2 11 interaction between adsorbate and metal 1

§ 2. 11 interaction between adsorbate and metal (1) Hydrophobic interaction (intermolecular reaction— non-covalent interaction , surface activity); (2) Electrostatic and chemical interaction (coordination); (3) Interaction between adsorbate molecules; (aggregate, island, crystallization) (4) replacement of adsorbed water molecules Different from gaseous adsorption

1) Difference between adsorption on gas/solid interface and solution/electrode interface Nearly all dissoluble organic compounds has more or less surface activity at the solution/electrode interface, i. e. , can adsorb on the interface. At solution/electrode interface, adsorbed species have to replace the formerly adsorbed water molecules and become contact physically or chemically with the electrode surface. RHslon + n. H 2 Oad RHad + n. H 2 Osoln

2) Adsorption isotherms: Isotherm describes the relationship between the activity (ai) of a species in solution and the amount of material adsorbed per unit area on electrode surface ( i). The isotherms are developed from the condition that the electrochemical potential for the adsorbed and solution species are equal:

The general form of an isotherm.

3) Langmuir isotherm Assumptions: 1) no interaction between species; i. e. , Gad is not a function of coverage. 2) No heterogeneity of surface, all surface sites are equal. 3) there is a maximum coverage, (monolayer) Surface concentration
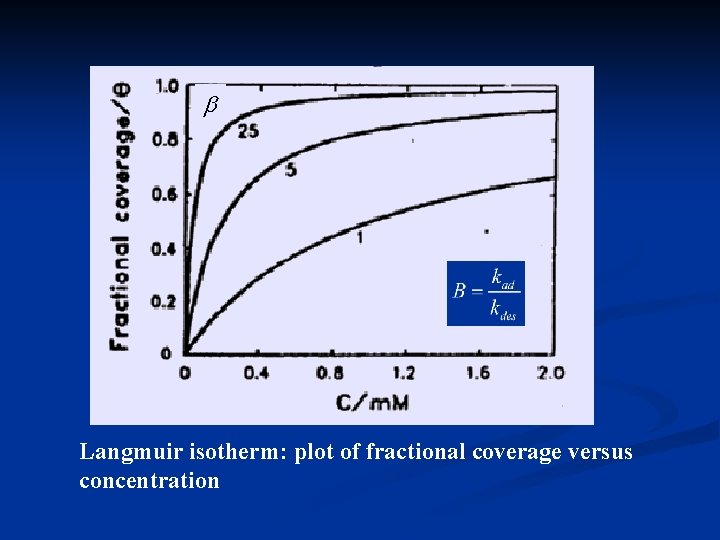
Langmuir isotherm: plot of fractional coverage versus concentration
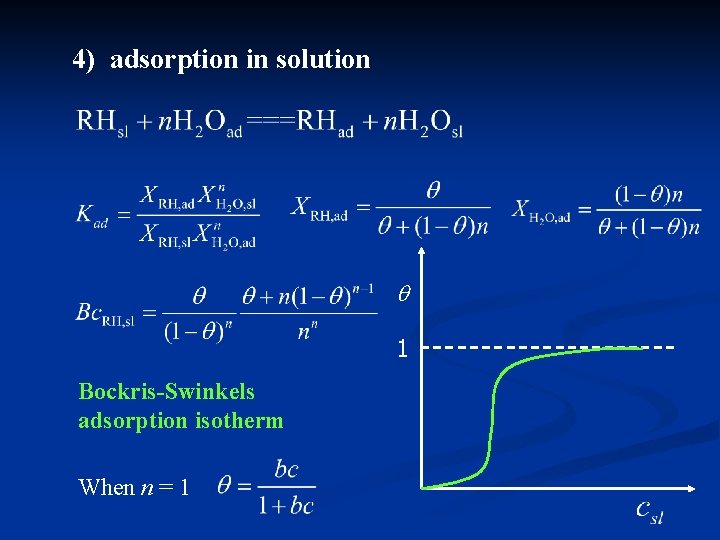
4) adsorption in solution 1 Bockris-Swinkels adsorption isotherm When n = 1

