1895 Wilhelm Roentgen Using a Cathode Ray Tube


1895 Wilhelm Roentgen Using a Cathode Ray Tube (CRT) he observed that nearby chemicals glowed with fluorescent properties. Further experiments found penetrating rays coming from the CRT that were not deflected by a magnetic field. He named them "X-rays". 1896 Henri Becquerel While studying the effect of x-rays on photographic film, he discovered some chemicals spontaneously decompose and give off high penetrating rays. He discovered “radioactivity”. 1898 Ernest Rutherford Studied radiations emitted from uranium and thorium and named them alpha and beta. 1898 Marie Curie Studied uranium and thorium and called their spontaneous decay process "radioactivity". She and her husband Pierre also discovered the radioactive elements polonium and radium.

1905 Albert Einstein Published the famous equation E=mc 2 1911 Ernest Rutherford Using alpha particles as atomic bullets, probed the atoms in a piece of thin (0. 00006 cm) gold foil. He established that the nucleus was: very dense, very small and positively charged. 1938 Enrico Fermi Conducted the first controlled chain reaction releasing energy from the atoms nucleus. Received the Nobel prize in physics for producing new radioactive elements via neutron irradiation, and work with nuclear reactions. 19391941 – 51 Glenn Seaborg Synthesized 6 transuranium elements and suggested a change in the layout of the periodic table. Pioneer of the modern periodic table. 1940 July 16, 1945, the first atomic bomb was tested in an isolated area of the New Mexico desert. 6 kg of plutonium detonated. Shortly thereafter we decided to end WWII.

Nuclear Radiation is a form of ionizing radiation that results from changes within an atoms nucleus. Atoms with unstable nuclei can spontaneously change their identity. All elements with atomic numbers greater than 83 (Bismuth) are radioactive. Nonionizing Radiation (low energy UV – Radio): Low energy radiation that causes matter to vibrate (infrared) or move electrons to higher energy levels (visible). Ionizing Radiation (high energy UV, X-Rays, Gamma Rays, Cosmic Rays): High energy radiation that carries more potential harm and can cause serious damage to cells.
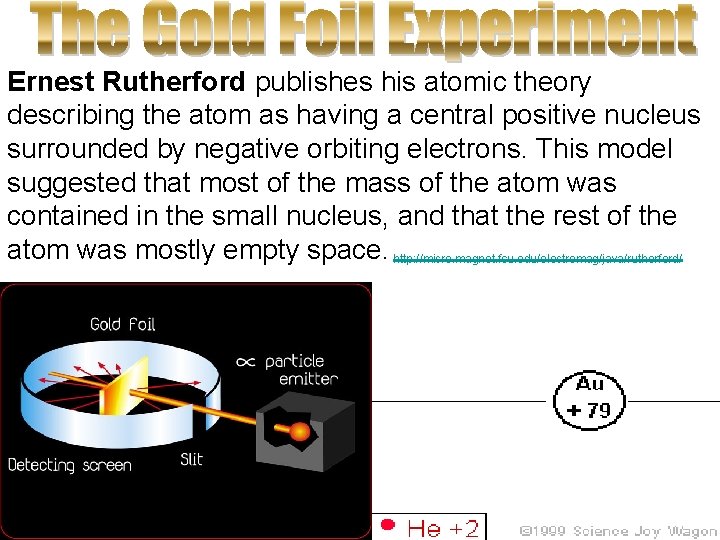
Ernest Rutherford publishes his atomic theory describing the atom as having a central positive nucleus surrounded by negative orbiting electrons. This model suggested that most of the mass of the atom was contained in the small nucleus, and that the rest of the atom was mostly empty space. http: //micro. magnet. fsu. edu/electromag/java/rutherford/

