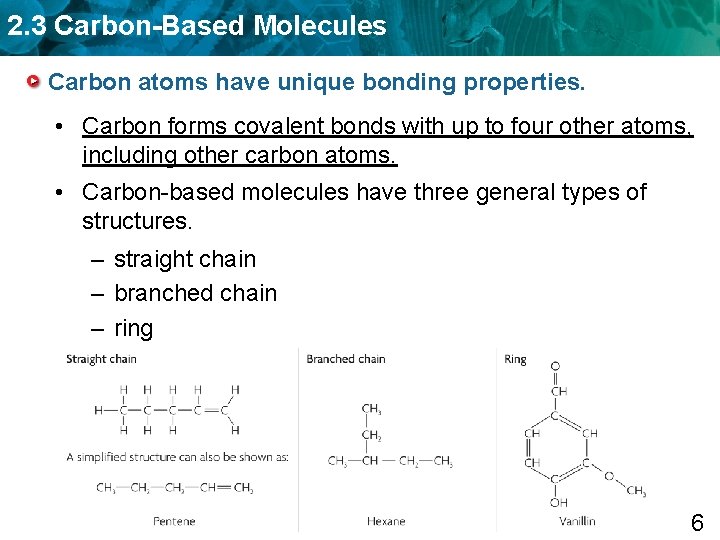
2. 3 Carbon-Based Molecules Carbon atoms have unique bonding properties. • Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. • Carbon-based molecules have three general types of structures. – straight chain – branched chain – ring 6
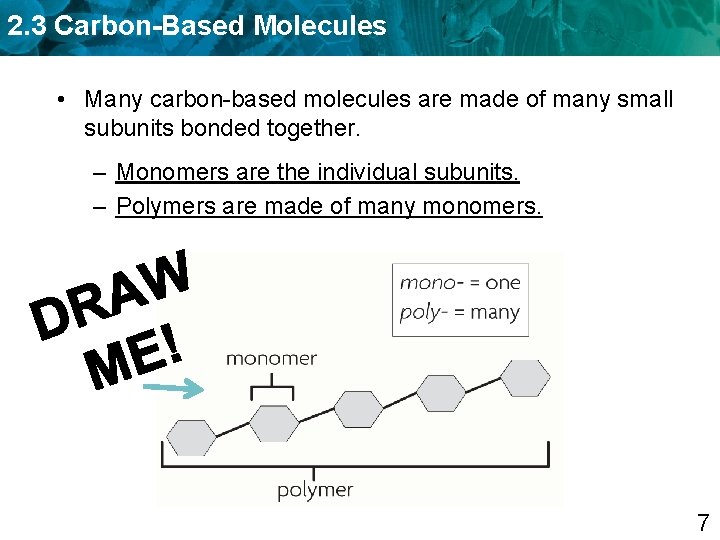
2. 3 Carbon-Based Molecules • Many carbon-based molecules are made of many small subunits bonded together. – Monomers are the individual subunits. – Polymers are made of many monomers. 7
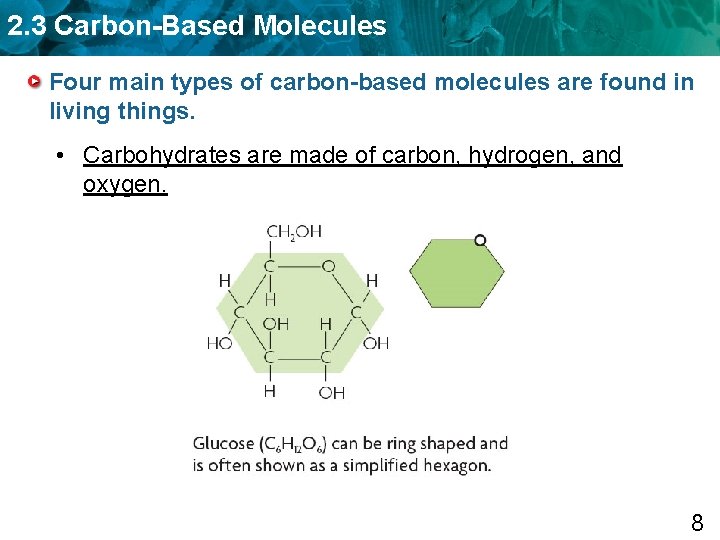
2. 3 Carbon-Based Molecules Four main types of carbon-based molecules are found in living things. • Carbohydrates are made of carbon, hydrogen, and oxygen. 8

2. 3 Carbon-Based Molecules Four main types of carbon-based molecules are found in living things. • Carbohydrates are made of carbon, hydrogen, and oxygen. – Carbohydrates