2 1 Basic Chemistry Matter is anything that

2. 1 Basic Chemistry • Matter is anything that takes up space and has mass. – The three states of matter are solid, liquid, and gas. • All matter, living or nonliving, is made up of elements. – Elements are substances that cannot be broken down into simpler substances by ordinary chemical means. 1
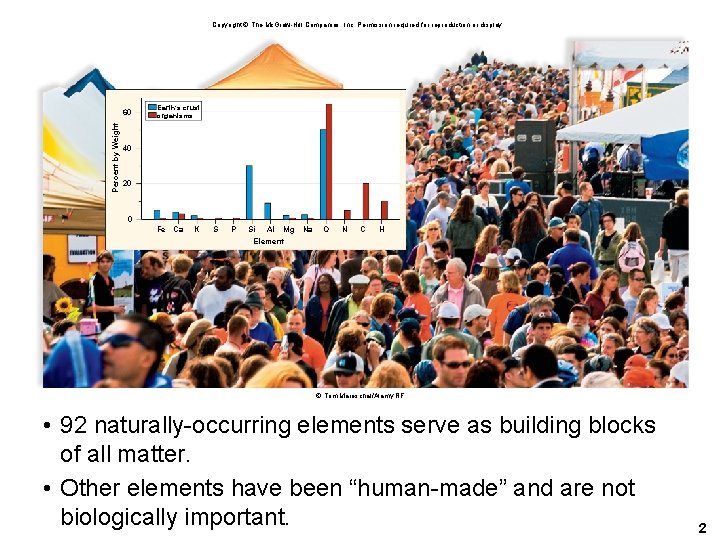
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Percent by Weight 60 Earth’s crust organisms 40 20 0 Fe Ca K S P Si Al Mg Na Element O N C H © Tom Mareschal/Alamy RF • 92 naturally-occurring elements serve as building blocks of all matter. • Other elements have been “human-made” and are not biologically important. 2

Elements That Make up 95% of Organisms (by weight) –C –H –N –O –P –S Carbon Hydrogen Nitrogen Oxygen Phosphorus Sulfur 3
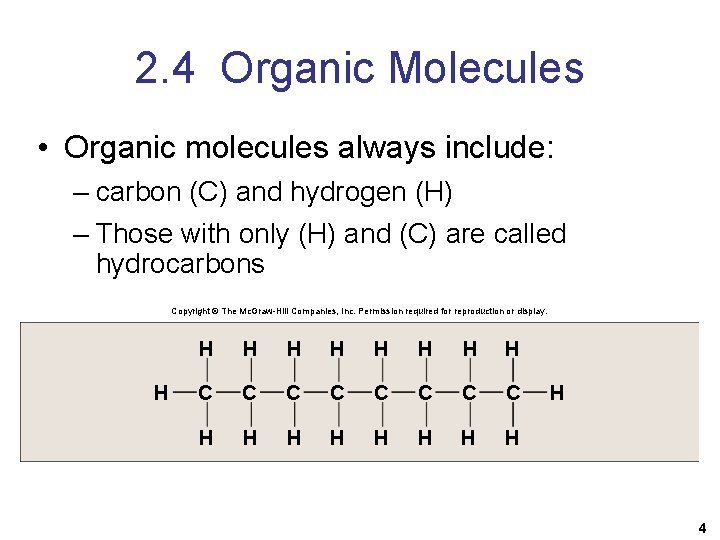
2. 4 Organic Molecules • Organic molecules always include: – carbon (C) and hydrogen (H) – Those with only (H) and (C) are called hydrocarbons Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H H H H H C C C C H H H H H 4

2. 4 Organic Molecules • The chemistry of carbon accounts for the formation of great variety of organic molecules. • Carbon atoms contain four valence electrons. • A carbon atom may share electrons with another carbon atom or other atoms in order to achieve eight electrons. • Satisfying the octet rule 5

2. 4 Organic Molecules • Functional groups are a specific combination of bonded atoms that always react in the same way. • The more common functional groups are listed in Table 2. 1. 6
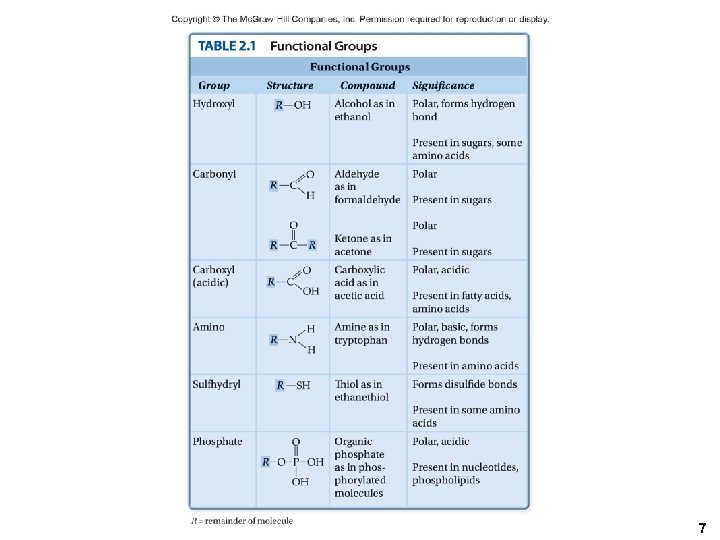
7