The nucleus is composed of nucleons (protons & neutrons) The nucleons have the same identical mass of about 1. 7 x 10 -24 grams. The mass for a proton & neutron is about 2000 times more than the mass of an electron. All atoms of an element have the same number of protons but all atoms of one element do not necessarily have the same number of neutrons. Atoms of the same element but having a different number of neutrons is called an isotope. (Iso- meaning “same”, tope- meaning “place”) Isotopes are distinguished by their different mass numbers. Remember the atomic mass is the total number of nucleons in an atom. (Protons + Neutrons) Isotopes are written with the name followed by the mass number. Example: Carbon – 12, Carbon – 13, Carbon – 14,
Isotopes are atoms of the same element having different masses due to varying numbers of neutrons. Isotope Protons Electrons Neutrons Hydrogen– 1 (protium) 1 1 0 Hydrogen-2 (deuterium) 1 1 1 Hydrogen-3 (tritium) 1 1 2 Nucleus Elements with atomic numbers greater than 83 (Bismuth) are naturally radioactive isotopes. Modern technology has made it possible to create a radioactive isotope of any element.

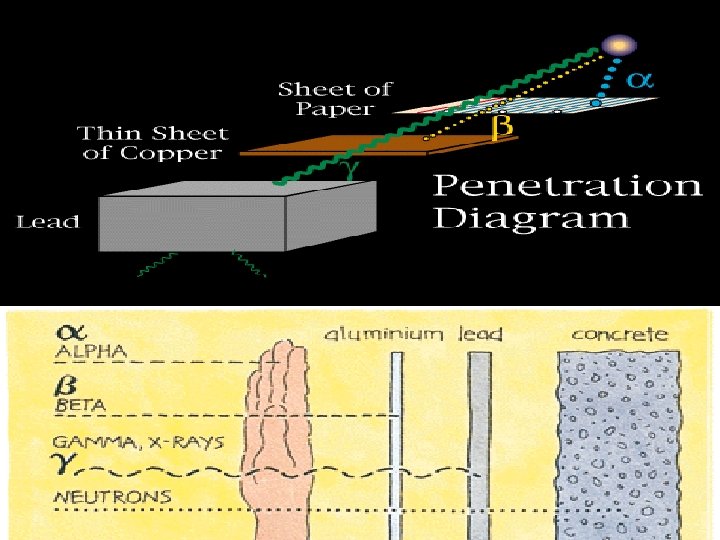
Types of Radiation Alpha particle ( ) w helium nucleus 2+ Beta particle ( -) w electron 1 - Positron ( +) w positron Gamma ( ) w high-energy photon paper lead 1+ concrete
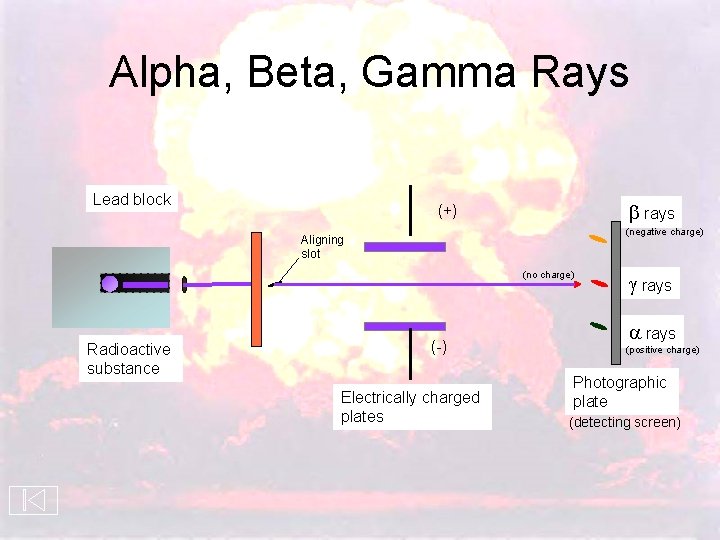
Alpha, Beta, Gamma Rays Lead block rays (+) (negative charge) Aligning slot (no charge) Radioactive substance (-) Electrically charged plates g rays a rays (positive charge) Photographic plate (detecting screen)

Types of Radiation • Alpha (ά) – a positively charged helium isotope • Beta (β) – an electron • Gamma (γ) – pure energy; called a ray rather than a particle

Other Nuclear Particles • Neutron • Positron – a positive electron • Proton – usually referred to as hydrogen-1