include sugars and starches. – Monosaccharides are simple sugars. (monomer) – Polysaccharides include starches, cellulose, and glycogen. (polymer) 9
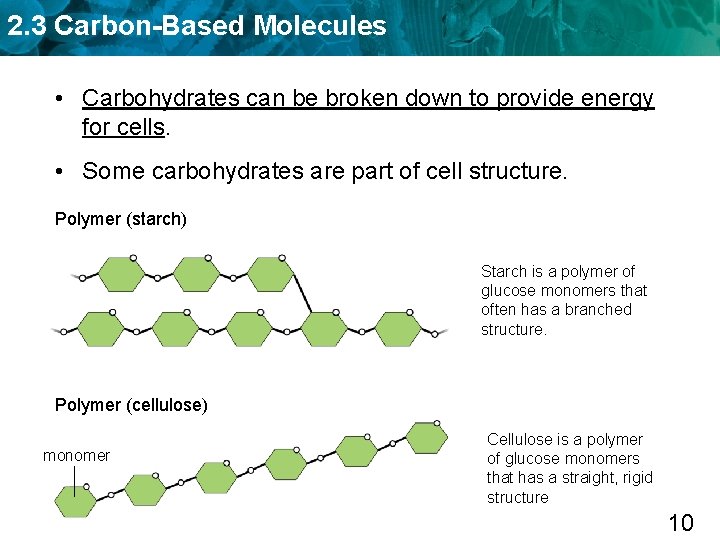
2. 3 Carbon-Based Molecules • Carbohydrates can be broken down to provide energy for cells. • Some carbohydrates are part of cell structure. Polymer (starch) Starch is a polymer of glucose monomers that often has a branched structure. Polymer (cellulose) monomer Cellulose is a polymer of glucose monomers that has a straight, rigid structure 10

2. 3 Carbon-Based Molecules • Lipids are nonpolar molecules that include fats, oils, and cholesterol. – Many contain carbon chains called fatty acids. – Fats and oils contain fatty acids bonded to glycerol. Triglyceride 11
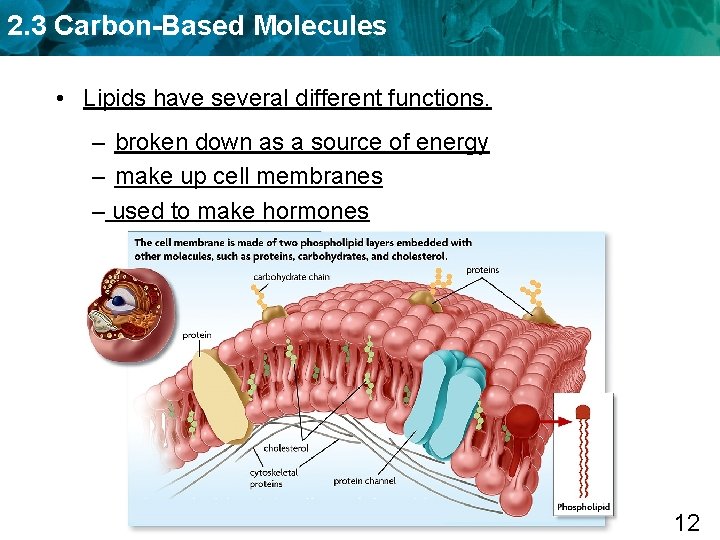
2. 3 Carbon-Based Molecules • Lipids have several different functions. – broken down as a source of energy – make up cell membranes – used to make hormones 12
