1976 U S Standard Atmosphere One of several

![For Incompressible Fluid (like Water) P [Pascal = N/m 2] P 1 The difference For Incompressible Fluid (like Water) P [Pascal = N/m 2] P 1 The difference](https://slidetodoc.com/presentation_image_h/223f012d8206dcfd5cc2bd095d2cc967/image-2.jpg)



- Slides: 8

1976 U. S. Standard Atmosphere • One of several models of the atmosphere. • Provides dependence of temperature, pressure and density on altitude. • Temperature is tabulated at 8 specific altitudes and temperatures at other altitudes, pressure and density computed from those data. 1976 US Standard Atmosphere 1
![For Incompressible Fluid like Water P Pascal Nm 2 P 1 The difference For Incompressible Fluid (like Water) P [Pascal = N/m 2] P 1 The difference](https://slidetodoc.com/presentation_image_h/223f012d8206dcfd5cc2bd095d2cc967/image-2.jpg)
For Incompressible Fluid (like Water) P [Pascal = N/m 2] P 1 The difference in pressure at the two depth is due to the weight of the column of water between them. z Known as “hydrostatic equation” 1976 US Standard Atmosphere 2

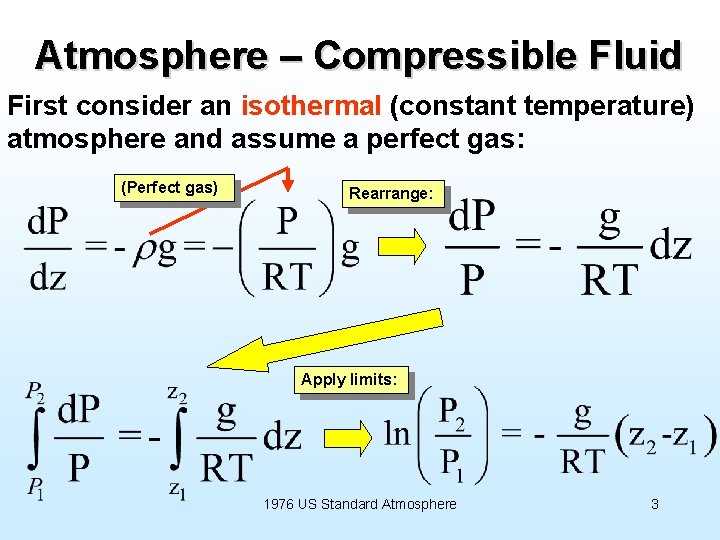
Atmosphere – Compressible Fluid First consider an isothermal (constant temperature) atmosphere and assume a perfect gas: (Perfect gas) Rearrange: Apply limits: 1976 US Standard Atmosphere 3

Atmosphere – Compressible Fluid So for isothermal (constant temperature) atmosphere: Note exponential drop of pressure with altitude, vs. linear with depth in an incompressible fluid like water. i. e. , expressure at one point in terms of that at a different altitude You can also handle (integrate) the case where the temperature is a linear function of altitude (constant “lapse rate”). * These are the two options in the VBA coding that goes with the USStand. Atmos. xls spreadsheet on webpage (www. people. virginia. edu/~rjr/modules/xls) 4 *See, e. g. , D. G. Shepherd, Elements of Fluid Mechanics, Harcourt, Brace and World, 1965.

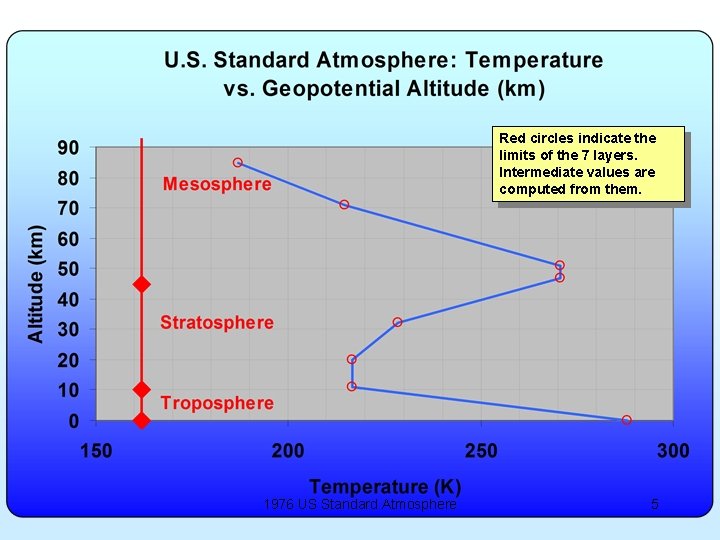
Red circles indicate the limits of the 7 layers. Intermediate values are computed from them. 1976 US Standard Atmosphere 5

U. S. Standard Atmosphere 1976 • Seven layers between sea level and ~85 km • Decrease of “g” with altitude is included in the calculation by use of “geopotential” altitude. – h = Altitude * Rearth / (Altitude + Rearth) • Two layers are isothermal • Five layers have constant “lapse rate” (d. T/dz) • Pressure tabulated at base of each layer and change computed between there and the point of interest. – Use equation we just derived for isothermal layer, similar equation for constant lapse rate layers. • Density found from P and T and perfect gas law. 1976 US Standard Atmosphere 6

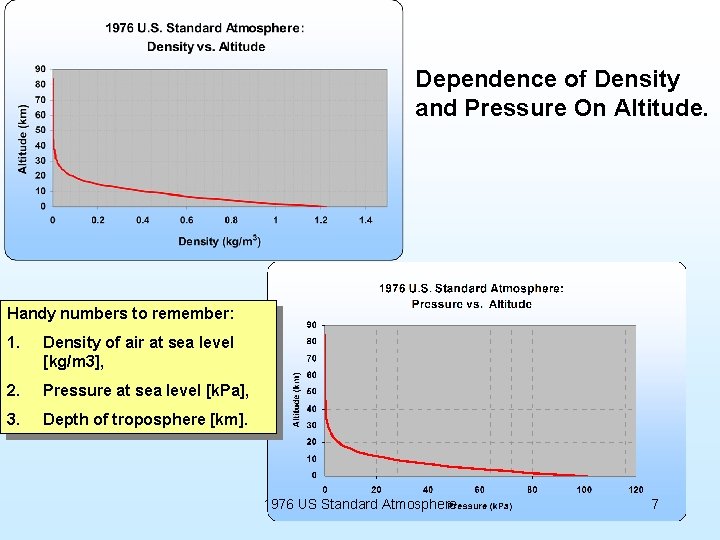
Dependence of Density and Pressure On Altitude. Handy numbers to remember: 1. Density of air at sea level [kg/m 3], 2. Pressure at sea level [k. Pa], 3. Depth of troposphere [km]. 1976 US Standard Atmosphere 7

Other notes: • Most sunlight comes through atmosphere (it’s largely transparent), hits surface of earth and the air in contact is heated up. That’s why the peak air temperature is at surface. • The other temperature peak at ~50 km is due to absorption of UV rays by ozone (and there isn’t much atmosphere to heat up). 1976 US Standard Atmosphere 8