2. 4 Organic Molecules • Macromolecules contain many molecules joined together. – Monomers: Simple organic molecules that exist individually – Polymers: Large organic molecules form by combining monomers 8

2. 4 Organic Molecules • Polymers in cells and their monomers Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Polymer Monomer carbohydrate (e. g. , starch) protein nucleic acid monosaccharide amino acid nucleotide 9

2. 4 Organic Molecules • Cells use common reactions to join monomers. – In a dehydration reaction an -OH and -H are removed as a water molecule. – In a hydrolysis reaction, components of water are added. 10
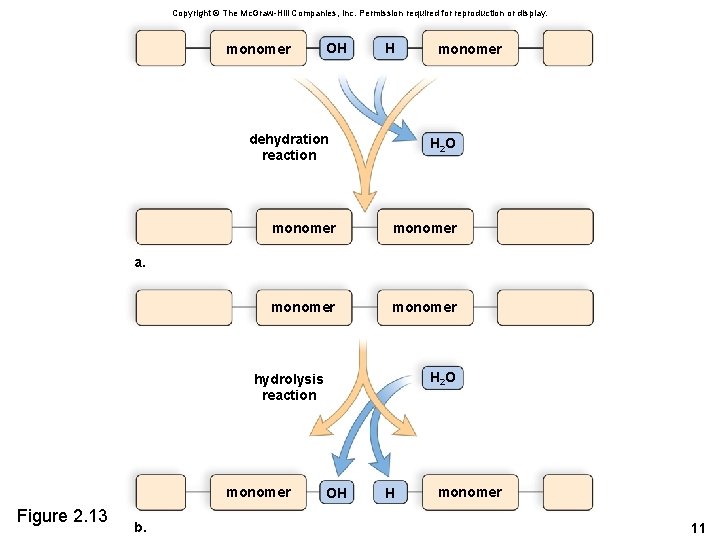
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. monomer OH H dehydration reaction monomer H 2 O monomer a. H 2 O hydrolysis reaction monomer Figure 2. 13 b. OH H monomer 11

2. 5 Carbohydrates • Carbohydrates function for quick fuel and short-term energy storage in organisms. – Play a structural role in woody plants, bacteria and insects – On cell surfaces, involved in cell-to-cell recognition 12
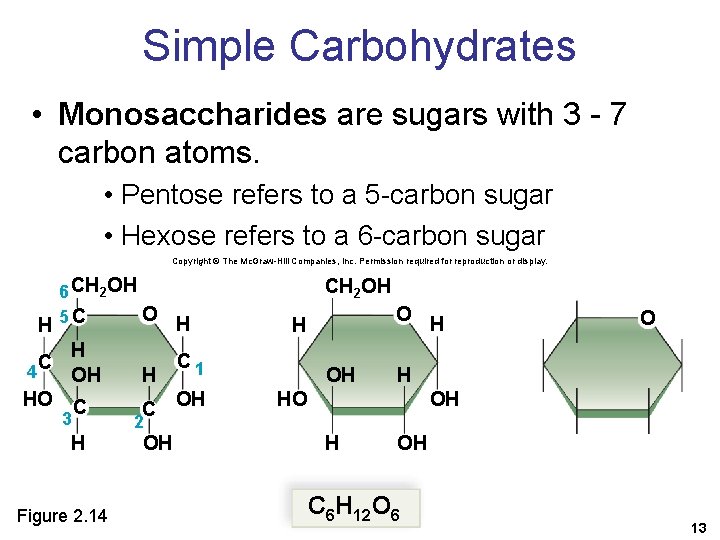
Simple Carbohydrates • Monosaccharides are sugars with 3 - 7 carbon atoms. • Pentose refers to a 5 -carbon sugar • Hexose refers to a 6 -carbon sugar Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H 6 CH 2 OH O 5 C CH 2 OH H C 4 H OH H HO C C OH 3 H Figure 2. 14 O H C 1 OH HO 2 OH H OH C 6 H 12 O 6 13
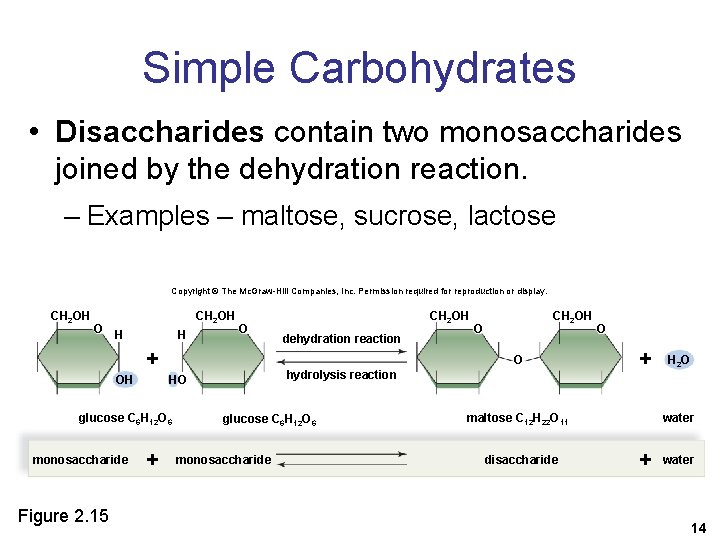
Simple Carbohydrates • Disaccharides contain two monosaccharides joined by the dehydration reaction. – Examples – maltose, sucrose, lactose Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. CH 2 OH O H H O + OH Figure 2. 15 O hydrolysis reaction HO + dehydration reaction CH 2 OH O glucose C 6 H 12 O 6 monosaccharide CH 2 OH glucose C 6 H 12 O 6 monosaccharide O + maltose C 12 H 22 O 11 disaccharide H 2 O water + water 14

