1953 Watson Crick DNA double helix 1970 E


基因 程技術發展 1953 年 Watson 與 Crick 提出 DNA 雙股螺旋結構 模型 (double helix) 1970 年首先由大腸桿菌 (E. coli) 中分離出第二型限 制酶 (type II restriction endonuclease) Eco. RI 1972 年 Berg 在試管中完成第一個重組 DNA 1973 年 Boyer 與 Cohen 建立利用質體選殖 DNA 的技術 1976 年 Sanger 與 Gilbert 建立 DNA 定序技術 2
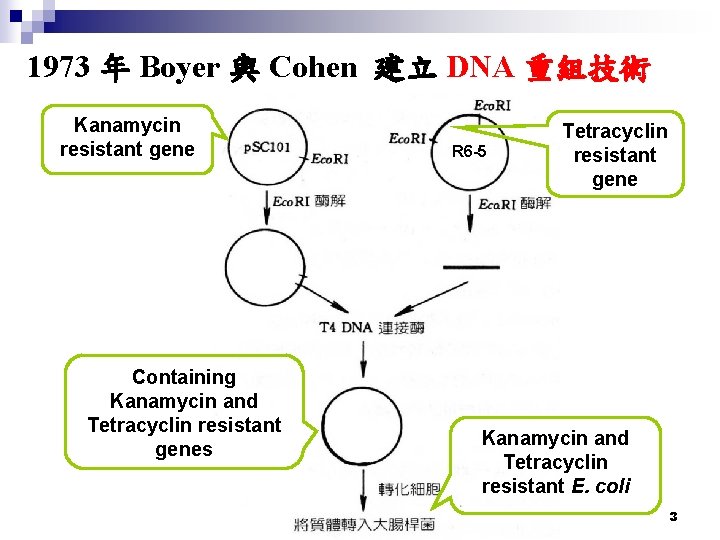
1973 年 Boyer 與 Cohen 建立 DNA 重組技術 Kanamycin resistant gene Containing Kanamycin and Tetracyclin resistant genes R 6 -5 Tetracyclin resistant gene Kanamycin and Tetracyclin resistant E. coli 3



限制酶 (restriction endonuclease) 的發現-1970 年 Eco. RI 1978 Nobel prize 1970 年首先由大腸桿菌 (E. coli) 中分離出第二 型限制酶 (type II restriction endonuclease) Eco. RI 6
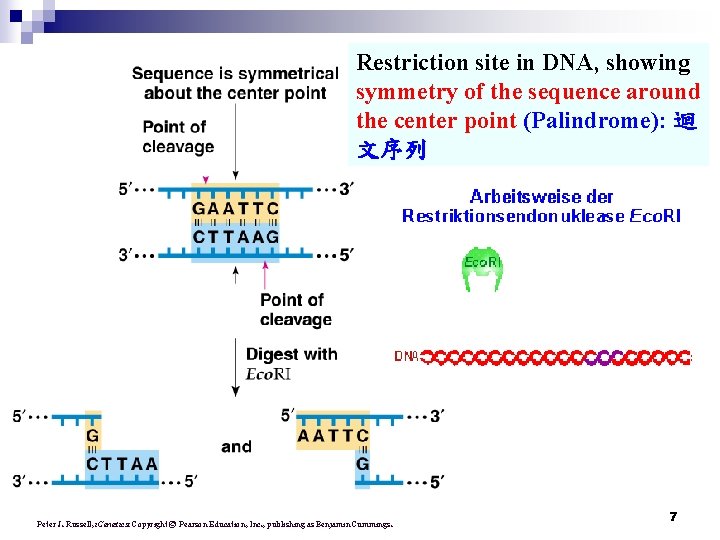
Restriction site in DNA, showing symmetry of the sequence around the center point (Palindrome): 迴 文序列 Peter J. Russell, i. Genetics: Copyright © Pearson Education, Inc. , publishing as Benjamin Cummings. 7
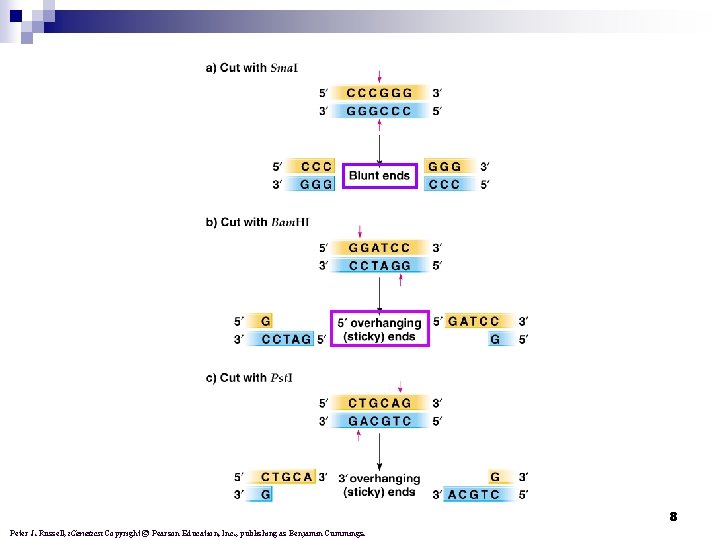
8 Peter J. Russell, i. Genetics: Copyright © Pearson Education, Inc. , publishing as Benjamin Cummings.