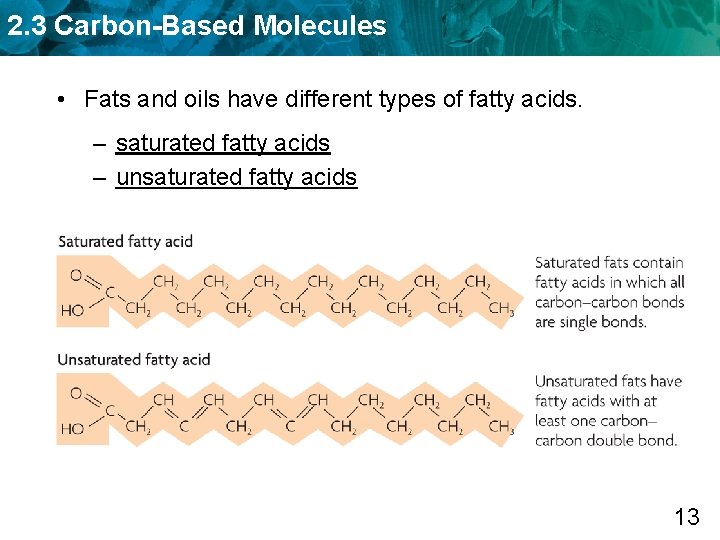
2. 3 Carbon-Based Molecules • Fats and oils have different types of fatty acids. – saturated fatty acids – unsaturated fatty acids 13
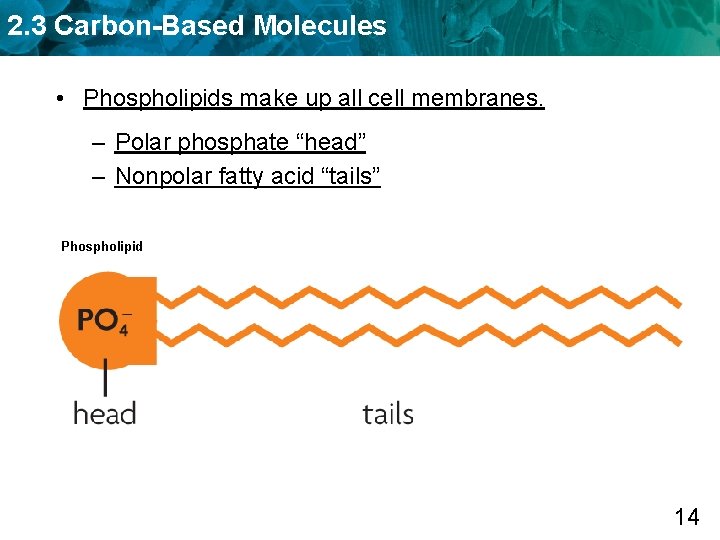
2. 3 Carbon-Based Molecules • Phospholipids make up all cell membranes. – Polar phosphate “head” – Nonpolar fatty acid “tails” Phospholipid 14
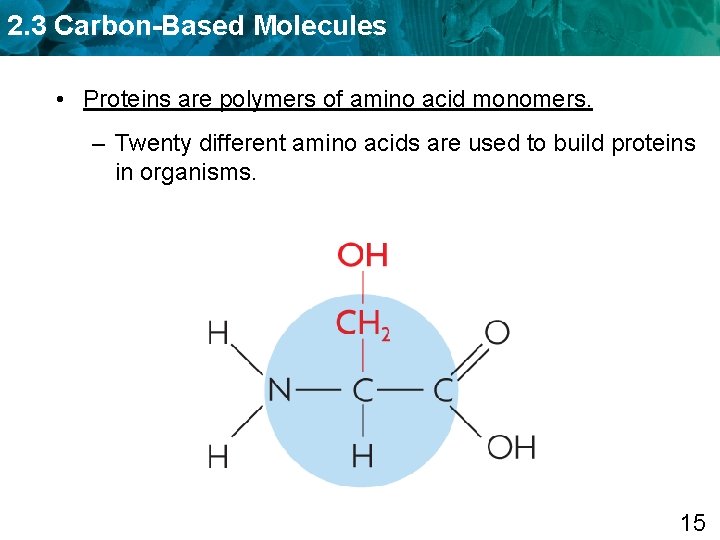
2. 3 Carbon-Based Molecules • Proteins are polymers of amino acid monomers. – Twenty different amino acids are used to build proteins in organisms. 15
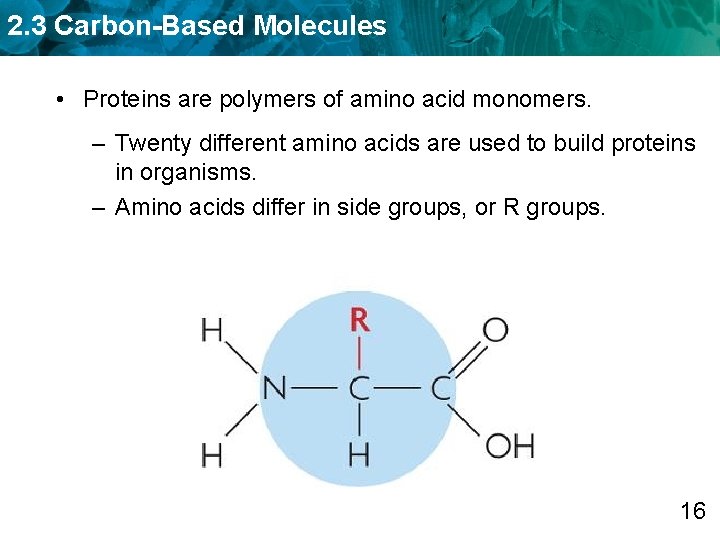
2. 3 Carbon-Based Molecules • Proteins are polymers of amino acid monomers. – Twenty different amino acids are used to build proteins in organisms. – Amino acids differ in side groups, or R groups. 16
