2 1 Classes of Hydrocarbons Hydrocarbons Aliphatic Aromatic

2. 1 Classes of Hydrocarbons
Hydrocarbons Aliphatic Aromatic
Hydrocarbons Aliphatic Alkanes Aromatic Alkenes Alkynes

Hydrocarbons Aliphatic Alkanes are hydrocarbons in which all of the bonds are single bonds. H H H C C H H H
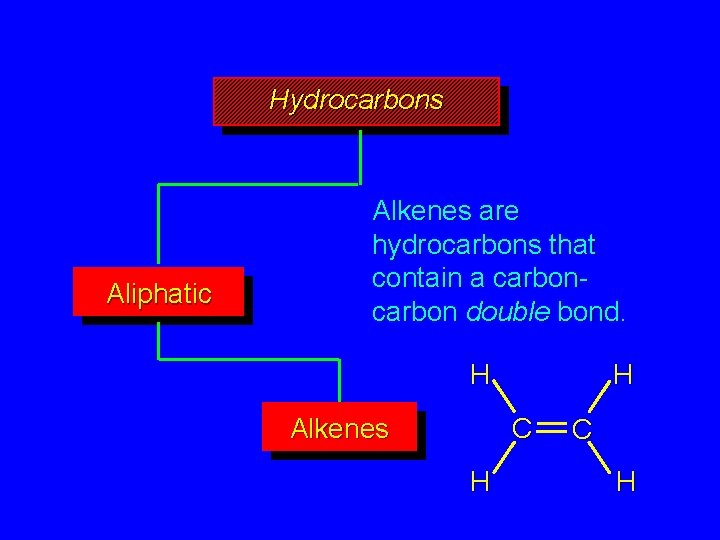
Hydrocarbons Aliphatic Alkenes are hydrocarbons that contain a carbon double bond. H H C Alkenes H C H
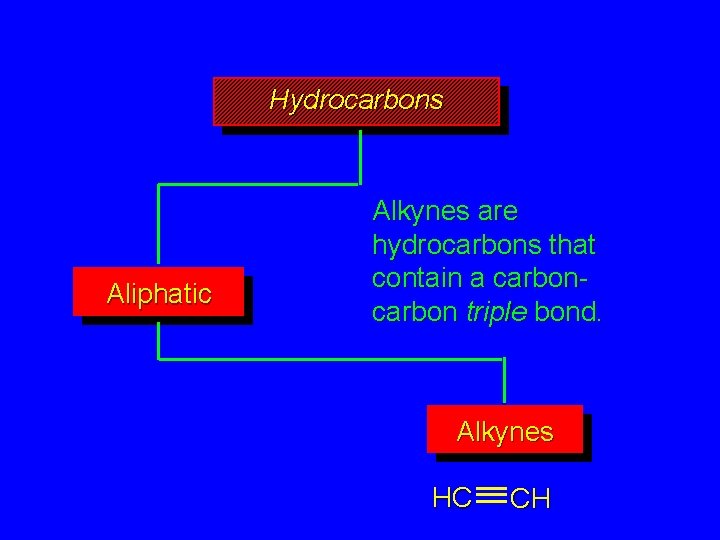
Hydrocarbons Aliphatic Alkynes are hydrocarbons that contain a carbon triple bond. Alkynes HC CH
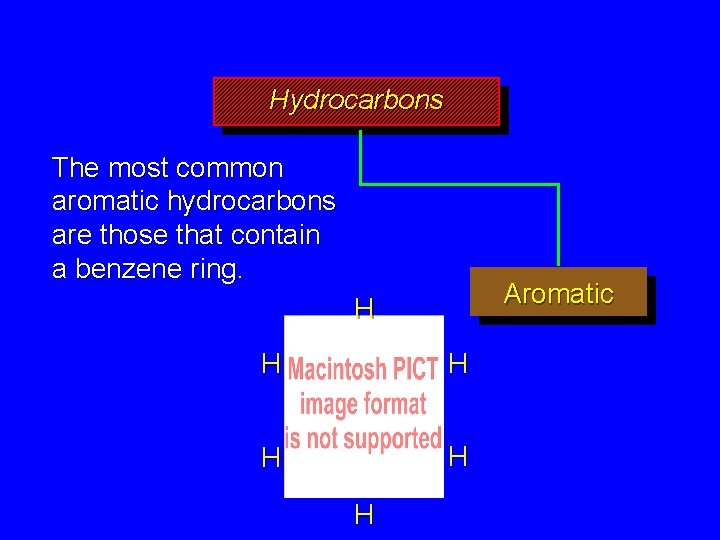
Hydrocarbons The most common aromatic hydrocarbons are those that contain a benzene ring. Aromatic H H H