RHslon + n. H 2 Oad RHad + n. H 2 Osoln Bockris-Swinkels adsorption isotherm Frumkin adsorption isotherm Water concentration
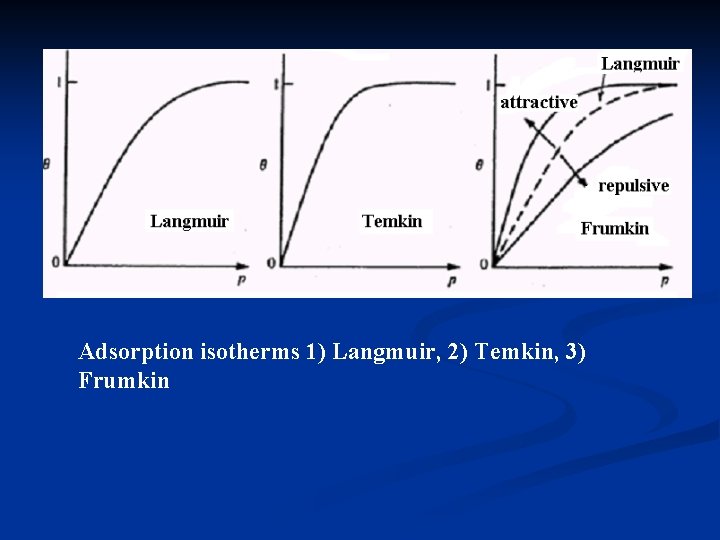
Adsorption isotherms 1) Langmuir, 2) Temkin, 3) Frumkin
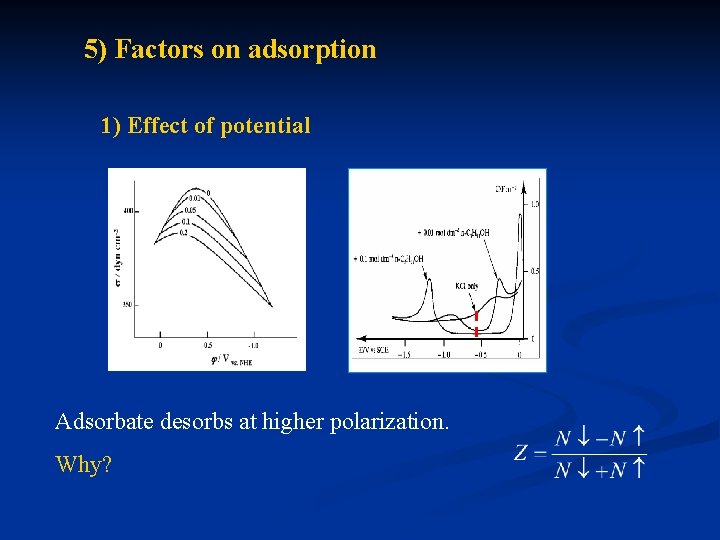
5) Factors on adsorption 1) Effect of potential Adsorbate desorbs at higher polarization. Why?

Frumkin Bockris -dipole moment of water molecule, E-electric field strength in compact layer, factor of intermolecular interaction, n- number of water molecule replaced by organic adsorbant. Z-orientation coefficient
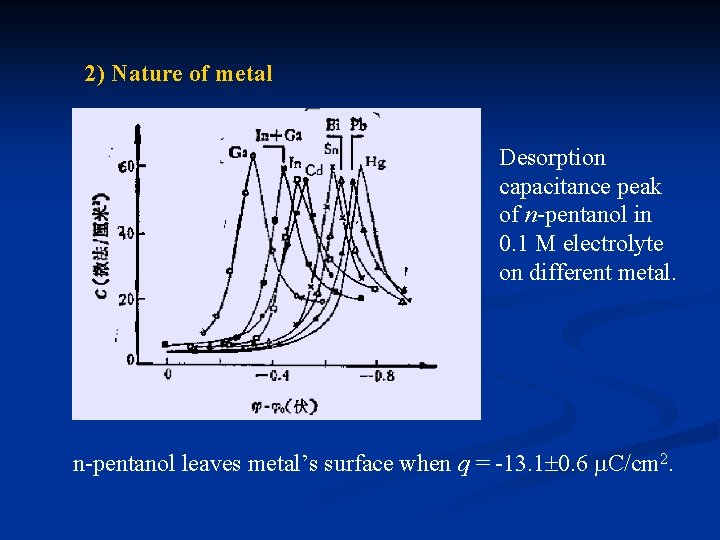
2) Nature of metal Desorption capacitance peak of n-pentanol in 0. 1 M electrolyte on different metal. n-pentanol leaves metal’s surface when q = -13. 1 0. 6 C/cm 2.

Attention: Because surfactant can affect the surface state of electrode significantly, therefore, the purification of solution used for electrochemical study is very important. Deionized water triple distilled water

Homework 1. The PZC measured in Na. F and Na. Cl are different, try to make explanation to this difference and make comparison between their values. 2. Describe the variation of surface excess of cation and anion in KBr solution with respect to rational potential and give explanation. 3. Why PZC can be only measured on metals with high hydrogen overpotential? 4. Adsorbates with aromatic rings can adsorbed on electrode surface with different configuration, explain the effect of potential on their configuration.