DNA ligase will seal a nick in a DNA strand can therefore be used to join two DNA fragments. It’s natural role is in repairing double strand breaks in DNA molecules. T 4 DNA ligase is able to join fragments with either blunt or sticky ends but bacterial ligases are only able to join fragments with sticky ends. It is commonly used for the insertion of restriction enzymegenerated DNA fragments into vector backbones. Commercial ligases are supplied with a reaction buffer containing ATP and Mg 2+, which are both essential for ligase activity. 16
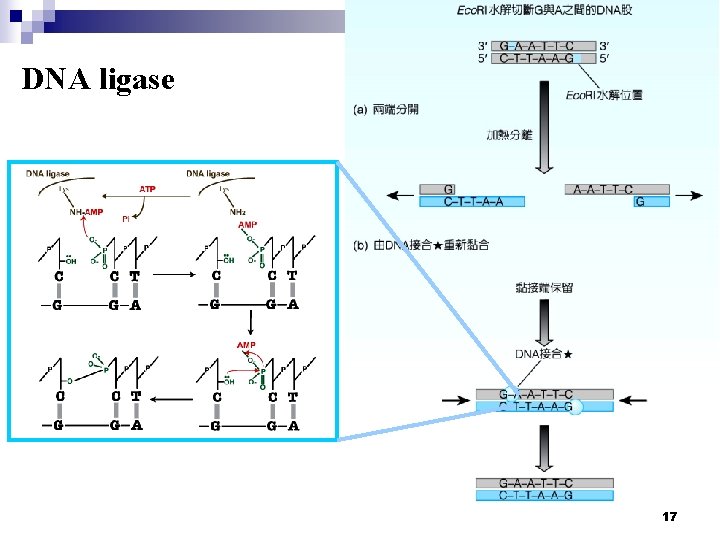
DNA ligase 17