Polysaccharides • Polysaccharides such as starch, glycogen, and cellulose are long polymers that contain many glucose subunits. 15

Starch and Glycogen • Starch is the storage form of glucose in plants. – May contain up to 4, 000 glucose units – Fewer side branches than glycogen • Glycogen is the storage form of glucose in animals. – Liver stores glucose as glycogen – In between meals, the liver releases glucose stored in glycogen 16
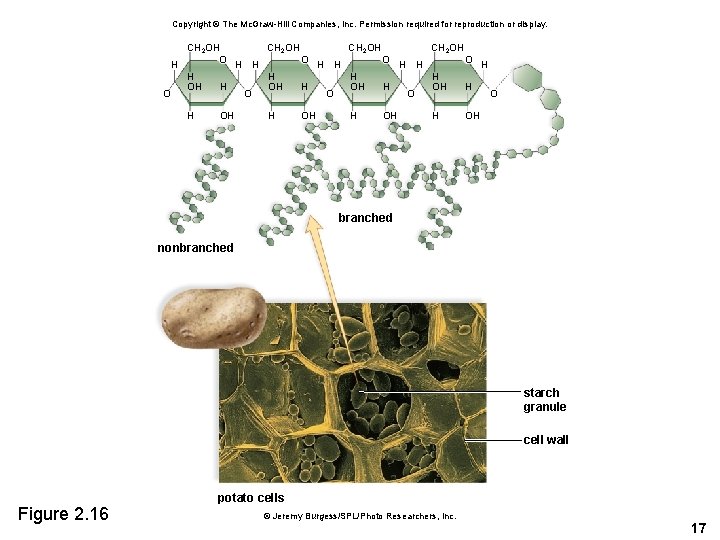
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. CH 2 OH O O H H H OH H O O O H OH branched nonbranched starch granule cell wall Figure 2. 16 potato cells © Jeremy Burgess/SPL/Photo Researchers, Inc. 17
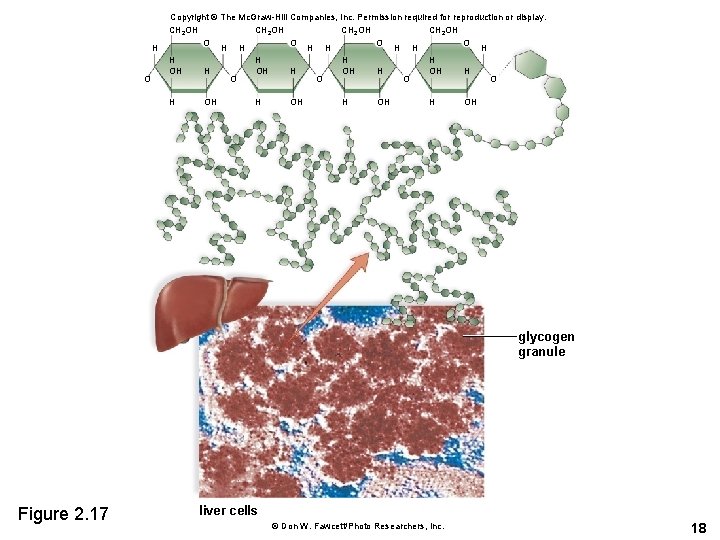
H O Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. CH 2 OH O O H H H OH H O O H OH glycogen granule Figure 2. 17 liver cells © Don W. Fawcett/Photo Researchers, Inc. 18