Natural or Background Radiation • We are all being exposed daily to a variety of radiation • We receive about 100 m. REM/year from background – The average non-occupational worker receives about 200 m. REM/year of chronic radiation exposure • Present at all times as a result of radiation naturally present in the environment – Cosmic rays – Uranium, thorium and radon in soil – Building materials • We receive an additional 100 m. REM/year from – Medical and dental x-rays – Smoke detectors – Dials on watches, etc. – Cell phones, TV’s, • Differs depending on geographical location

Radiation Dosages Dose (Amount + Energy) rad = radiation absorbed dose – absorbed radiation energy per kg of material Gray (Gy) SI Base Unit for absorbed dosage = 100 rad rem = radiation for roentgen equivalent man Sievert (Sv) SI Base Unit for radiation equivalent man = 100 rem (1 Gy = 100 rad, 1 Sv = 100 rem)
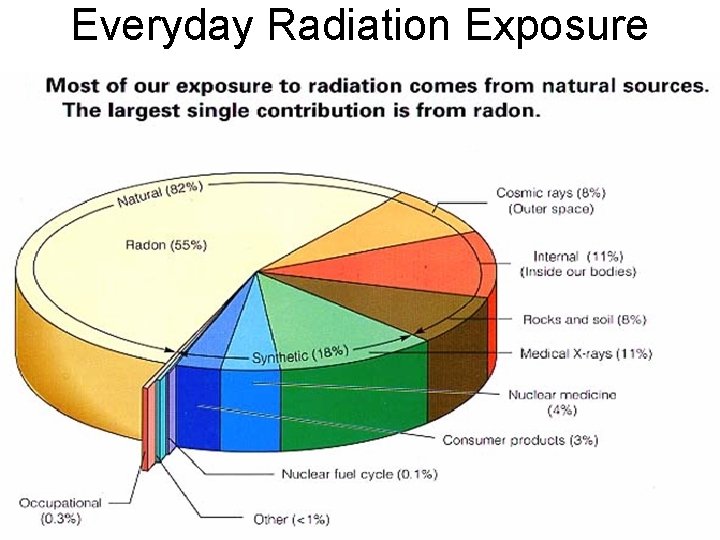
Everyday Radiation Exposure

Factors to Reduce Exposure • Time • Distance • Shielding

Time • If you decrease the time exposed to a given isotope you will decrease the dose of that exposure
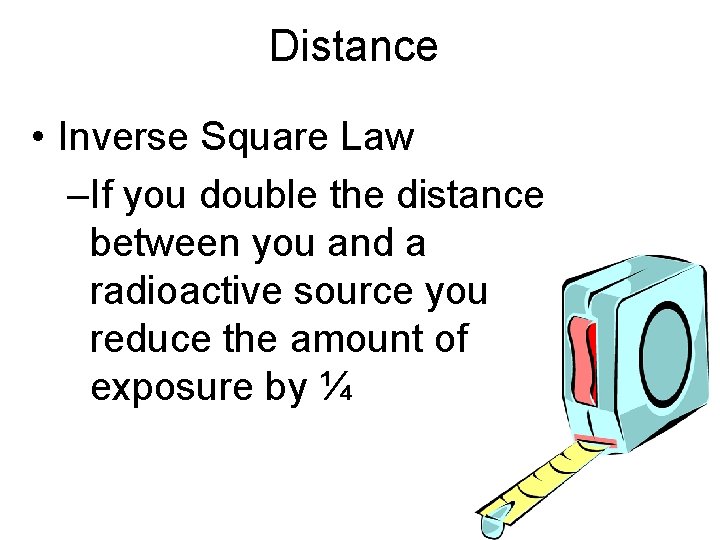
Distance • Inverse Square Law –If you double the distance between you and a radioactive source you reduce the amount of exposure by ¼