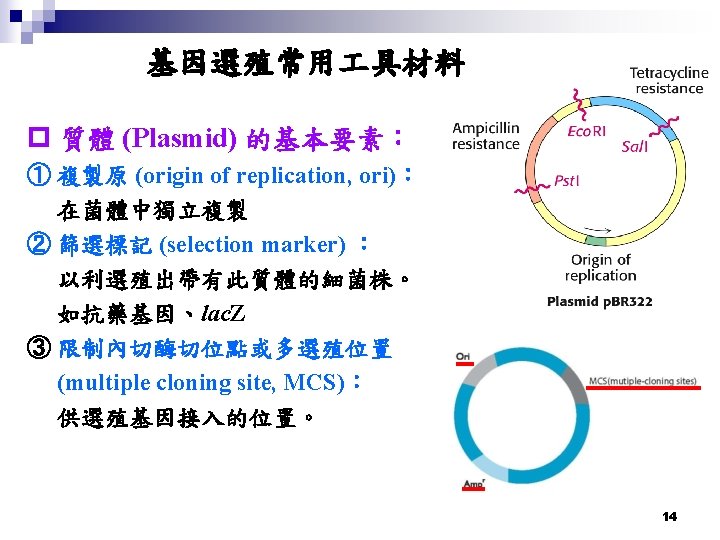
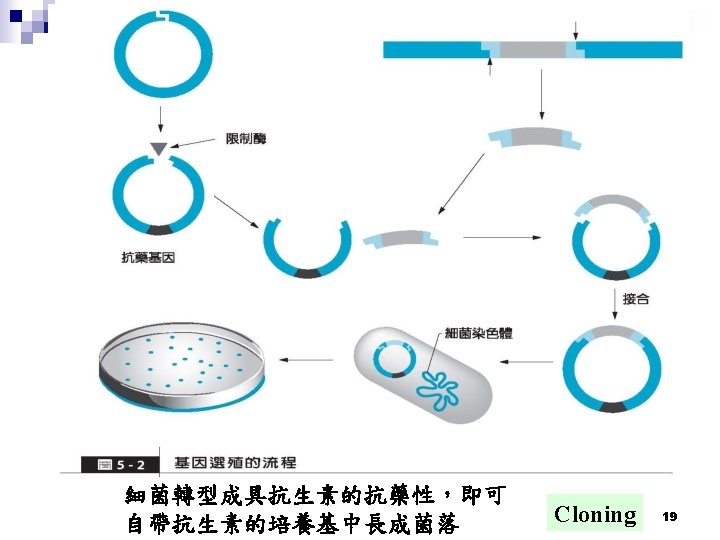
基因選殖技術操作的一般流程 ․專一性 DNA 內切酵素 (Restriction Enzymes) ․DNA 片段連接酵素 (T 4 DNA Ligase) ․DNA 片段轉接質/載體 (DNA fragment carrier. Plasmids/Vectors) ․將選殖基因轉型入微生物或細胞 (Transformation) ․轉型株的篩選 (Screening) 18
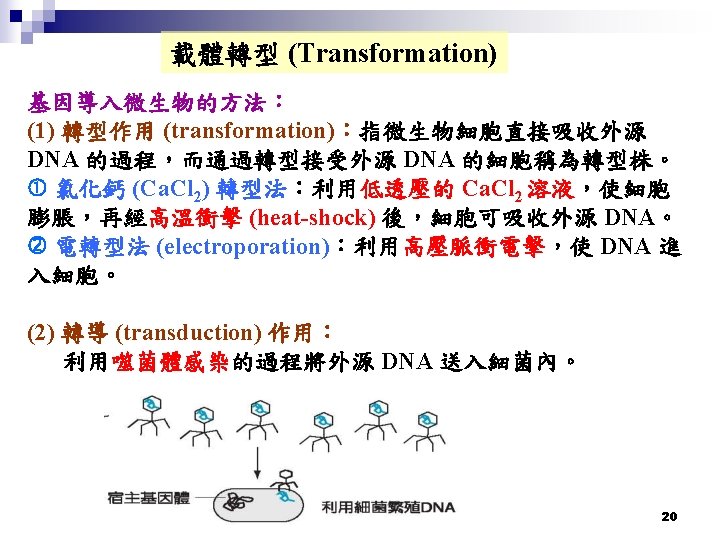
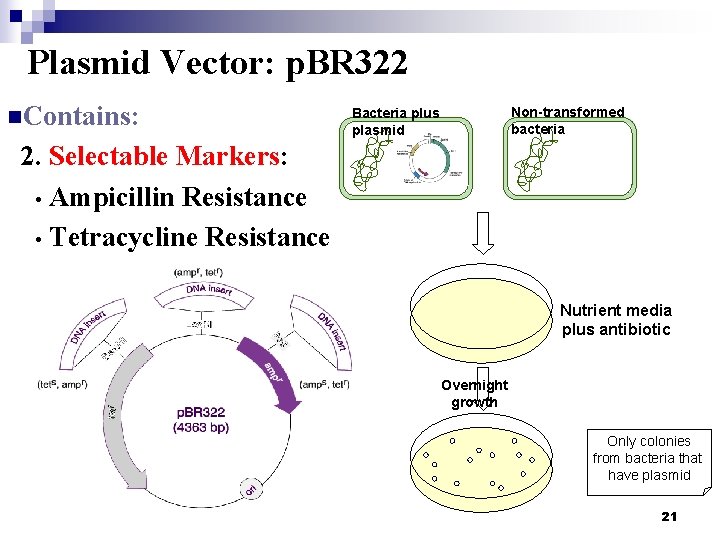
Plasmid Vector: p. BR 322 n. Contains: Non-transformed bacteria Bacteria plus plasmid 2. Selectable Markers: • Ampicillin Resistance • Tetracycline Resistance Nutrient media plus antibiotic Overnight growth Only colonies from bacteria that have plasmid 21
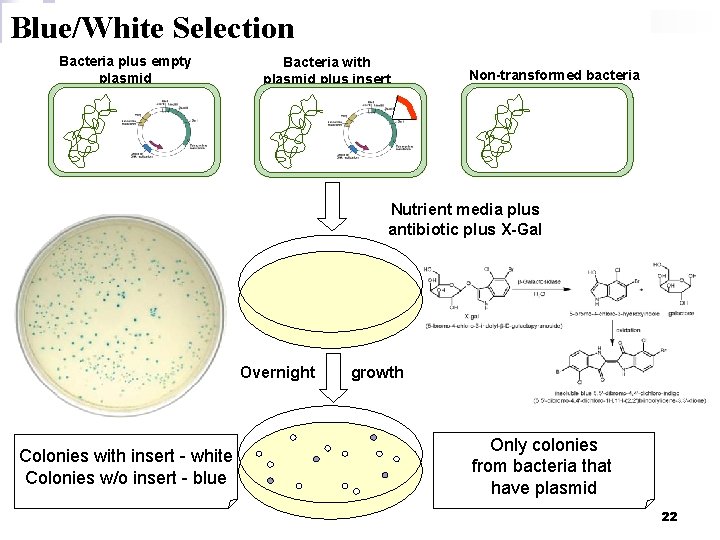
Blue/White Selection Bacteria plus empty plasmid Bacteria with plasmid plus insert Non-transformed bacteria Nutrient media plus antibiotic plus X-Gal Overnight Colonies with insert - white Colonies w/o insert - blue growth Only colonies from bacteria that have plasmid 22