Cellulose • Some polysaccharides function as structural components of cells. • Cellulose is found in the cell walls of plants. – Accounts for the strong nature of the cell walls – Has different chemical linkage than starch or glycogen • Prevents us from digesting foods with cellulose • Chiton, found in the exoskeleton of crabs, is another structural polysaccharide. 19
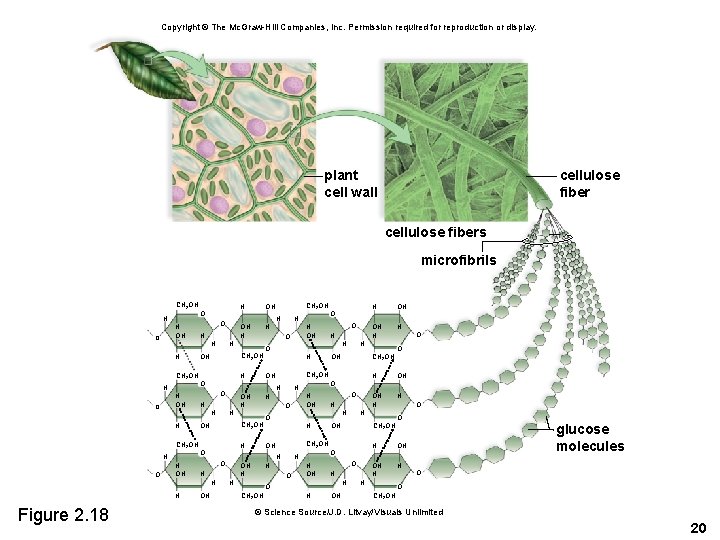
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. plant cell wall cellulose fibers microfibrils CH 2 OH H H OH O H CH 2 OH H H OH OH H H O CH 2 OH H O H H OH O H H H O O H OH OH H H CH 2 OH OH O CH 2 OH H H OH H O CH 2 OH OH OH CH 2 OH H H OH O CH 2 OH OH OH H OH OH H O O H H H OH O CH 2 OH O H H H OH H H Figure 2. 18 O H CH 2 OH OH H H O OH O O H OH OH H glucose molecules O O CH 2 OH © Science Source/J. D. Litvay/Visuals Unlimited 20

2. 6 Lipids • Lipids contain more energy per gram than other biological molecules. • Types – Fats and oils used for energy storage – Phospholipds from membranes – Steroids include sex hormones 21

2. 6 Lipids • Lipids are diverse in structure and function. • Lipids have one common characteristic – they do not dissolve in water (hydrophobic). 22

Fats and Oils • Fats – Usually of animal origin – Solid at room temperature – Store energy, insulate against heat loss, form protective cushion • Oils – Usually of plant origin – Liquid at room temperature 23

Fats and Oils • A fat molecule is also known as a triglyceride or neutral fat. • A triglyceride consists of – One glycerol backbone – Three fatty acids 24
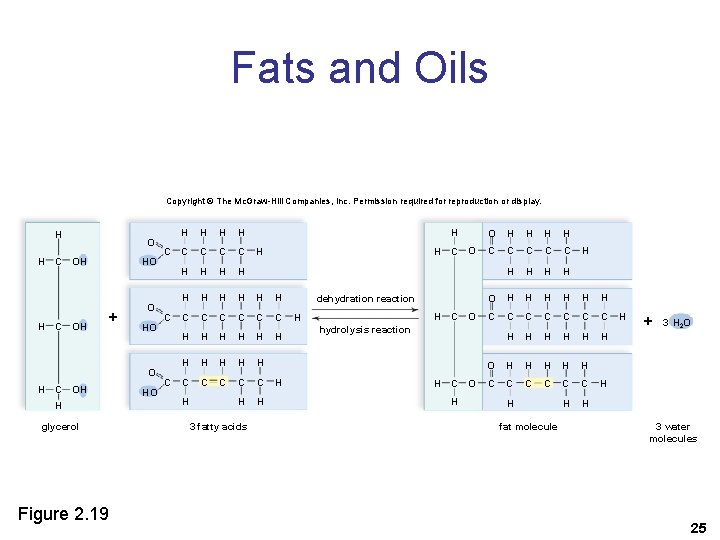
Fats and Oils Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H H H O C OH HO + O HO O H C OH H glycerol Figure 2. 19 HO C C C H H H H H C C C H H H 3 fatty acids H H C O dehydration reaction H H C O O H H C C C H H O H H H C C C C H H H O H H H C C C H H hydrolysis reaction H C H O H fat molecule H H + 3 H 2 O H 3 water molecules 25

• Emulsification – Fat droplets disperse in water. – Emulsifiers contain molecules with a polar and nonpolar end. 26