2. 2 Reactive Sites in Hydrocarbons

Functional Group a structural unit in a molecule responsible for its characteristic behavior under a particular set of reaction conditions

Alkanes R—H R—X functional group is a hydrogen reaction that takes place is substitution one of the hydrogens is replaced by some other atom or group
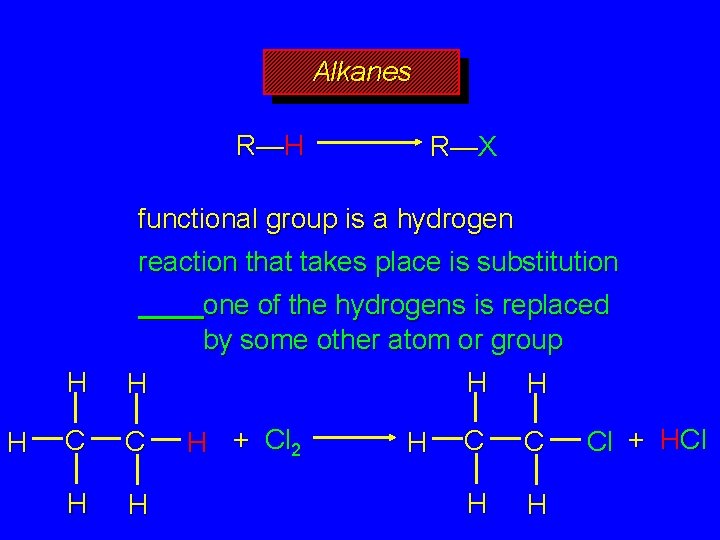
Alkanes R—H R—X functional group is a hydrogen reaction that takes place is substitution H H H C C H H one of the hydrogens is replaced by some other atom or group H H H + Cl 2 H C C H H Cl + HCl

Functional Groups in Hydrocarbons alkanes RH alkenes double bond alkynes triple bond arenes Ar. H

2. 3 The Key Functional Groups

Families of organic compounds and their functional groups Alcohols ROH Alkyl halides Amines RX (X = F, Cl, Br, I) primary amine: RNH 2 secondary amine: R 2 NH tertiary amine: R 3 N
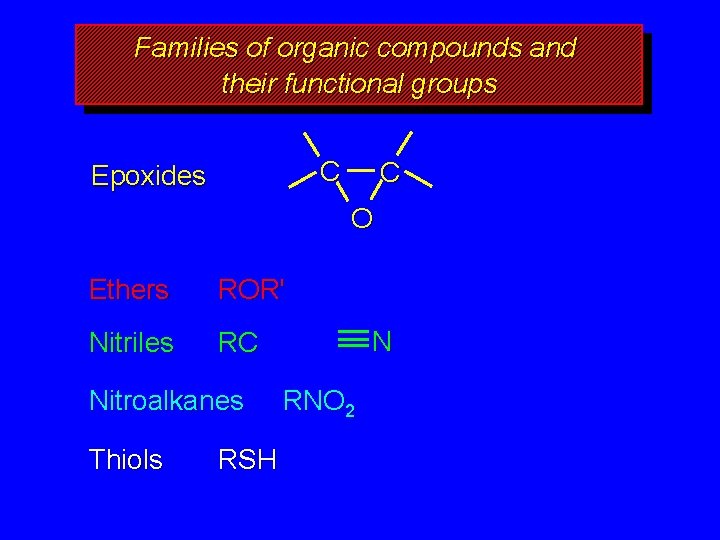
Families of organic compounds and their functional groups C Epoxides C O Ethers ROR' Nitriles RC Nitroalkanes Thiols RSH N RNO 2