Chapter 3 transport phenomena in electrolytic systems and concentration overpotential
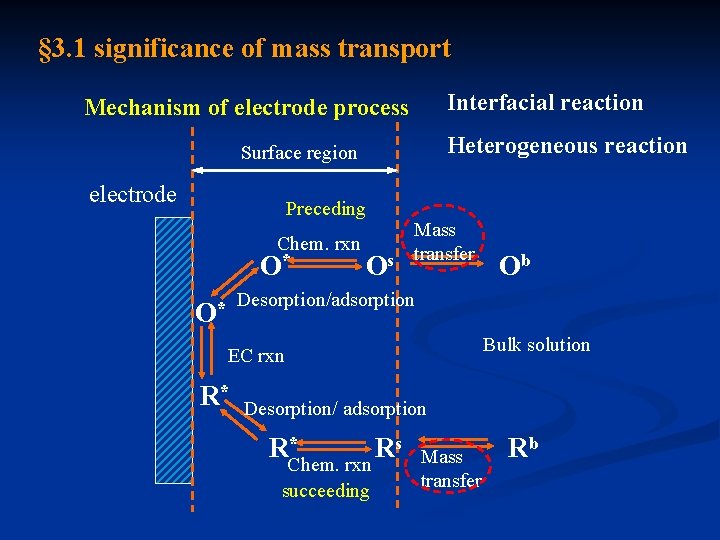
§ 3. 1 significance of mass transport Interfacial reaction Mechanism of electrode process Heterogeneous reaction Surface region electrode Preceding Mass transfer Os Chem. rxn O* Ob Desorption/adsorption * O Bulk solution EC rxn R* Desorption/ adsorption * RChem. rxn Rs succeeding Mass transfer Rb
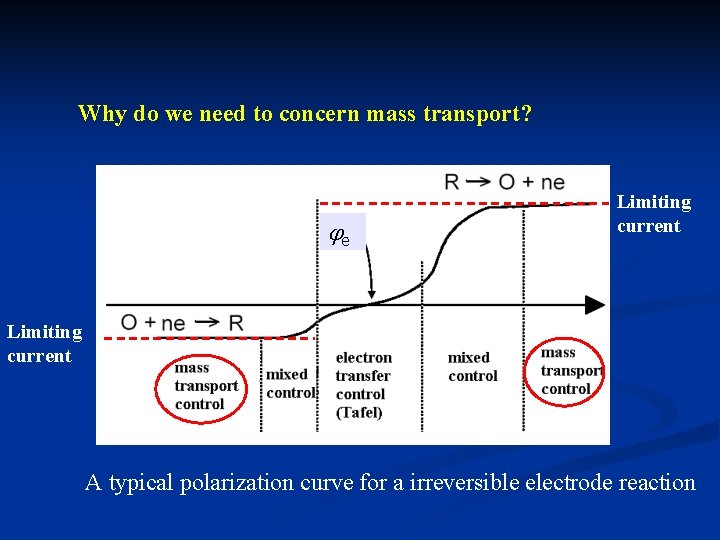
Why do we need to concern mass transport? e Limiting current A typical polarization curve for a irreversible electrode reaction
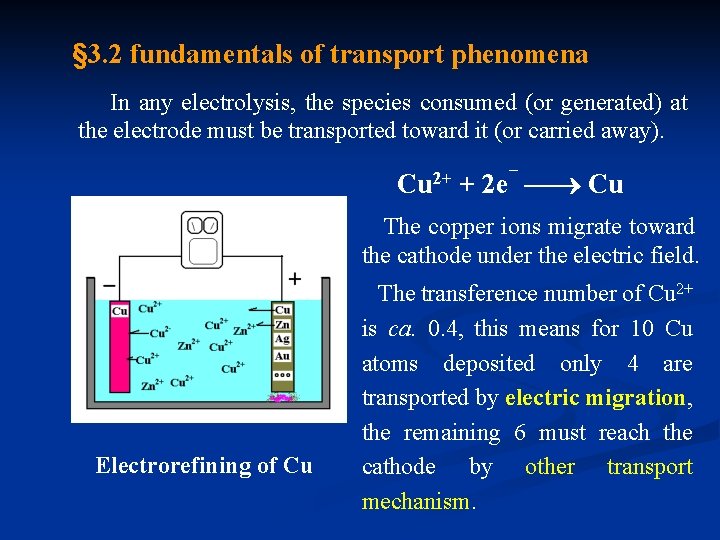
§ 3. 2 fundamentals of transport phenomena In any electrolysis, the species consumed (or generated) at the electrode must be transported toward it (or carried away). Cu 2+ + 2 e¯ Cu The copper ions migrate toward the cathode under the electric field. Electrorefining of Cu The transference number of Cu 2+ is ca. 0. 4, this means for 10 Cu atoms deposited only 4 are transported by electric migration, the remaining 6 must reach the cathode by other transport mechanism.

1 Flux density and velocity Driving force: gradients of the electrochemical potential a physical parameter with dimension of Newton per mole is a force.

For a certain species j, consider two points in the solution, r and s (an infinitesimal distance form one another ) and: This difference of over a distance can arise because of differences of concentration (or activity) of species j at r and s (a concentration gradient) or because of difference of at r and s (an electric field or potential gradient). In general, a flux of species j will occur to alleviate this difference of . The flux, J (mol s-1 cm-2), is proportional to the gradient, grad or of . or

2 Basic concepts transport modes: 1) electric migration 2) diffusion 3) convection

1) Electric migration B A C movement of charged species under electric field due to electrostatic interaction (potential gradient) C - , Z - , U- ; C + , Z + , U+ ; species move with respect to liquid. flux Ex strength of electric field, u 0 ionic mobility, (3. 1)
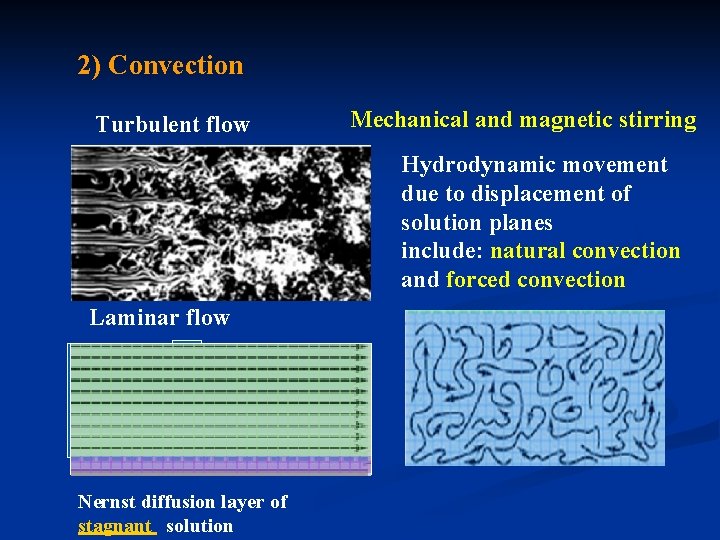
2) Convection Turbulent flow Mechanical and magnetic stirring Hydrodynamic movement due to displacement of solution planes include: natural convection and forced convection Laminar flow Nernst diffusion layer of stagnant solution