Saturated, Unsaturated, and Trans-Fatty Acids • A fatty acid is a hydrocarbon chain that ends with the acidic group —COOH. • Saturated fatty acids have no double bonds between carbon atoms. • Unsaturated fatty acids have one or more double bonds between carbon atoms. 27
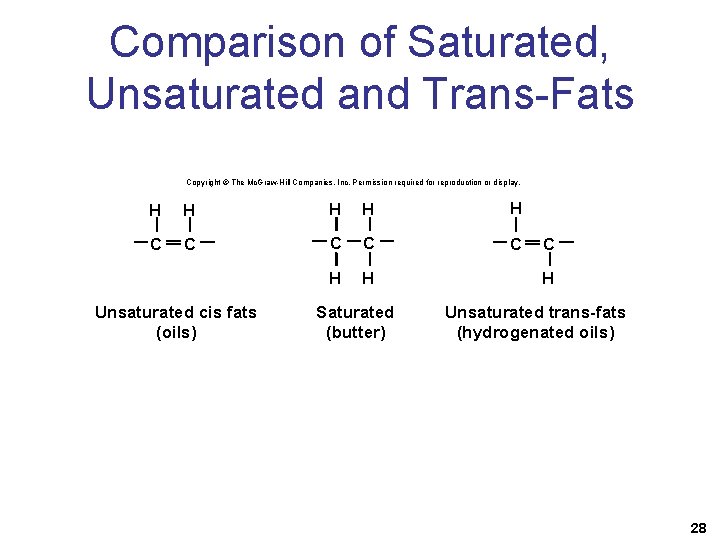
Comparison of Saturated, Unsaturated and Trans-Fats Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H H H C C C H H Unsaturated cis fats (oils) Saturated (butter) C H Unsaturated trans-fats (hydrogenated oils) 28