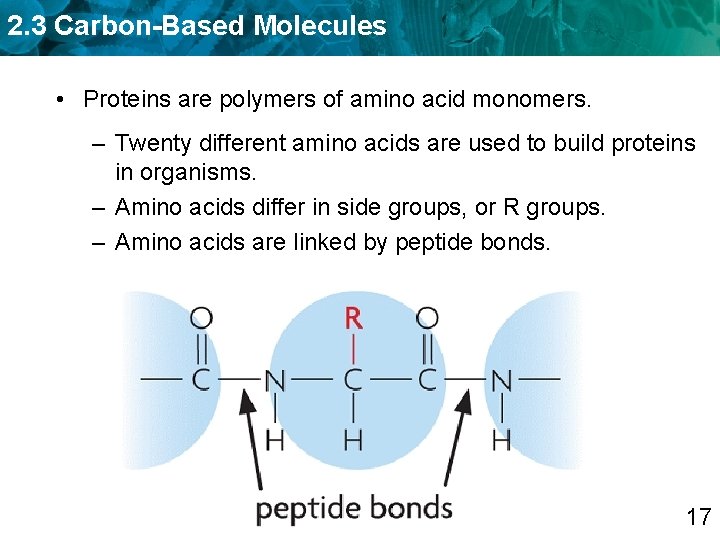
2. 3 Carbon-Based Molecules • Proteins are polymers of amino acid monomers. – Twenty different amino acids are used to build proteins in organisms. – Amino acids differ in side groups, or R groups. – Amino acids are linked by peptide bonds. 17
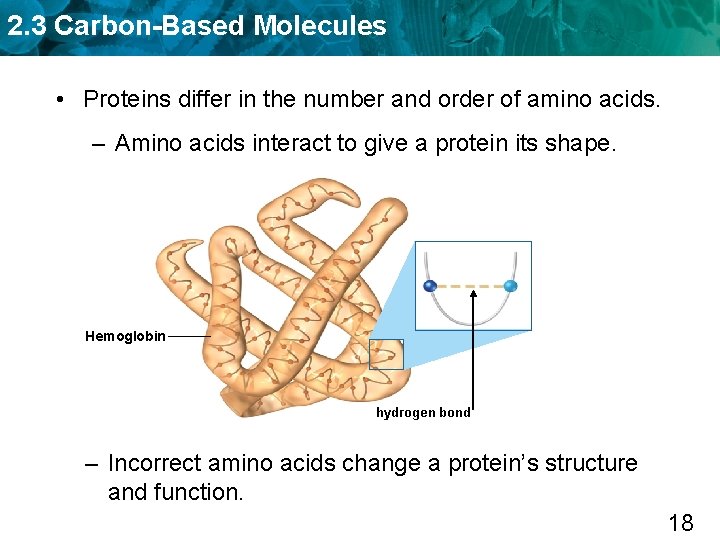
2. 3 Carbon-Based Molecules • Proteins differ in the number and order of amino acids. – Amino acids interact to give a protein its shape. Hemoglobin hydrogen bond – Incorrect amino acids change a protein’s structure and function. 18
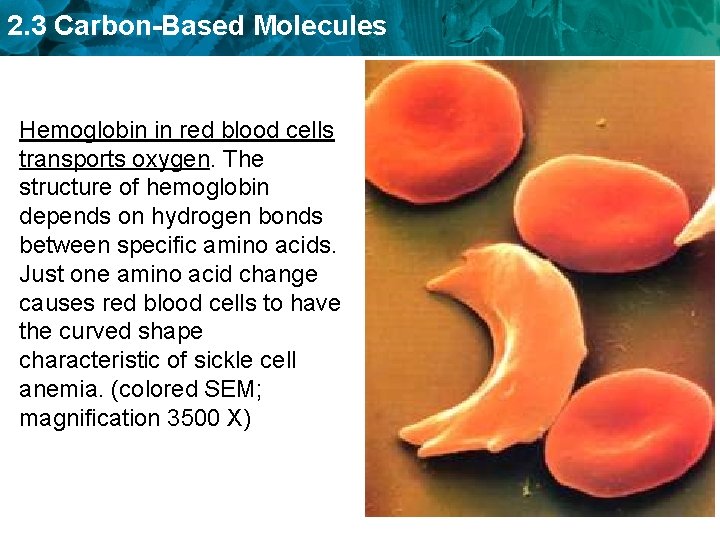
2. 3 Carbon-Based Molecules Hemoglobin in red blood cells transports oxygen. The structure of hemoglobin depends on hydrogen bonds between specific amino acids. Just one amino acid change causes red blood cells to have the curved shape characteristic of sickle cell anemia. (colored SEM; magnification 3500 X)