寄主細胞與基因表現 (一) Host cell (1) High density of cell mass ; (2) Low culture or fermentation cost; (3) No toxicity; (4) Easy for metabolic controlling; (6) Easy for recombinant technique handling; (7) Highly yield, (8) Easy for product extraction and purification。 23
(一) Host cell Prokaryotic cells:大腸桿菌 (E. coli)、枯草芽胞桿 菌 (Bacillus subtilis)、鏈黴菌 (Streptomyces spp. ) 等; Eukaryotic cells:酵母菌 (yeast)、絲狀真菌 (filamentous fungi)、哺乳動物細胞等。 24

(一) Host cell: Prokaryotic cells (1) E. coli (gram negative bacteria) Advantages: The bacteria can also be grown easily and its genetics are comparatively simple and easily-manipulated, making it one of the best-studied prokaryotic model organisms. Disadvantages: a) Endocellular protein b) Inclusion body c) More difficult for product purification d) Lack of post-translational modification e) Endotoxin f) f) Proteases 25
(一) Host cell: Prokaryotic cells (2) Bacillus subtilis (枯草芽胞桿菌, gram positive bacteria) Advantages: have protein secretion ability and more easy for protein purification. Disadvantages: a) Less useful expression vectors b) Production yield is less than E. coli c) Lack of post-translational modification d) Higher extracellular protease activities 26

(一) Host cell: Prokaryotic cells (3) Streptomyces spp. (鏈黴菌, gram positive bacteria) In recent years, biotechnology researchers have begun to use Streptomyces spp. for production of recombinant human proteins. Advantages: p. No toxicity p. Have the ability to secrete correctly folded recombinant proteins into the medium p. Simplifying the purification steps p. Glycosylation p. Less RM ability in mutants 27
(一) Host cell: Eukaryotic cells (1) Yeast (酵母菌) - Saccharomyces cerevisiae p Yeasts are unicellular eukaryotic microorganisms. p Genetics are comparatively simple and easily-manipulated. p Short life cycle; Grown fast; lower fermentation cost; p No toxicity; Generally recognized as safe (GRAS) p Well developed gene expression systems p Extracellular expression (secretion to medium) p Reduced the protein purification steps p Glycosylation and post-translational modifications are similar to the multicellular eukaryotic cells 28

(一) Host cell: Eukaryotic cells (2) Filamentous fungi (絲狀真菌) Aspergillus niger (黑麴菌) DNA transformation systems have been developed for filamentous fungi. Advantages: a) Strong protein secretion ability b) Glycosylation and post-translational modifications are similar to the multicellular eukaryotic cells c) The fermentation processes are well developed d) Many filamentous fungi are defined as safe microorganism 29