Phospholipids • Phospholipids are comprised of two fatty acids and a phosphate group • The phosphate group is polar so the molecules are not electrically neutral. • The phosphate group forms a polar head (hydrophilic) while the rest of the molecule is a nonpolar (hydrophobic) tail. 29
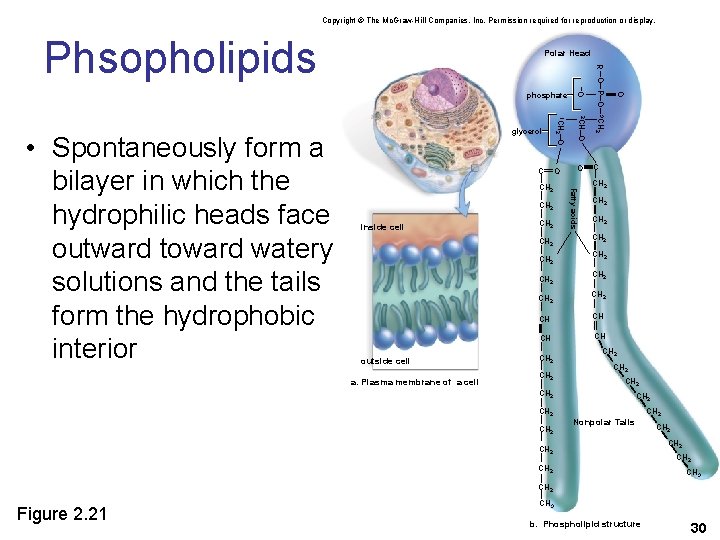
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Phsopholipids Polar Head CH 2 outside cell a. Plasma membrane of a cell C CH 2 CH 2 CH CH CH 2 O R – O– P– O – 3 CH 2 O Fatty acids inside cell –O CH 2 2 CH CH 2 –O C 1 CH 2 • Spontaneously form a bilayer in which the hydrophilic heads face outward toward watery solutions and the tails form the hydrophobic interior glycerol –O phosphate CH 2 CH 2 Nonpolar Tails CH 2 CH 2 CH 3 CH 2 Figure 2. 21 CH 3 b. Phospholipid structure 30
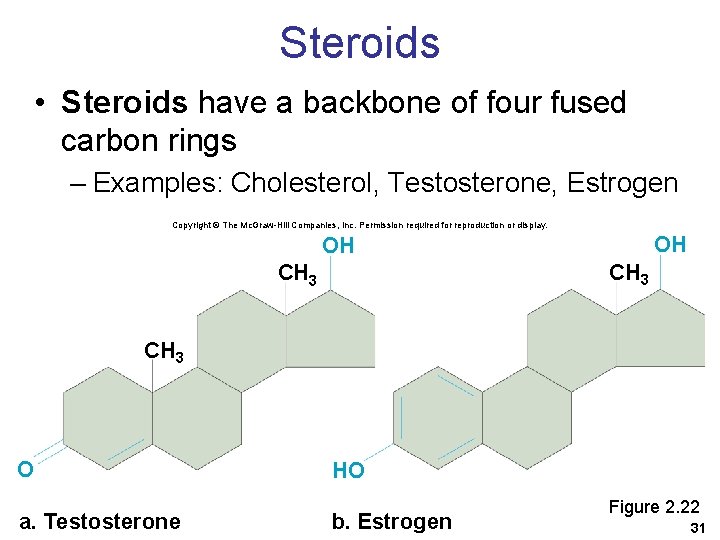
Steroids • Steroids have a backbone of four fused carbon rings – Examples: Cholesterol, Testosterone, Estrogen Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. OH OH CH 3 O a. Testosterone HO b. Estrogen Figure 2. 22 31
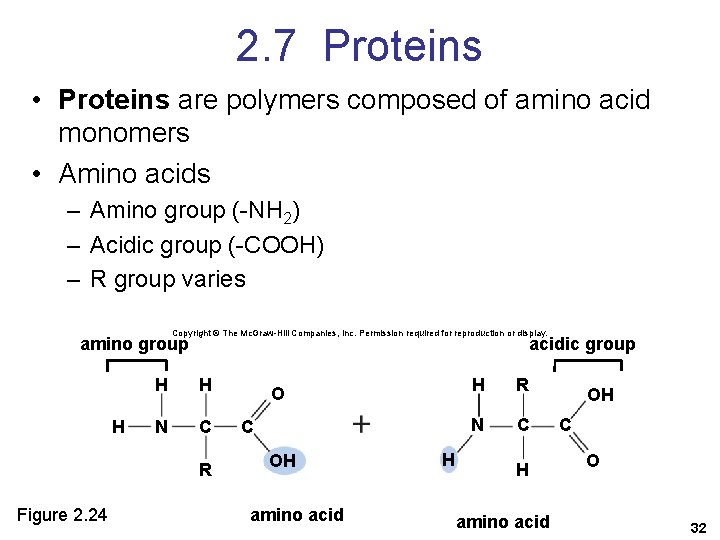
2. 7 Proteins • Proteins are polymers composed of amino acid monomers • Amino acids – Amino group (-NH 2) – Acidic group (-COOH) – R group varies Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. amino group H acidic group H H N C R Figure 2. 24 O C OH amino acid H H R N C H amino acid OH C O 32
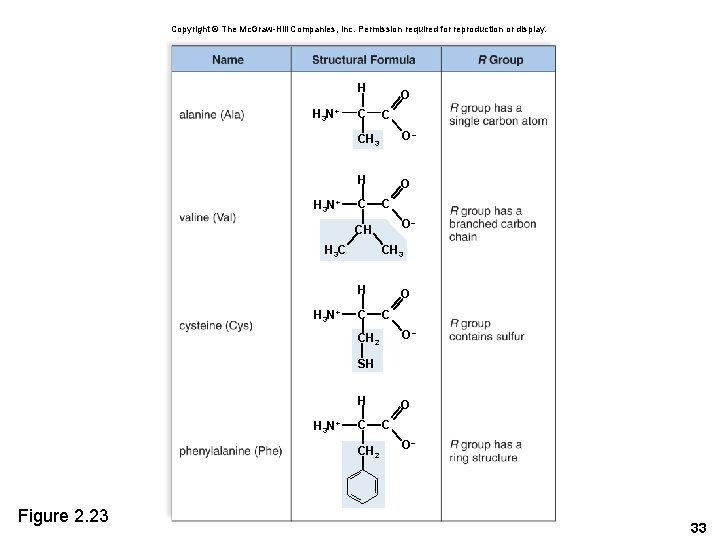
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H H 3 N+ C O C CH 3 O– H O C C O– CH CH 3 H 3 C H H 3 N+ C O– CH 2 SH H H 3 N+ C CH 2 Figure 2. 23 O C O– 33