Shielding (Barrier between you and the source) • Type needed depends on type of radiation produced – Alpha • Air • Paper • Clothes – Beta • Metal • Wood • Plexiglass – Gamma • Concrete • Lead
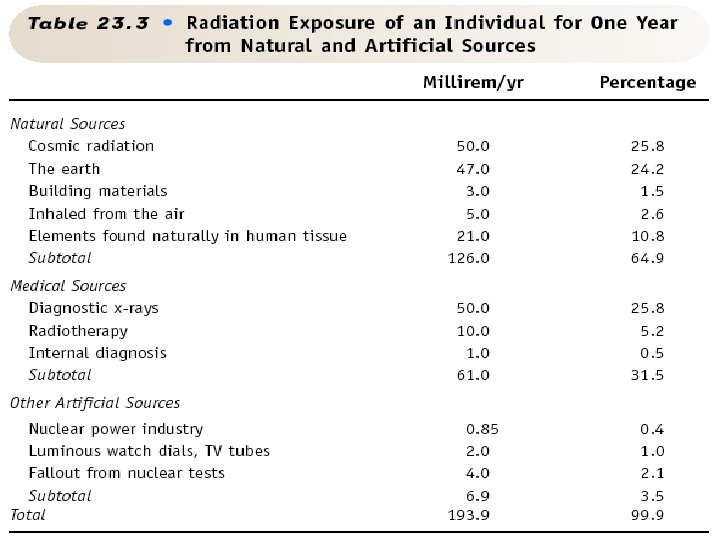

Dosages Required for Certain Immediate Effects • 0 -100 REM’s – Survival certain – No obvious symptoms – Maybe some clinical signs if lab tests are done • 100 -200 REM’s – Survival probable – Begins signs of light radiation sickness • Nausea • Vomiting • Listlessness • 200 -700 REM’s – Survival questionable. Some will survive, some won’t. – Severe radiation sickness – Radiation burns • Over 700 REM’s – Survival impossible

Radiation Measurement A Geiger counter • detects beta and gamma radiation. • uses ions produced by radiation to create an electrical current.
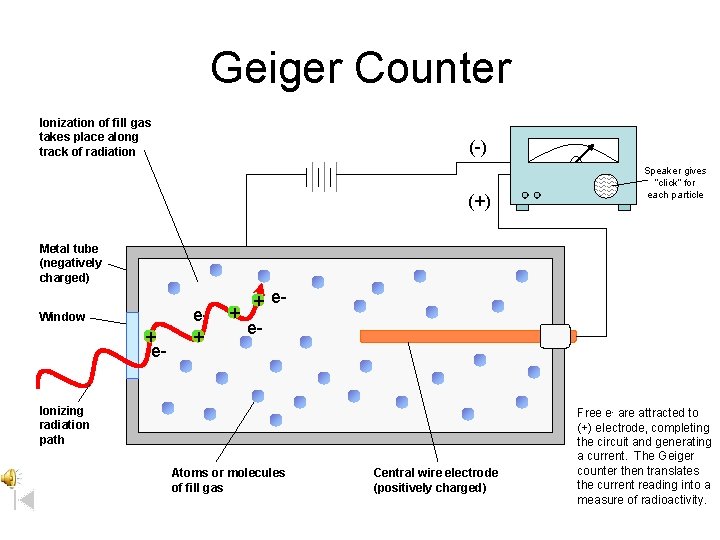
Geiger Counter Ionization of fill gas takes place along track of radiation (-) Speaker gives “click” for each particle (+) Metal tube (negatively charged) Window + e- e+ + + ee- Ionizing radiation path Atoms or molecules of fill gas Central wire electrode (positively charged) Free e- are attracted to (+) electrode, completing the circuit and generating a current. The Geiger counter then translates the current reading into a measure of radioactivity.

Most of the isotopes which occur naturally are stable. A few naturally occurring isotopes and all of the manmade isotopes are unstable. Unstable isotopes can become stable by releasing different types of particles. This process is called radioactive decay and the elements which undergo this process are called radioisotopes/radionuclides.

Radioactive Decay Radioactive decay results in the emission or capture of either: • an alpha particle (a), • a beta particle ( -), • a gamma ray (g), • a postitron ray ( +), • a proton (p+), • a neutron (no).