(一) Host cell: Eukaryotic cells (3) Mammalian (哺乳動物細胞) Advantages: p Extracellular expression (secretion to medium) p Easy for products purification p Glycosylation and post-translational modifications Disadvantages: p cell growth slow; low yield; high cost of growth medium 30
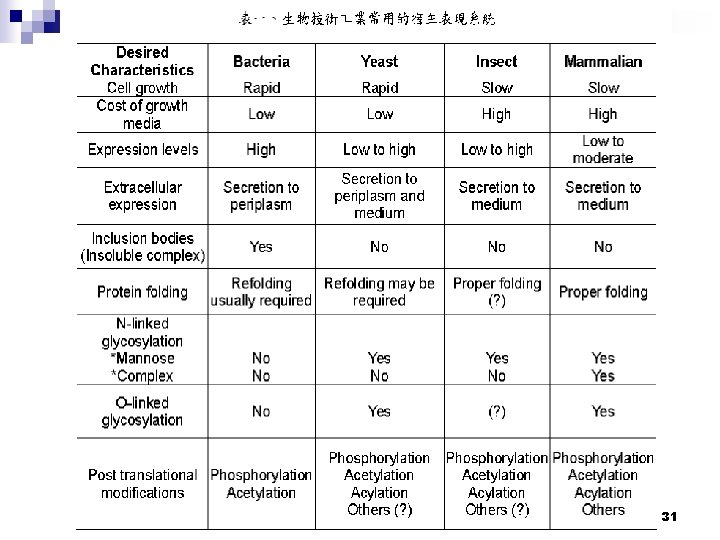
31

寄主細胞與基因表現 (二) Gene expression in E. coli The factors effect on the gene expression in E. coli (1) Copy number (2) Gene expression efficiency ① Promoter、② SD sequence、③ distance of SD to initiation codon (ATG)、④ codon usage (3) Product stability (4) Culture condition 32

(二) Gene expression in E. coli Promoter and inducting factor for gene expression in E. coli 33

Expression vector:p. QE system 34

寄主細胞與基因表現 (三) Gene expression in yeast 1. Vectors:A wide range of vectors are available to meet various requirements for insertion, deletion alteration and expression of genes in yeast (1) Classification by replication sequence of vector ① YEp (yeast episomal plasmid, 附加體型載體): - The YEp vectors replicate autonomously - The 2 μm ori is responsible for the high copy-number (20~100 plasmids/cell) and high frequency of transformation (103~105 transformants/μg DNA) of YEp vectors. 35

(三) Gene expression in yeast ② YIp 類 (yeast integrative plasmid,酵母整合型質體): - The Yp. I vectors do not replicate autonomously, but integrate into the genome at low frequencies (1~100 transformants/μg DNA) by homologous recombination - Linearization increases the efficiency of integrative transformation from 10 - to 50 -fold. - The YIp vectors typically integrate as a single copy - Strains transformed with YIp plasmids are extremely stable 36
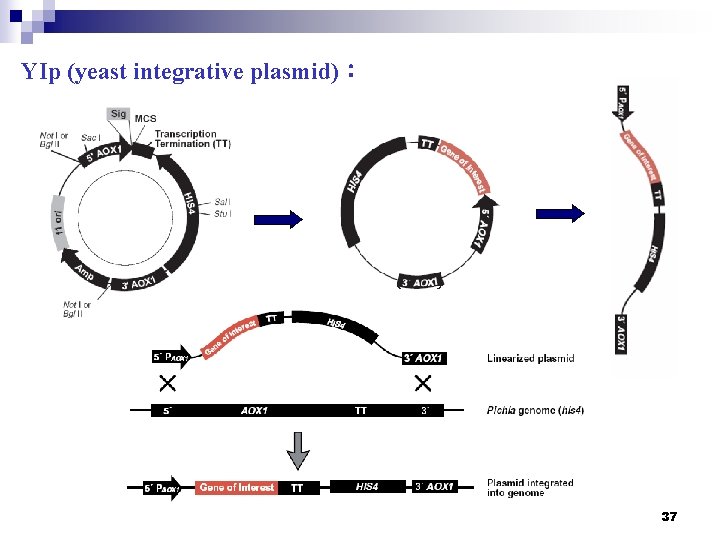
YIp (yeast integrative plasmid): 37

③ YCp 類 (yeast centromeric plasmid,酵母著絲粒質體): - The YCp vectors are autonomously replicating vectors - Very low copy numbers, from 1 to 3 per cell - Transformation efficiency is 103~104 transformants/μg DNA) - YCp is replicate during mitosis and meiosis - High stability - The stability and low copy-number of YCp vectors make them the ideal choice for cloning vectors, for construction of yeast genomic DNA libraries, and for investigating the function of genes altered in vivo. 38