2. 7 Proteins • Proteins perform many functions – Structural proteins give support (keratin, collagen) – Enzymes speed up chemical reactions – Hormones are chemical messengers – Actin and myosin move cells and muscles – Some proteins transport molecules in blood – Antibodies protect cells – Channels allow substances to cross membranes 34
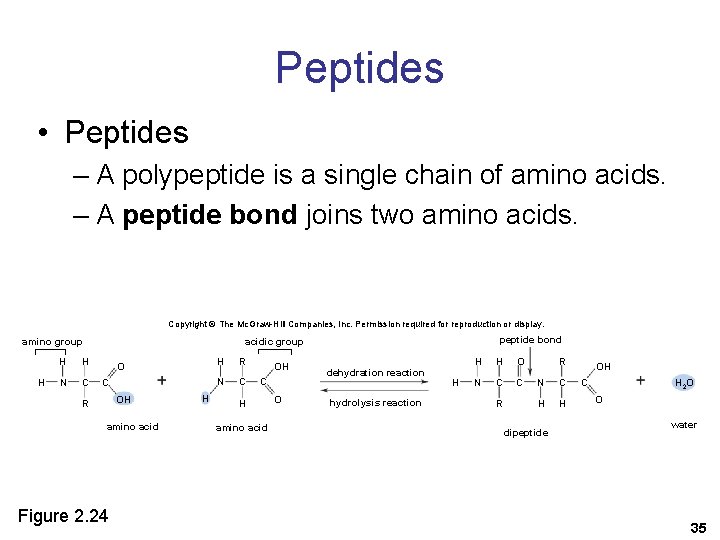
Peptides • Peptides – A polypeptide is a single chain of amino acids. – A peptide bond joins two amino acids. Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. amino group H H H N C peptide bond acidic group O C OH R amino acid Figure 2. 24 H H R N C OH C H amino acid O dehydration reaction hydrolysis reaction H H H O N C C R R N C H H dipeptide OH C H 2 O O water 35
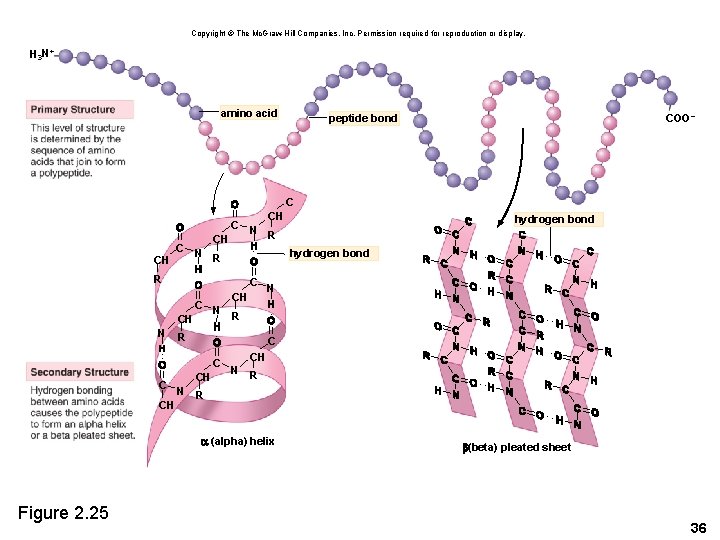
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H 3 N+ amino acid COO– peptide bond C CH C N R CH hydrogen bond R C CH N R R C C CH C N R CH CH N R (alpha) helix Figure 2. 25 hydrogen bond (beta) pleated sheet 36
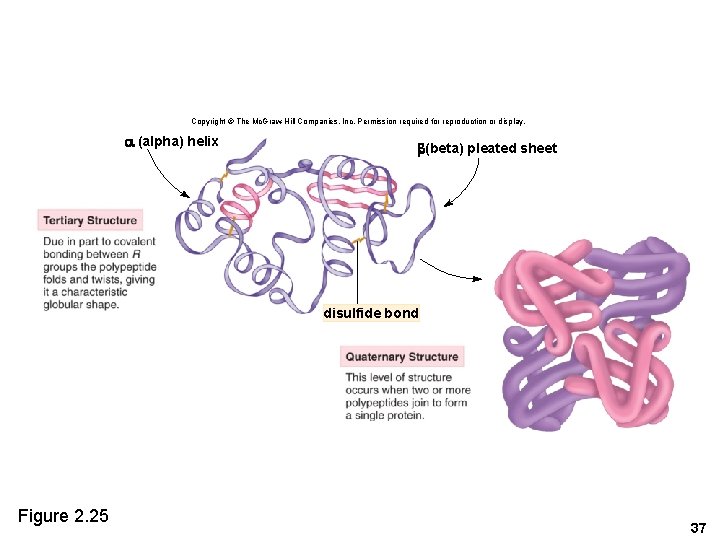
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. (alpha) helix (beta) pleated sheet disulfide bond Figure 2. 25 37
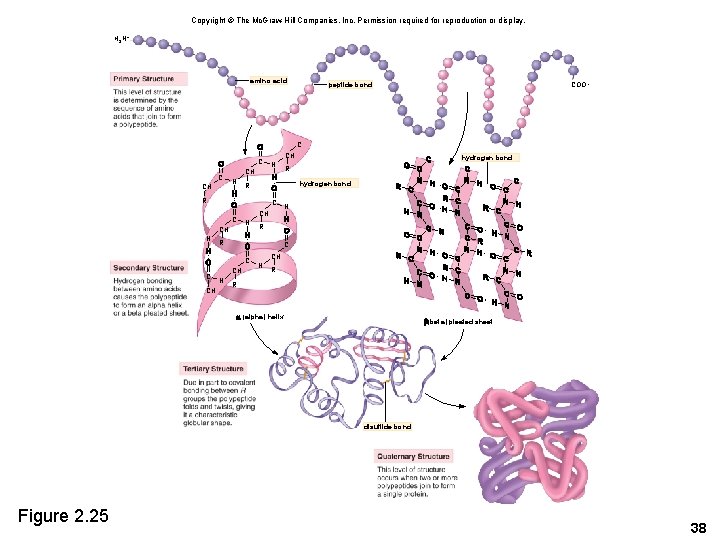
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H 3 N+ amino acid COO– peptide bond C CH CH N C C CH N R R CH N hydrogen bond R hydrogen bond C C C N R R N CH CH N R R (alpha) helix (beta) pleated sheet disulfide bond Figure 2. 25 38

