2 2 What are some ways to describe

2. 2 What are some ways to describe matter? Concept 2: Matter can be described by its chemical properties Concept 3: Matter can be described based on physical and chemical changes Name: _______________ Date: _______________

Chemical Properties: Qualitative Observations • Matter can also be described using chemical properties • Chemical property describes the ability of matter to react. • Chemical properties can only be observed in a chemical reaction. • Reactivity with acids: • Some substances react vigorously with acids and others do not • Example: baking soda and vinegar produce a gas

Chemical Properties: Qualitative Observations • Reactivity with oxygen: • Substances in some foods react with oxygen when exposed to air • Example: avocadoes turning brown • Combustibility: • Some material will catch fire and burn in the air • Example: burning wood • Lack of reactivity: • Some substances do not react with other substances are known to be “inert” • Example: helium in balloons

Matter can be described based on physical and chemical changes. Chemical Change: cooking toast Physical Change: Boiling Water Chemical and Physical Change: cooking bacon Refer to page 121 in the textbook

Describing Matter: Physical Changes • Physical Change is a change that alters a substance without changing its chemical identity or composition. • Ex: crumpling up a piece of aluminum foil, folding a piece of paper into a bird • Can you think of another example of physical change? • _______________________ • Changing the state of matter of a substance is also a physical change • Example: freezing water into ice, boiling water until it forms steam.

Describing Matter: Chemical Changes • A chemical change can also be known as a chemical reaction • In a chemical reaction we have reactants and products reactants products • During chemical changes, new substance are going to be produced • Example: • steel forming rust • food rotting • gasoline producing smoke

Describing Matter: Chemical Changes • Can you give an example of a chemical change? • _____________________ • Evidence of new substances forming: • Change in colour • Change in texture • New smells • Release of gas • Change of energy in the form of heat

Law of Conservation of Mass • Antoine and Marie Anne Lavoisier carried out many chemical reactions and found that the mass does not change during a chemical reaction. • The law of conservation of mass states: mass of the reactants = mass of the products Antoine and Marie Anne Lavoisier

Law of Conservation of Mass
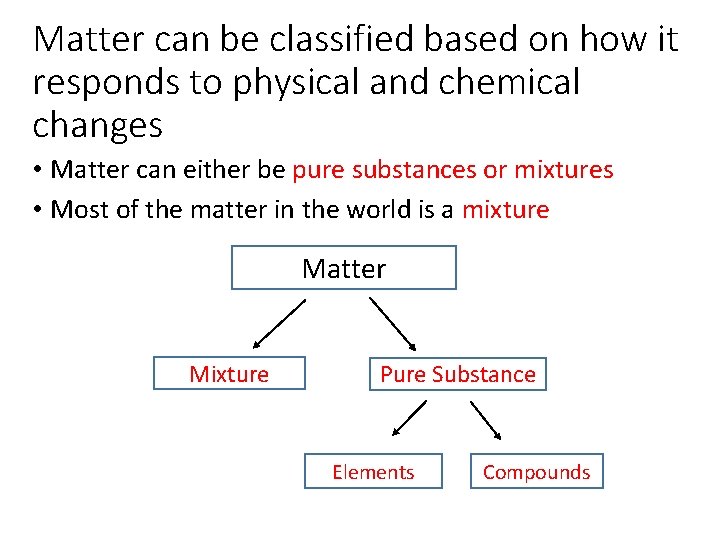
Matter can be classified based on how it responds to physical and chemical changes • Matter can either be pure substances or mixtures • Most of the matter in the world is a mixture Matter Mixture Pure Substance Elements Compounds

Types of Matter: Mixtures • A mixture is two or more different kinds of particles • Example: salt water • Example: iron and sand • Salt can be separated from mixture water by evaporation • Iron can be separated from sand by using a magnet

Types of Matter: Pure Substances • In a pure substance, all the particles look alike • There are two different kinds of pure substances Compounds Elements CAN be broken down further into elements by chemical means CANNOT be broken down further Examples: water (H 2 O), carbon dioxide (CO 2), Glucose (C 6 H 12 O 6) Examples: solid gold, solid silver Diagrams:
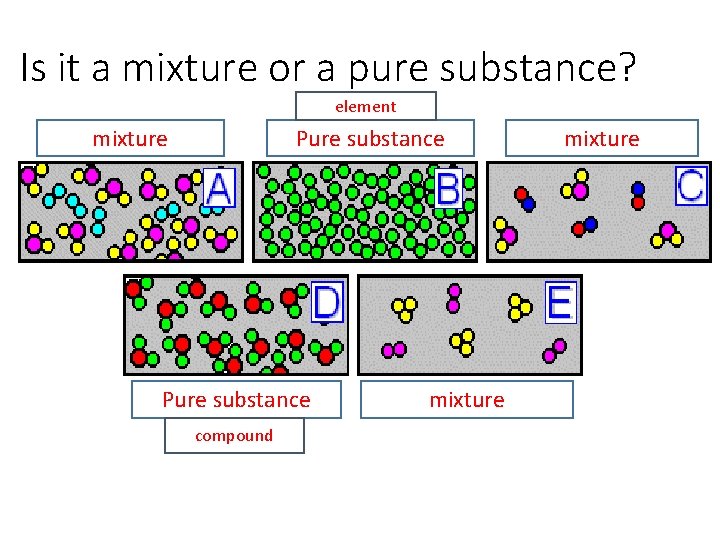
Is it a mixture or a pure substance? element mixture Pure substance compound mixture

Is it a mixture or a pure substance? Pure substance Pure Substance compound 1 2 Mixture 3 Mixture 4
- Slides: 14