④ YRp 類 (yeast replication plasmid,酵母複製型質體): - YRp vectors, containing ARS but lacking functional CEN elements, transform yeast at high frequencies, but are lost at too high a frequency (unstable), over 10% per generation, making them undesirable for general vectors. - Low copy numbers, from 5 to 10 per cell 39

(2) Shuttle vector: Which contain sequences permitting them to be selected and propagated in E. coli. The most common yeast vectors originated from p. BR 322 and contain an ori, promoting high copy-number maintenance in E. coli, and the selectable antibiotic markers, the β-lactamase gene and sometime to tetracycline-resistance gene. (3) Expression vector: Constructed the yeast promoter and terminator elements in the yeast vector for gene expression. 40
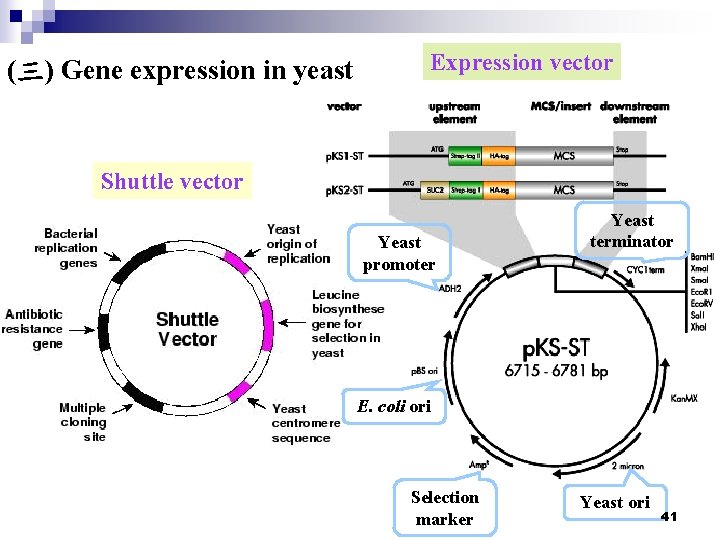
(三) Gene expression in yeast Expression vector Shuttle vector Yeast promoter Yeast terminator E. coli ori Selection marker Yeast ori 41