2. 8 Nucleic Acids • The two types of nucleic acids are – DNA (deoxyribonucleic acid) • Stores genetic information in the cell and in the organism • DNA replicates to transmit its information when a cell divides or organism reproduces – RNA (ribonucleic acid) 39

Structure of DNA and RNA • Both DNA and RNA are polymers of nucleotides – Every nucleotide is a molecular complex of • Phosphate • Pentose sugar (ribose or deoxyribose) • Nitrogen-containing base – DNA contains: Adenine (A), Thymine (T), Guanine (G) and Cytosine (C) – In RNA, uracil (U) replaces thymine 40
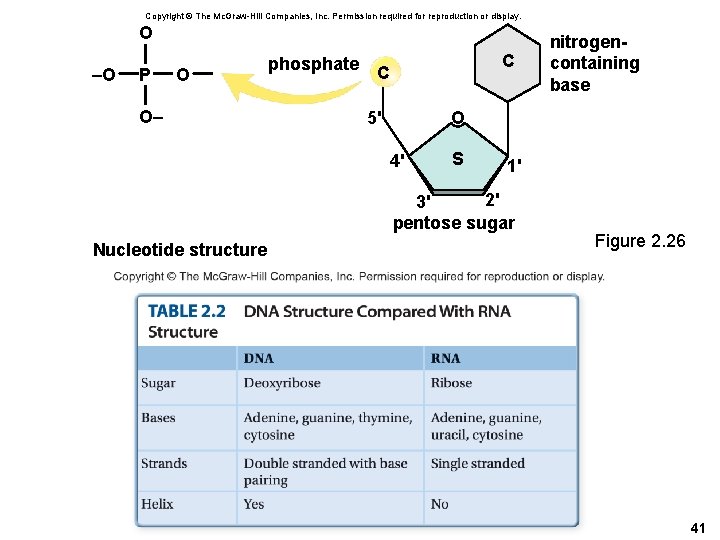
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. O –O P O O– phosphate C C O 5' 4' S 1' 2' 3' pentose sugar Nucleotide structure nitrogencontaining base Figure 2. 26 41

Structure of DNA and RNA • The nucleotides form a linear molecule called a strand. • DNA is a double helix of two strands. • The two strands are held together by hydrogen bonds. • Rungs of the ladder are formed by complementary paired bases. – Adenine (A) always pairs with thymine (T) – Cytosine (C) always pairs with guanine (G) 42
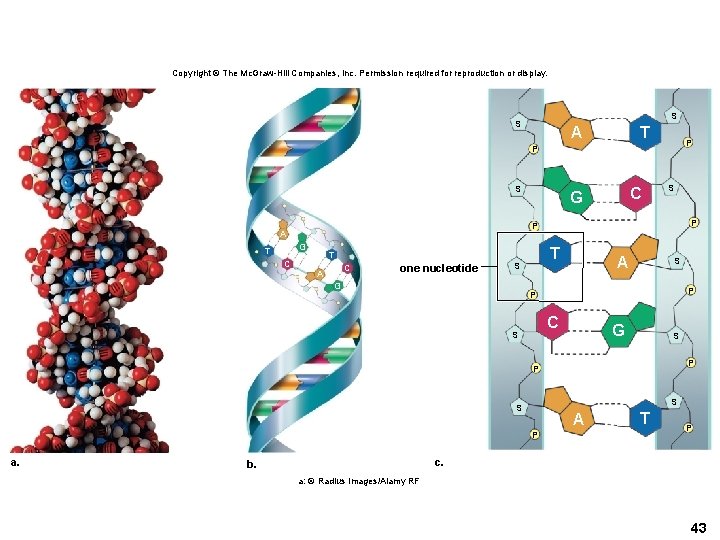
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. S S A T P P S G C S P P A T C G T T C A one nucleotide S G A S P P C S G S P P S S A P a. T P c. b. a: © Radius Images/Alamy RF 43

Structure of DNA and RNA • RNA is single-stranded. – Several types are involved in carrying information from DNA to make proteins. • ATP (Adenosine Triphosphate) – ATP is a high-energy molecule. – ATP undergoes hydrolysis and energy is released. – ATP is the energy “currency” of the cell. 44

Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H 2 O P P P + energy • Last two phosphate bonds are unstable and easily broken. • Hydrolization forms ADP (adenosine diphosphate). • ATP can be rebuilt. – Add P to ADP to make ATP Figure 2. 28 45
- Slides: 45