Convection: species move with liquid due to density different (density gradient) caused by either concentration or temperature difference. (3. 2)

3) Diffusion: A molecular mode of transport which tends to equalize concentrations and which is set up only if concentration difference / gradient exist. species move with respect to liquid Fick’s first law for stationary state D diffusion coefficient, ( c/ x) concentration gradient.
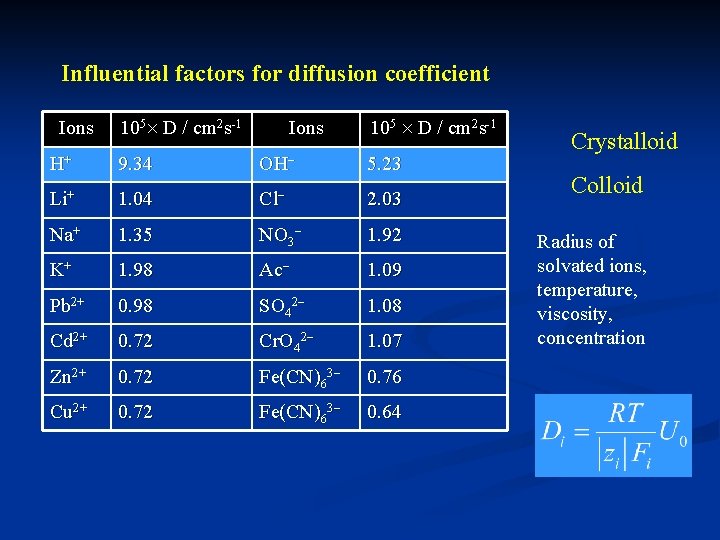
Influential factors for diffusion coefficient Ions 105 D / cm 2 s-1 Ions 105 D / cm 2 s-1 9. 34 OH 5. 23 Li+ 1. 04 Cl 2. 03 Na+ 1. 35 NO 3 1. 92 K+ 1. 98 Ac 1. 09 Pb 2+ 0. 98 SO 42 1. 08 Cd 2+ 0. 72 Cr. O 42 1. 07 Zn 2+ 0. 72 Fe(CN)63 0. 76 Cu 2+ 0. 72 Fe(CN)63 0. 64 H+ Crystalloid Colloid Radius of solvated ions, temperature, viscosity, concentration

Theoretical calculation of diffusion co-efficient Leveling effect Diffusion coefficient is a function of concentration.

Extension of diffusion layer c 0 After the electrolysis current is switched on, the diffusion layer is set up and extends progressively toward the interior of the solution; i. e. , its thickness of diffusion layer grows with time. depleted layer – diffusion layer

diffusion Microscopic, molecule by molecule transport mechanism. Brownian motion, no directionality except that resulting form a concentration gradient. Movement of a species under the influence of a gradient of chemical potential (i. e. , a concentration gradient) migration Microscopic, molecule by molecule transport mechanism, driven by electric field. Has directionality governed by field. Movement of a charged body under the influence of an electric field (a gradient of electrical potential) convection Macroscopic (segmented by solution element) transport mechanism, directional, based on external forces. Stirring or hydrodynamic transport, but also natural convection (convection caused by density gradients) and forced convection.

transport modes: 1) electric migration 2) diffusion 3) convection

3 simplification of mass transport The general equation for mass transport It is rather complicated.
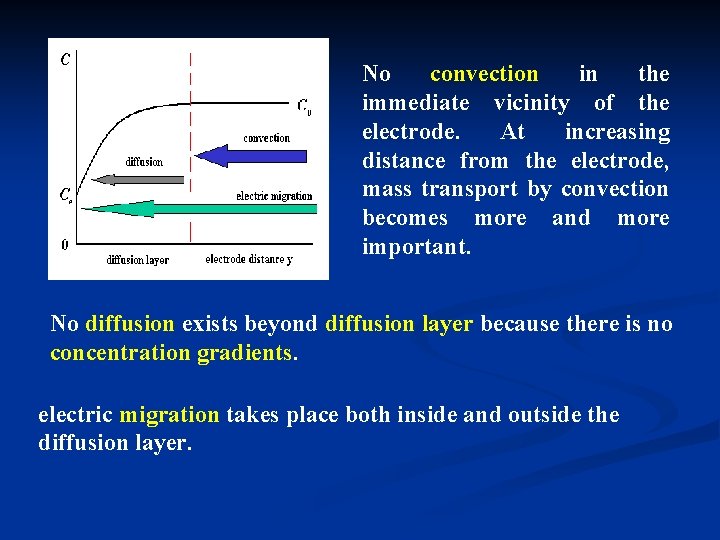
No convection in the immediate vicinity of the electrode. At increasing distance from the electrode, mass transport by convection becomes more and more important. No diffusion exists beyond diffusion layer because there is no concentration gradients. electric migration takes place both inside and outside the diffusion layer.

Elimination of electric migration: When supporting electrolyte of high concentration (nonelectroactive species) was added (10 times more than electroactive species), the mass transport due to electric migration of electroactive species can be neglected. Elimination of convection: At immediate vicinity to electrode within stationary liquid layer or make mass transport occur in stationary medis, no convection occurs.

§ 3. 4 Diffusion in absence of convection: steady state and nonsteady state 1) Fick’s first law unidirectional diffusion within a isotropic medium, can be generalized to a three-dimentional diffusional flux Where grad or is a vector operator, i is the unit vector along the axis and x is distance. For 3 D mass transfer depends on the choice of a suitable coordinate system for each particular case.