Many classes of organic compounds contain a carbonyl group O O C C Carbonyl group R Acyl group
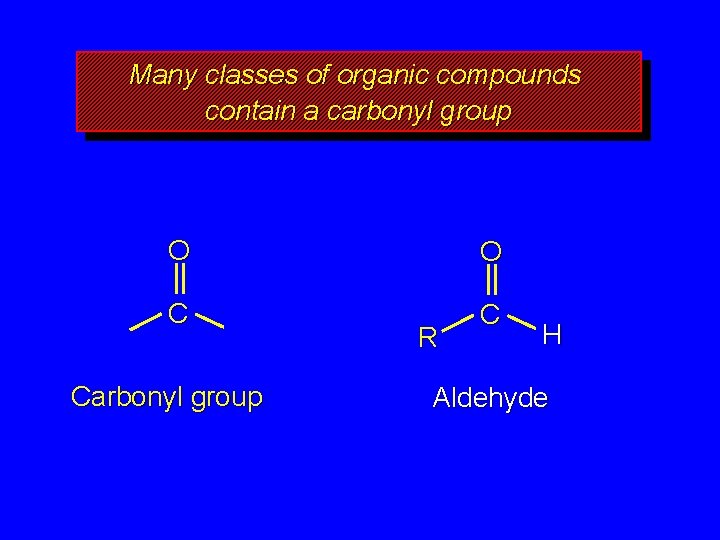
Many classes of organic compounds contain a carbonyl group O O C C Carbonyl group R H Aldehyde
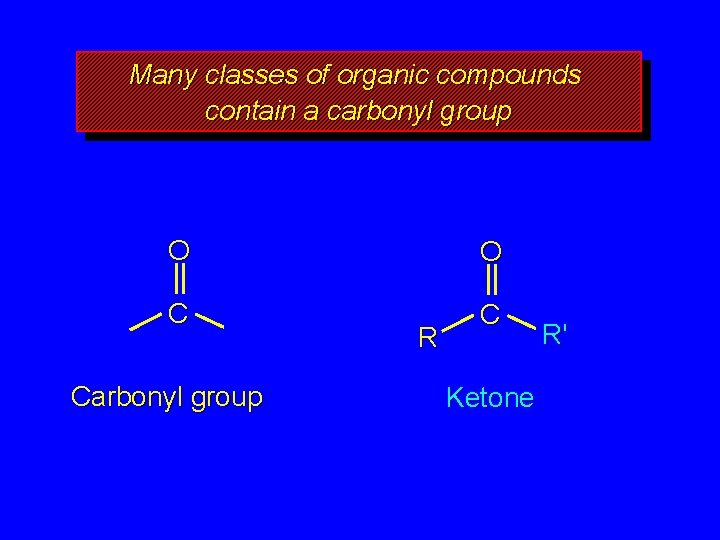
Many classes of organic compounds contain a carbonyl group O O C C Carbonyl group R Ketone R'
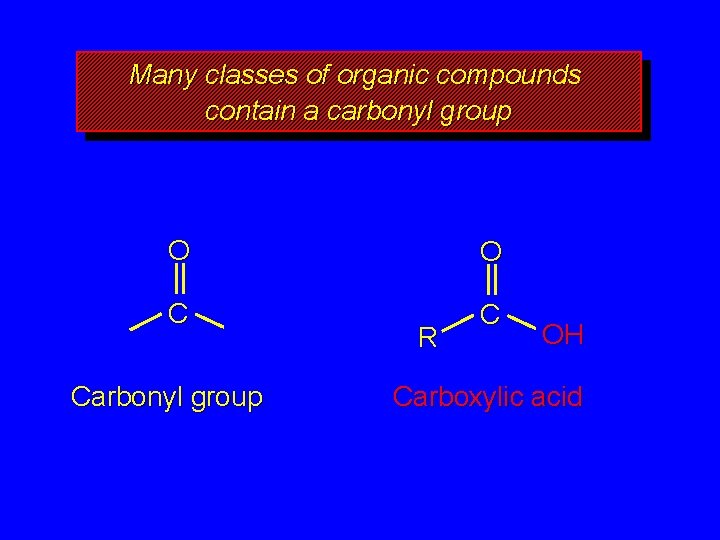
Many classes of organic compounds contain a carbonyl group O O C C Carbonyl group R OH Carboxylic acid
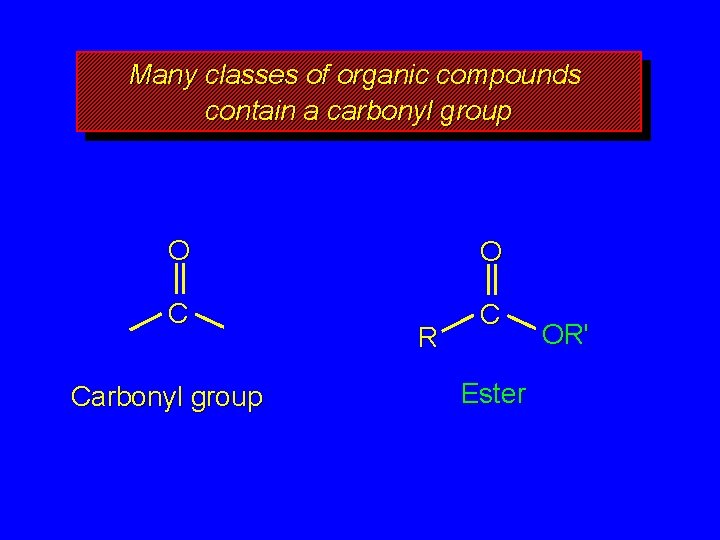
Many classes of organic compounds contain a carbonyl group O O C C Carbonyl group R Ester OR'
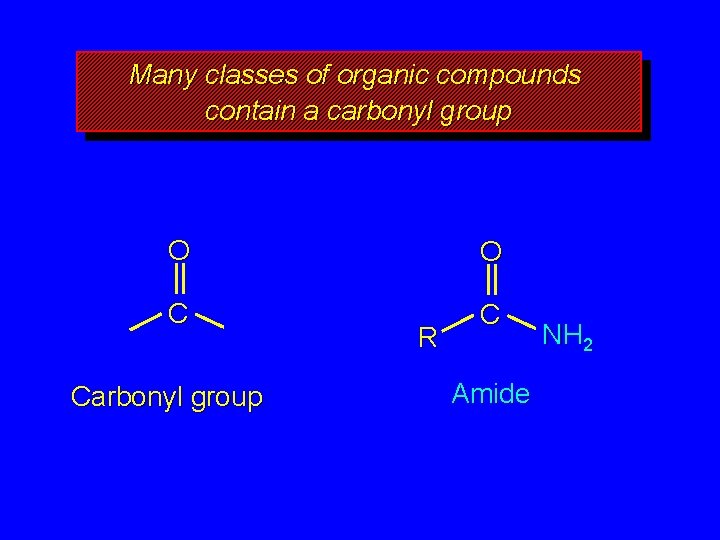
Many classes of organic compounds contain a carbonyl group O O C C Carbonyl group R Amide NH 2