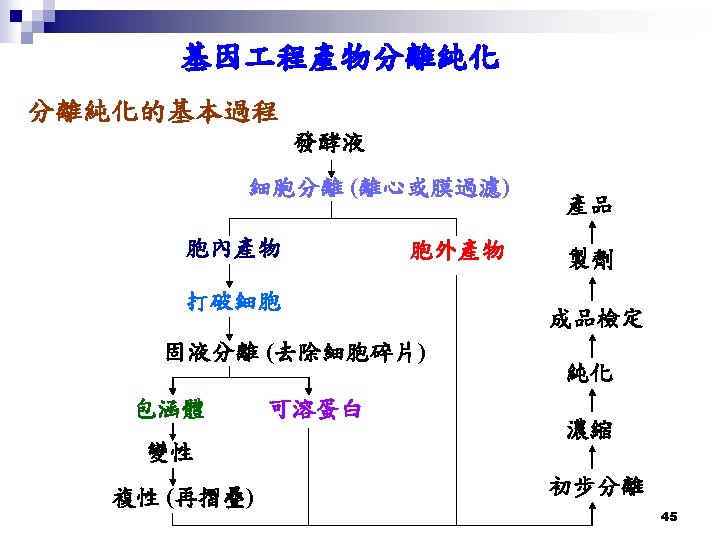

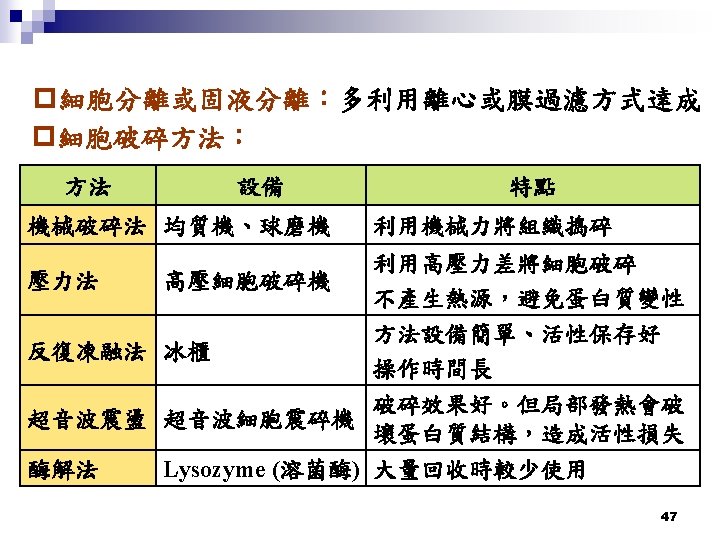



鹽析法 (salting-out) 50

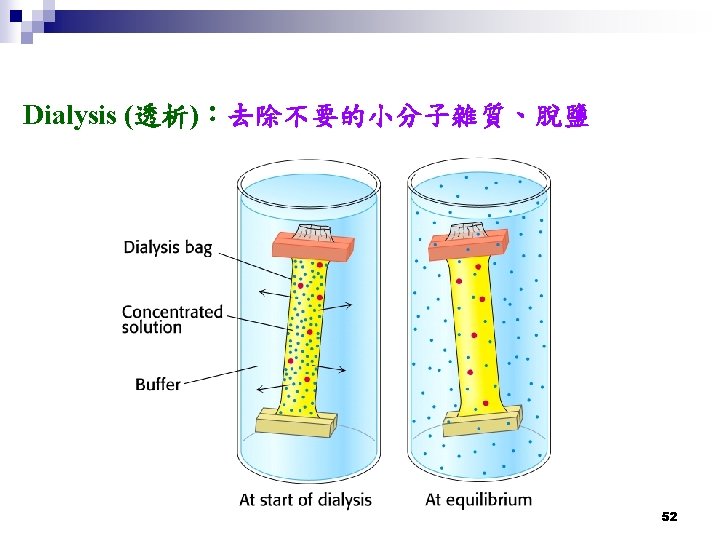
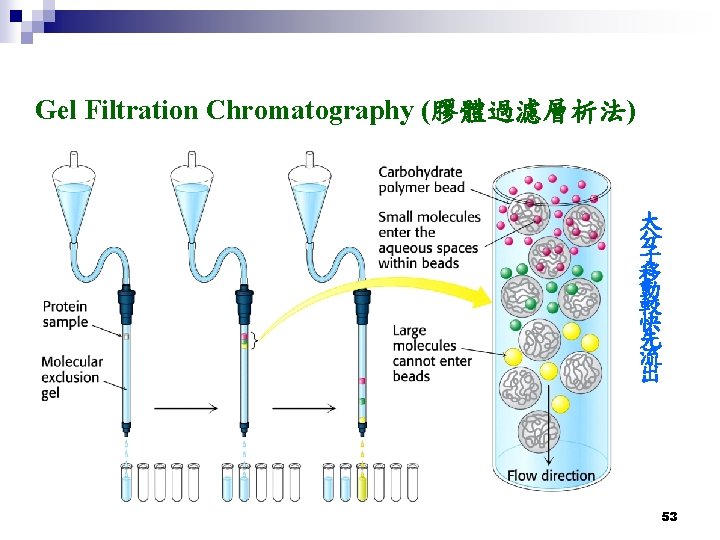
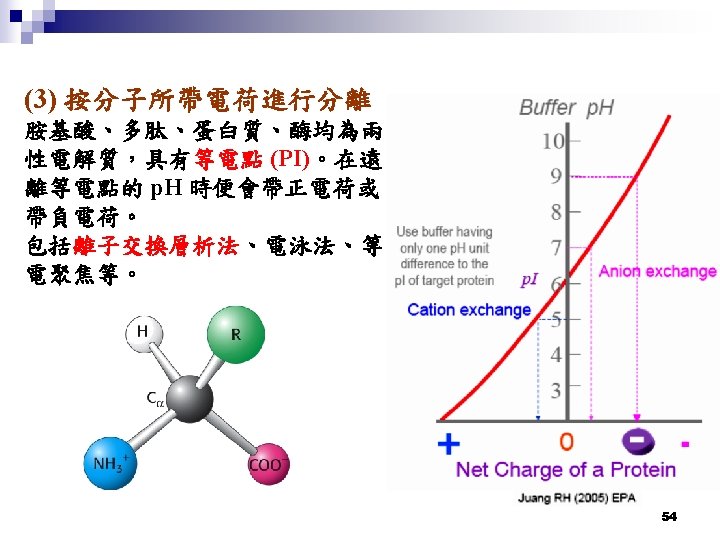
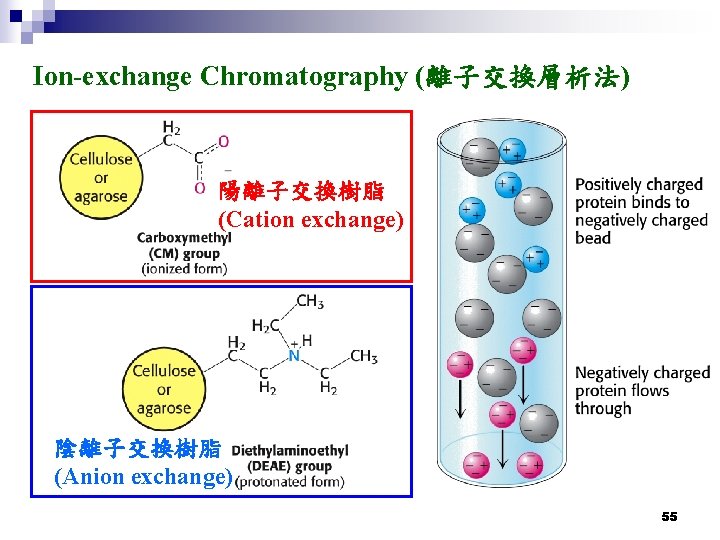