2) Fick’s second law Generally, most diffusional processes of electrochemical interest involve time-dependent concentration characteristics. The amount of species accumulated within the volume element due to the x -directional flux is A B Jx=x+dx D C dx The change of the concentration in the reference volume can be expressed as

For Cartesian coordinates: Fick’s second law The unidirectional diffusion for a real solution For stationary state

3) Stationary state diffusion c 0 Transition state): state (nonsteady the thickness of diffusion layer ( ) grows with time. The stationary state under pure diffusion is only an approximation. At the initiation of diffusion, large variations of either the flux toward the interface or the local concentration are produced. At long times, either the increase or the depletion of the reacting species at the interface causes density gradients, which generate the bulk fluid motions of natural convection.
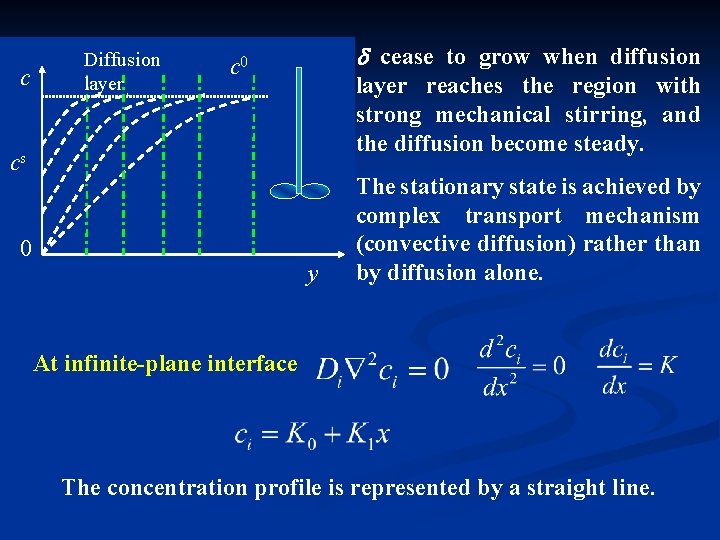
c Diffusion layer cease to grow when diffusion c 0 layer reaches the region with strong mechanical stirring, and the diffusion become steady. cs 0 y The stationary state is achieved by complex transport mechanism (convective diffusion) rather than by diffusion alone. At infinite-plane interface The concentration profile is represented by a straight line.
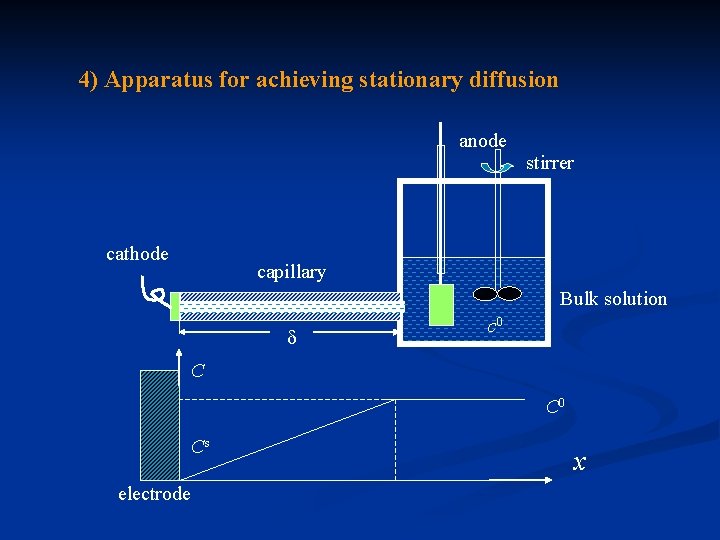
4) Apparatus for achieving stationary diffusion anode stirrer cathode capillary Bulk solution c 0 C C 0 Cs electrode x
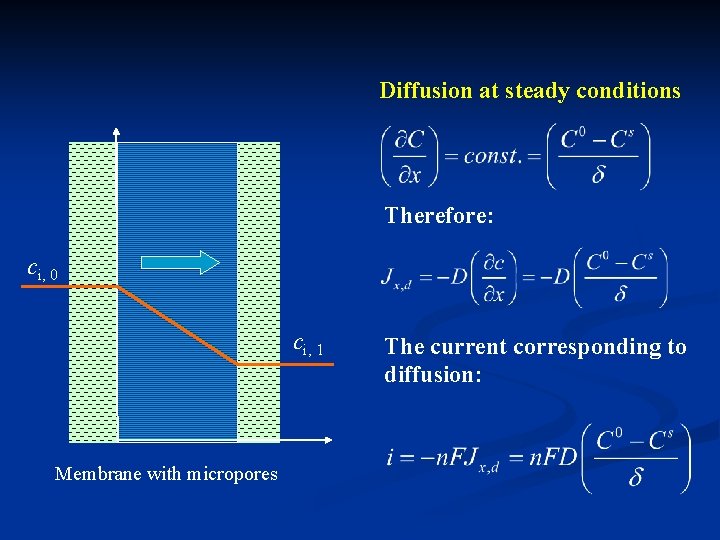
Diffusion at steady conditions Therefore: ci, 0 ci, 1 Membrane with micropores The current corresponding to diffusion:

When the surface concentration becomes 0 (complete concentration polarization), the current reaches maximum – limiting current density The plane-interface diffusion equation can be applied to real systems as long as the diffusion distance is much larger than the microscopic unevenness of the plane. Question: using limiting diffusion current, how to determine D and c 0?
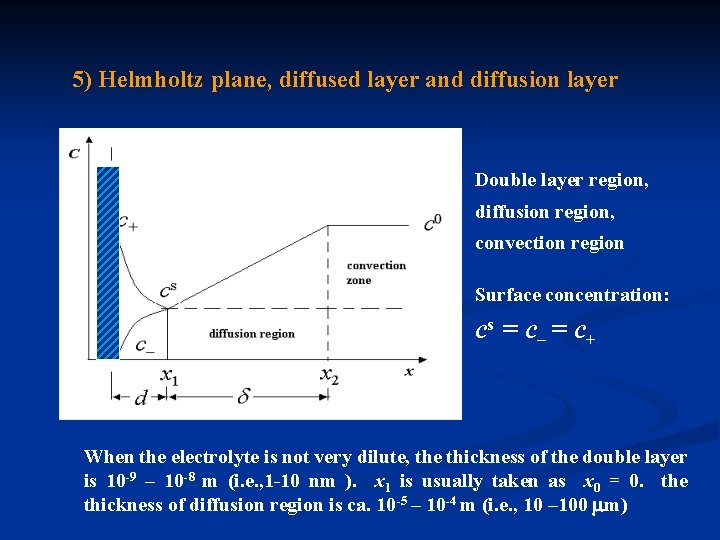
5) Helmholtz plane, diffused layer and diffusion layer Double layer region, diffusion region, convection region Surface concentration: cs = c– = c+ When the electrolyte is not very dilute, the thickness of the double layer is 10 -9 – 10 -8 m (i. e. , 1 -10 nm ). x 1 is usually taken as x 0 = 0. the thickness of diffusion region is ca. 10 -5 – 10 -4 m (i. e. , 10 – 100 m)

3. 5 Diffusion with convection and RDE 1 Diffusion with convection Ideal / nonideal stationary state Difference between stationary and non-stationary states: 1) ci = f(x); ci = f(x, t); 2) the thickness of diffusion layer 3) relativity
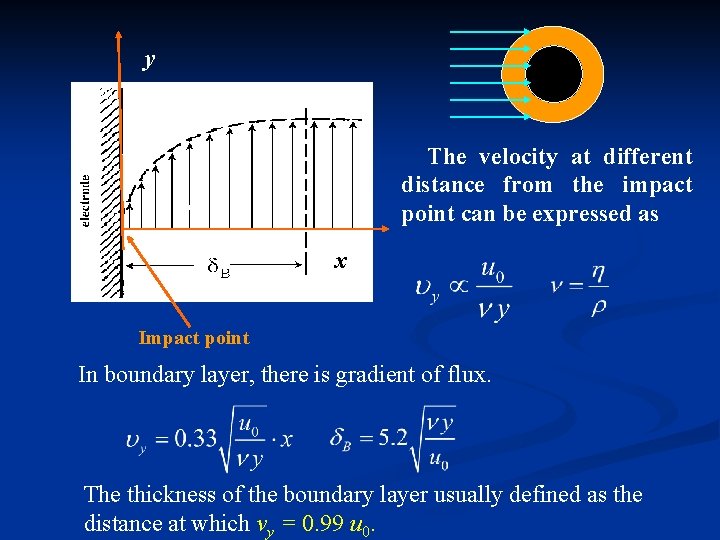
y The velocity at different distance from the impact point can be expressed as x Impact point In boundary layer, there is gradient of flux. The thickness of the boundary layer usually defined as the distance at which vy = 0. 99 u 0.
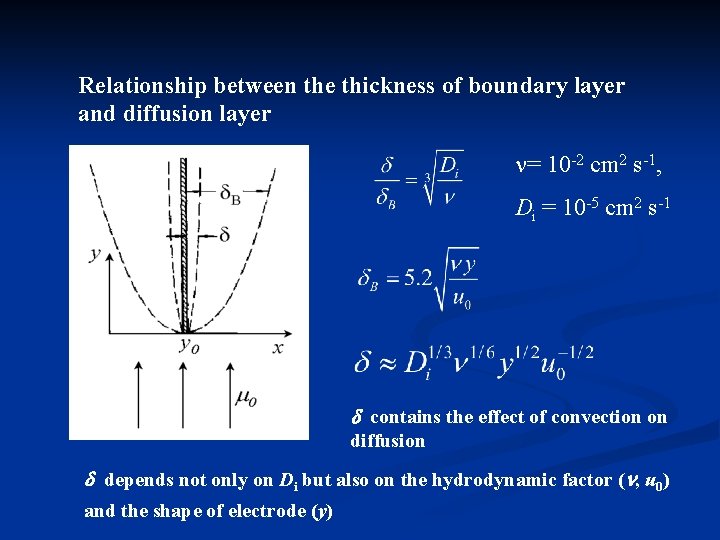
Relationship between the thickness of boundary layer and diffusion layer = 10 -2 cm 2 s-1, Di = 10 -5 cm 2 s-1 contains the effect of convection on diffusion depends not only on Di but also on the hydrodynamic factor ( , u 0) and the shape of electrode (y)
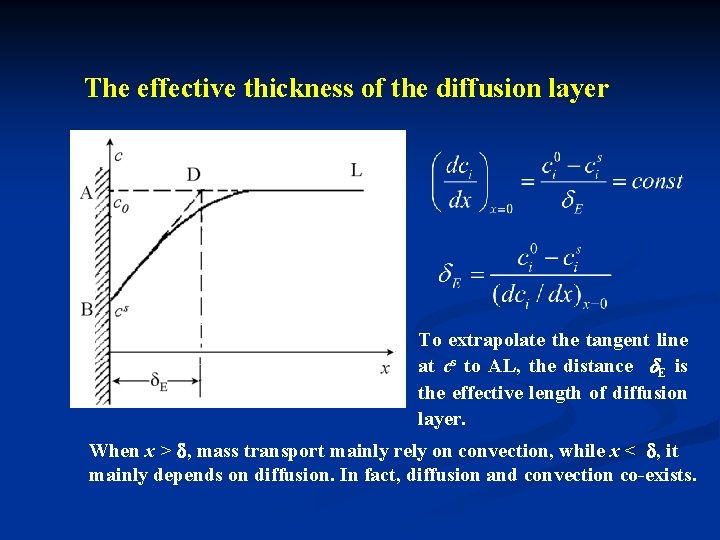
The effective thickness of the diffusion layer To extrapolate the tangent line at cs to AL, the distance E is the effective length of diffusion layer. When x > , mass transport mainly rely on convection, while x < , it mainly depends on diffusion. In fact, diffusion and convection co-exists.