Cn. H 2 n+2 2. 4 Introduction to Alkanes: Methane, Ethane, and Propane

The simplest alkanes Methane (CH 4) CH 4 Ethane (C 2 H 6) CH 3 Propane (C 3 H 8) CH 3 CH 2 CH 3 bp -160°C bp -89°C bp -42°C

C 4 H 10 2. 5 Isomeric Alkanes: The Butanes

n-Butane CH 3 CH 2 CH 3 Isobutane (CH 3)3 CH bp -0. 4°C bp -10. 2°C

2. 6 Higher n-Alkanes
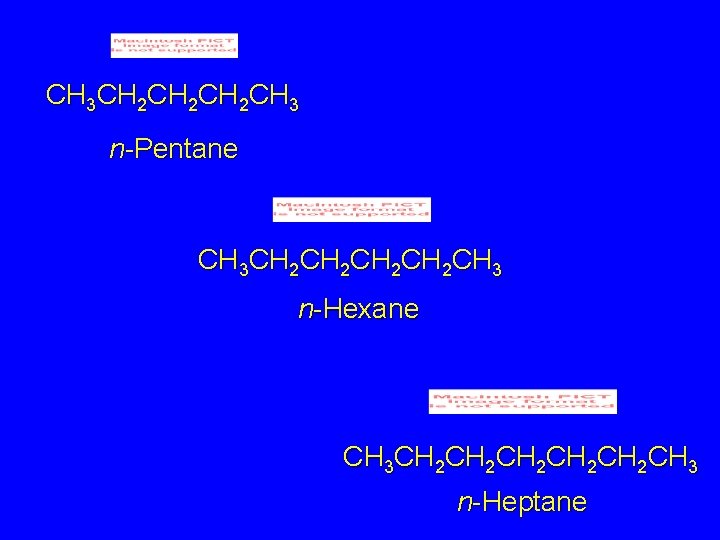
CH 3 CH 2 CH 2 CH 3 n-Pentane CH 3 CH 2 CH 2 CH 3 n-Hexane CH 3 CH 2 CH 2 CH 2 CH 3 n-Heptane