2. 3 Carbon-Based Molecules Functions of proteins • Functions—many, including enzymes, oxygen transport, and muscle movement
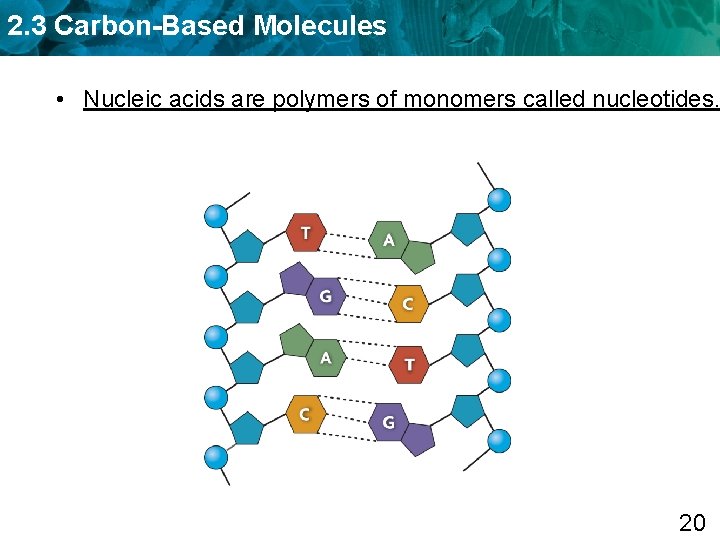
2. 3 Carbon-Based Molecules • Nucleic acids are polymers of monomers called nucleotides. 20
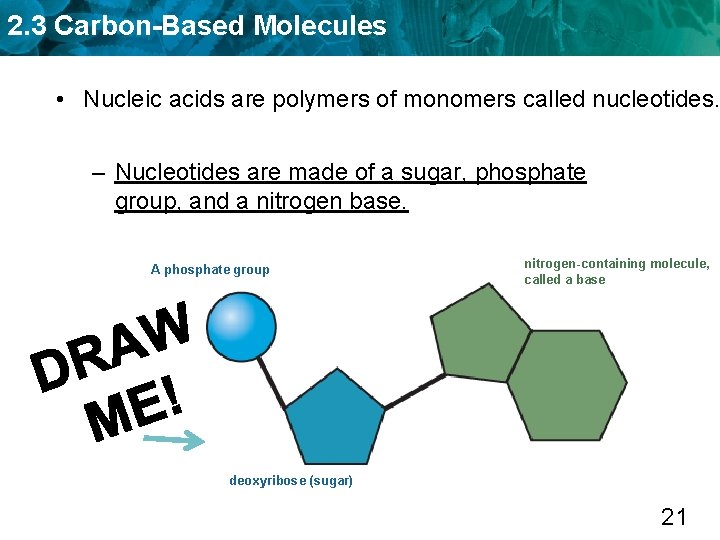
2. 3 Carbon-Based Molecules • Nucleic acids are polymers of monomers called nucleotides. – Nucleotides are made of a sugar, phosphate group, and a nitrogen base. A phosphate group nitrogen-containing molecule, called a base deoxyribose (sugar) 21
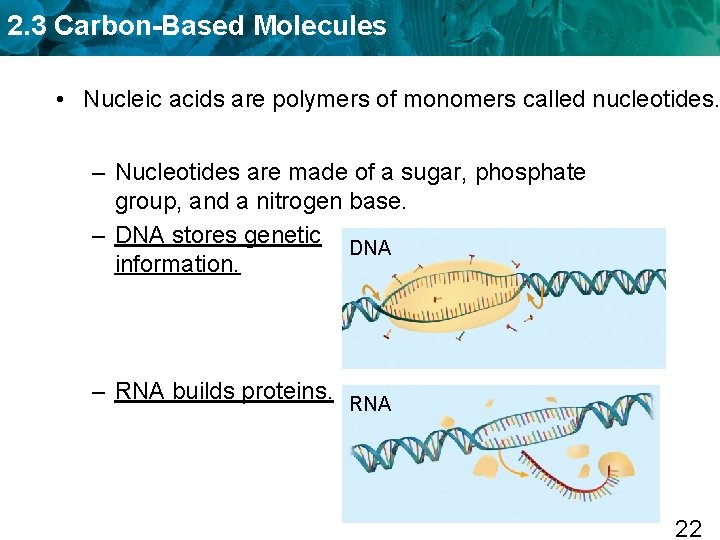
2. 3 Carbon-Based Molecules • Nucleic acids are polymers of monomers called nucleotides. – Nucleotides are made of a sugar, phosphate group, and a nitrogen base. – DNA stores genetic DNA information. – RNA builds proteins. RNA 22

2. 3 Carbon-Based Molecules Reteach - Identifying Variables - SC. 912. N. 1. 1 • A scientist studied the effect of jogging on the number of Calories used. People jogged for three different lengths of time - 10 minutes, 20 minutes, 30 minutes. The number of Calories used was measured, recorded, and plotted on a bar graph. • What are the independent variables? • What are the dependent variables? 23

2. 3 Carbon-Based Molecules Review • To reinforce the concept of polymers(many) made from repeating monomers(one). Complete the following chart: Monomer Polymer

2. 3 Carbon-Based Molecules Check your answers • To reinforce the concept of polymers(many) made from repeating monomers(one). Complete the following chart: Monomer- (subunits) Polymer (macro molecules) Monosaccharide (simple sugars) polysaccharides starchescellulose Amino acids proteins Nucleotides nucleic acids Fatty acids lipids(triglycerides) 24
- Slides: 30