Discussion: jd Di 3/2, there exists convection j = jd + jc jd depends on stirring, viscosity, and position

3. 3. 2 Rotating disc electrode Diffusion under convection: This formula demonstrates that the current density of plane electrode depends significantly on the shape / dimension of the electrode. If this kind of electrode is used to study the kinetics of electrode process, only the statistic/averaged results over the total electrode surface can be obtained.

In 1942, an electrochemist in the former Soviet Union named Levich solved the problem by using rotating disc electrode (RDE) method. PTFE sheathing Embedded metal disc electrode High-speed electric motor The impact point is the center of the disc.
Rotating ring disk electrode (RRDE)

IL = (0. 620)∙z∙F∙A∙D 2/3∙ 1/2∙ – 1/6∙c Levich Equation w speed of rotation (rad∙s-1), kinematic viscosity of the solution (cm 2∙s-1), kinematic viscosity is the ratio between solution viscosity and its specific weight. For pure water: = 0, 0100 cm 2∙s-1, For 1. 0 mol∙dm-3 KNO 3 is = 0, 00916 cm 2∙s-1 (at 20°C). c concentration of electroactive species (in mol. cm-3, note unusual unit) D diffúsion coefficient (cm 2∙s-1), A electrode area in cm 2

r y Impact point As distance from the center increases, thickness of diffusion layer ( ) increases while the u 0 increase which cause decrease of . is a dimensionless parameter related to distance.
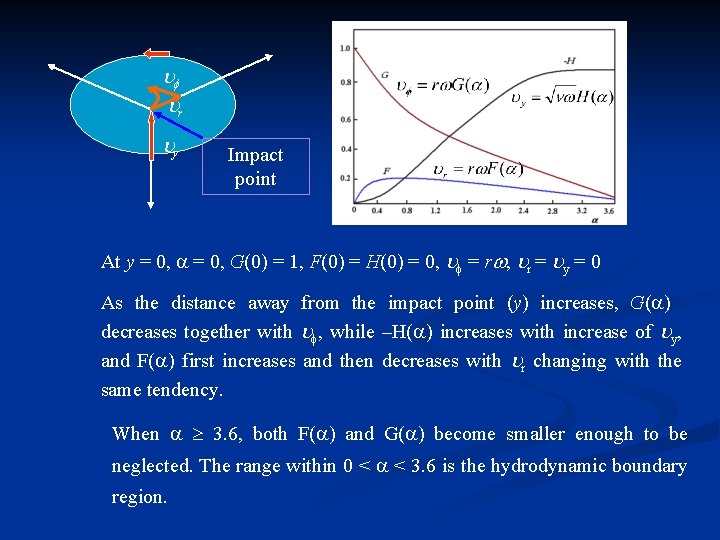
r y Impact point At y = 0, G(0) = 1, F(0) = H(0) = 0, = r , r = y = 0 As the distance away from the impact point (y) increases, G( ) decreases together with , while –H( ) increases with increase of y, and F( ) first increases and then decreases with r changing with the same tendency. When 3. 6, both F( ) and G( ) become smaller enough to be neglected. The range within 0 < < 3. 6 is the hydrodynamic boundary region.
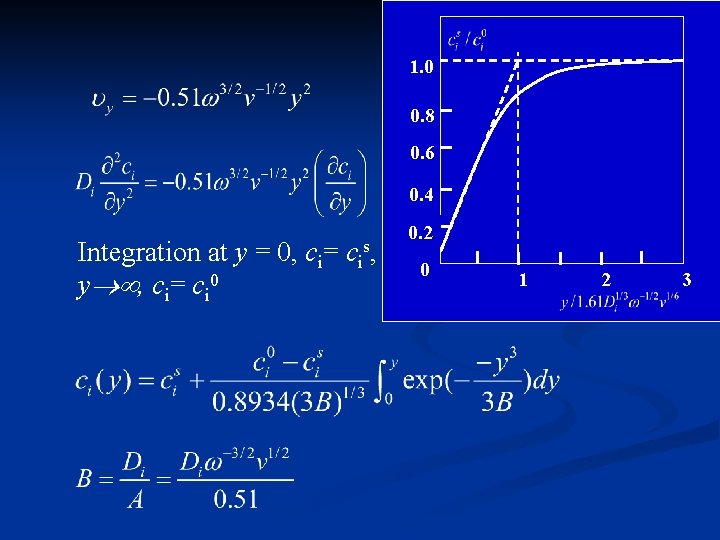
1. 0 0. 8 0. 6 0. 4 Integration at y = 0, ci= cis, y , ci= ci 0 0. 2 0 1 2 3
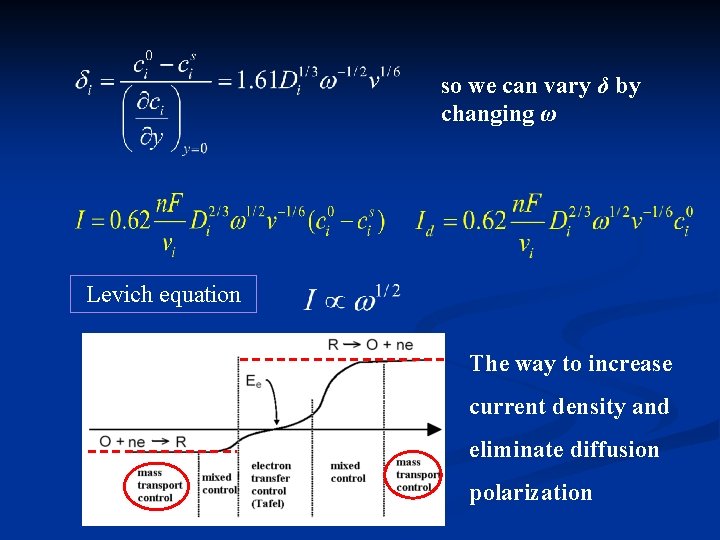
so we can vary δ by changing ω Levich equation The way to increase current density and eliminate diffusion polarization