2. 7 The C 5 H 12 Isomers
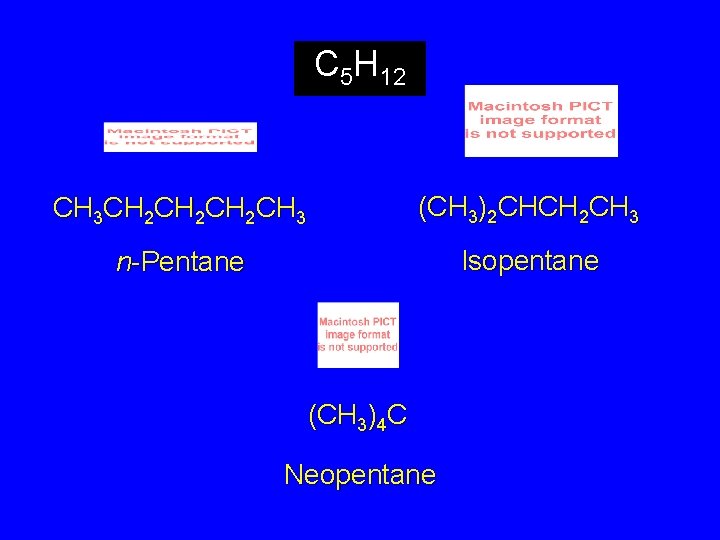
C 5 H 12 CH 3 CH 2 CH 2 CH 3 (CH 3)2 CHCH 2 CH 3 n-Pentane Isopentane (CH 3)4 C Neopentane

How many isomers? The number of isomeric alkanes increases as the number of carbons increase. There is no simple way to predict how many isomers there are for a particular molecular formula.
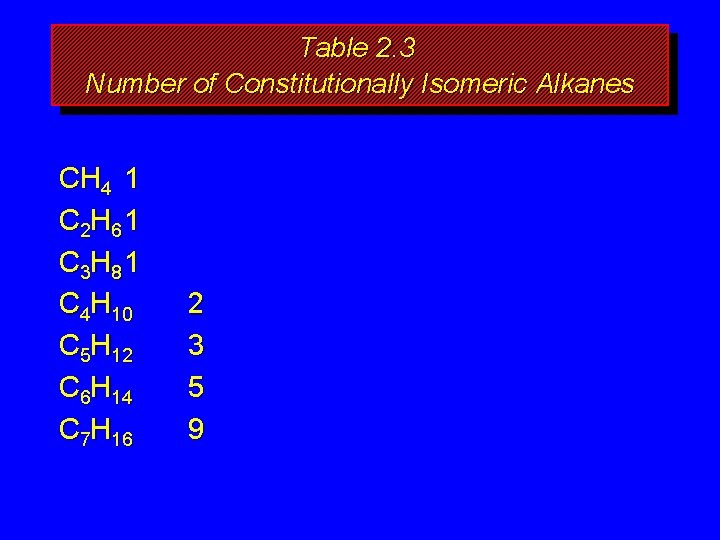
Table 2. 3 Number of Constitutionally Isomeric Alkanes CH 4 1 C 2 H 6 1 C 3 H 8 1 C 4 H 10 C 5 H 12 C 6 H 14 C 7 H 16 2 3 5 9
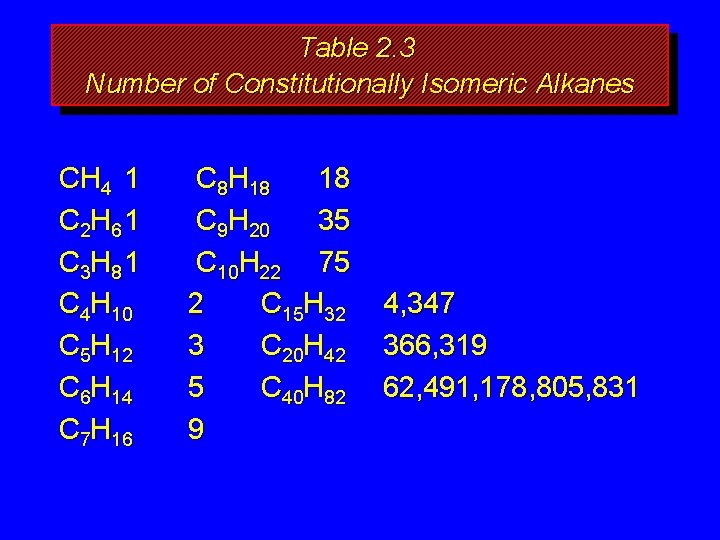
Table 2. 3 Number of Constitutionally Isomeric Alkanes CH 4 1 C 2 H 6 1 C 3 H 8 1 C 4 H 10 C 5 H 12 C 6 H 14 C 7 H 16 C 8 H 18 18 C 9 H 20 35 C 10 H 22 75 2 C 15 H 32 3 C 20 H 42 5 C 40 H 82 9 4, 347 366, 319 62, 491, 178, 805, 831
- Slides: 32