Ion-exchange Chromatography (離子交換層析法) 陽離子交換樹脂 (Cation exchange) 陰離子交換樹脂 (Anion exchange) 55
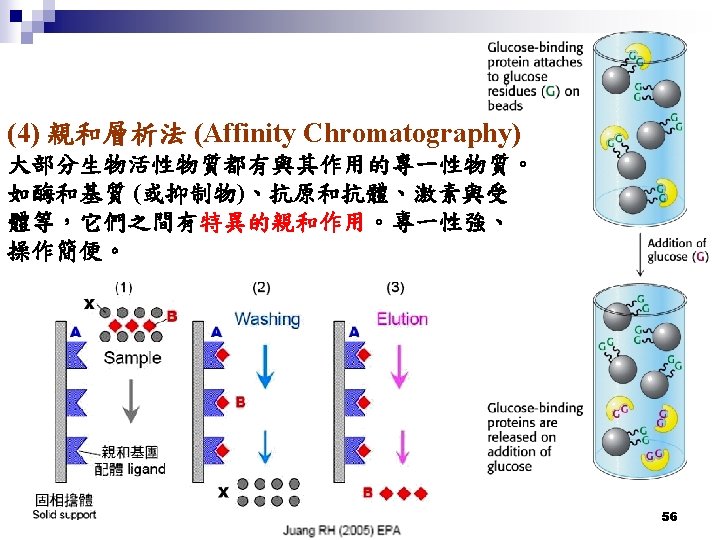
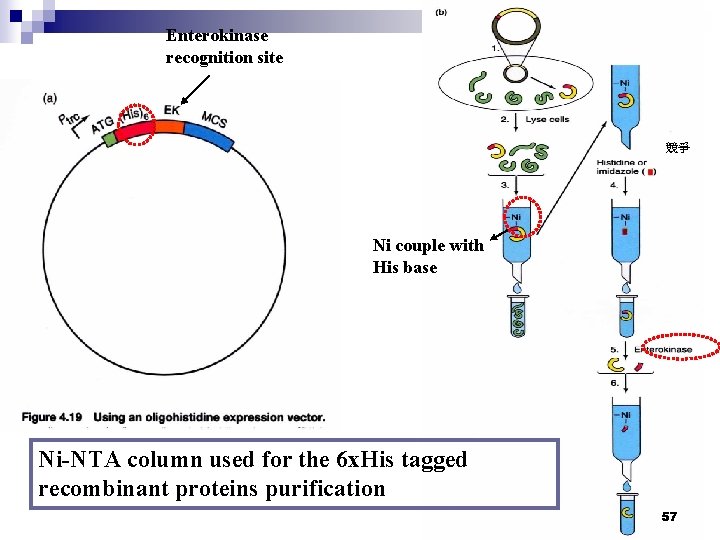
Enterokinase recognition site 競爭 Ni couple with His base Ni-NTA column used for the 6 x. His tagged recombinant proteins purification 57

Thanks for Your Attention 59
- Slides: 59