Validity: 1) laminar fluid 2) diffusion control Valid when concentrated supporting electrolyte presents to eliminate electric migration. Application: 1) measurement of D: from the slope of Id~ 1/2 2) on knowing of D, measure n and c 0 3) determine whether or not diffusion is r. d. s.
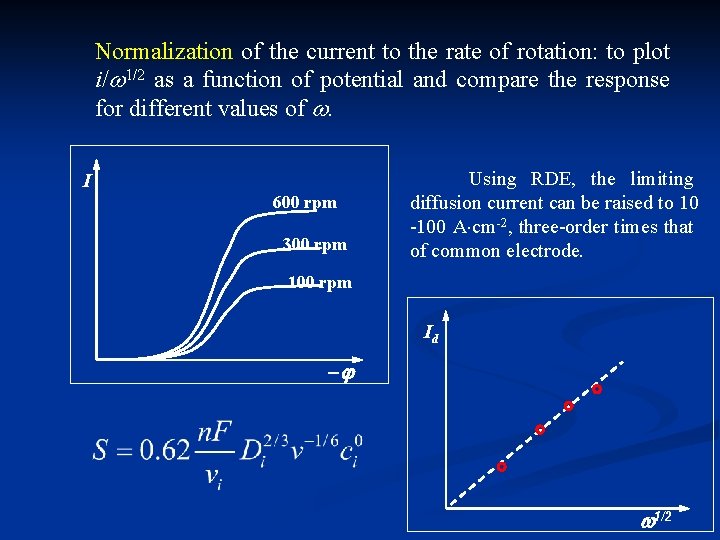
Normalization of the current to the rate of rotation: to plot i/ 1/2 as a function of potential and compare the response for different values of . I 600 rpm 300 rpm Using RDE, the limiting diffusion current can be raised to 10 -100 A cm-2, three-order times that of common electrode. 100 rpm Id 1/2
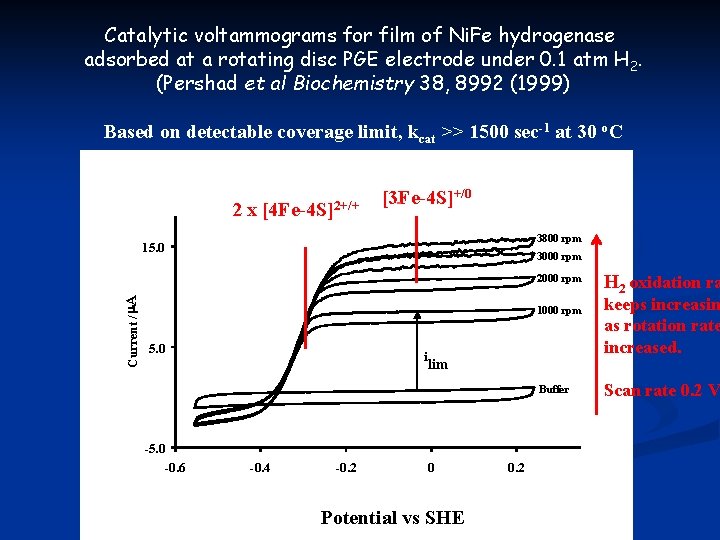
Catalytic voltammograms for film of Ni. Fe hydrogenase adsorbed at a rotating disc PGE electrode under 0. 1 atm H 2. (Pershad et al Biochemistry 38, 8992 (1999) Based on detectable coverage limit, kcat >> 1500 sec-1 at 30 o. C 2 x [4 Fe-4 S]2+/+ +/0 [3 Fe-4 S] 3800 rpm 15. 0 3000 rpm Current / A 2000 rpm 1000 rpm 5. 0 E (H+/H 2) i lim Buffer -5. 0 -0. 6 -0. 4 -0. 2 0 Potential vs SHE 0. 2 H 2 oxidation ra keeps increasin as rotation rate increased. Scan rate 0. 2 V
- Slides: 58