A X Z A (Mass Number) = number of protons + number of neutrons Z (Atomic Number) = number of protons A – Z = number of neutrons X = Element Symbol Number of neutrons = Mass Number – Atomic Number
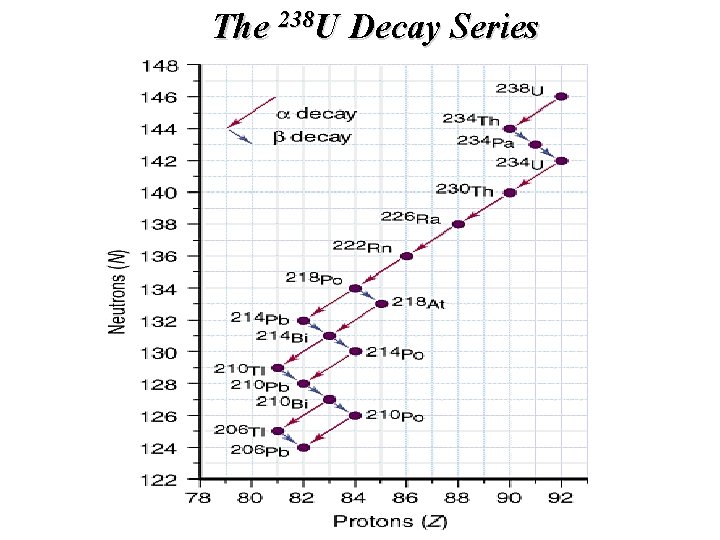
The 238 U Decay Series
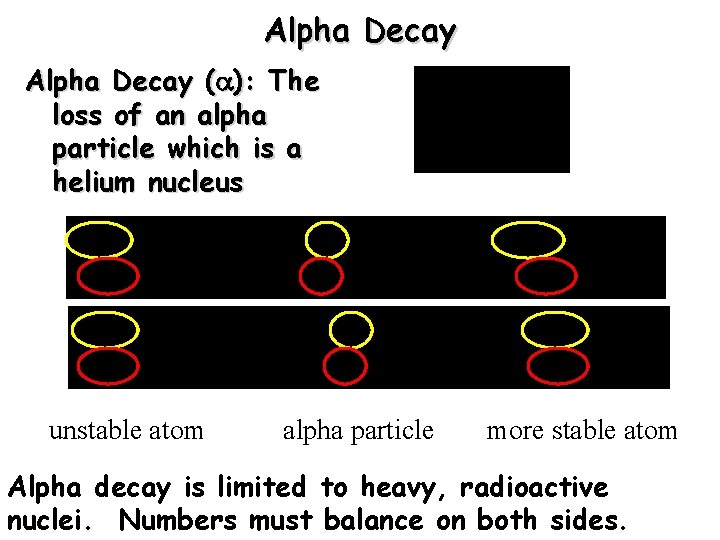
Alpha Decay ( ): The loss of an alpha particle which is a helium nucleus unstable atom alpha particle more stable atom Alpha decay is limited to heavy, radioactive nuclei. Numbers must balance on both sides.
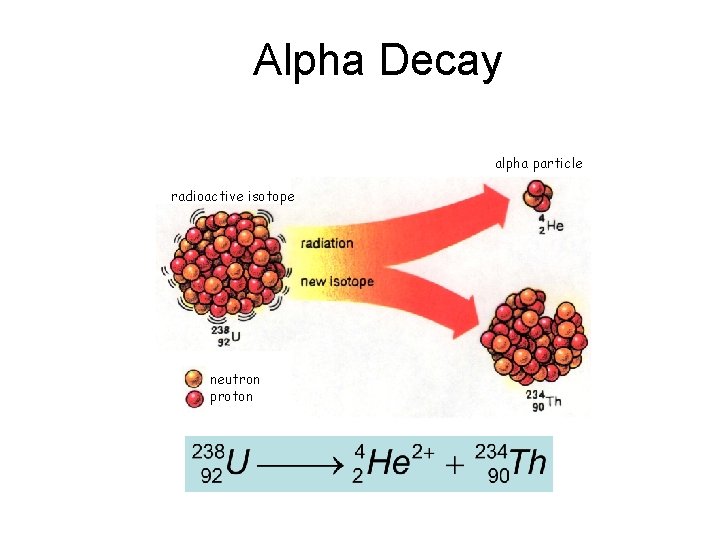
Alpha Decay alpha particle radioactive isotope neutron proton

Alpha Decay A A-4 4 226 222 4 X Z Ra 88 Y + Z-2 Rn + 86 Numbers must balance on both sides He 2
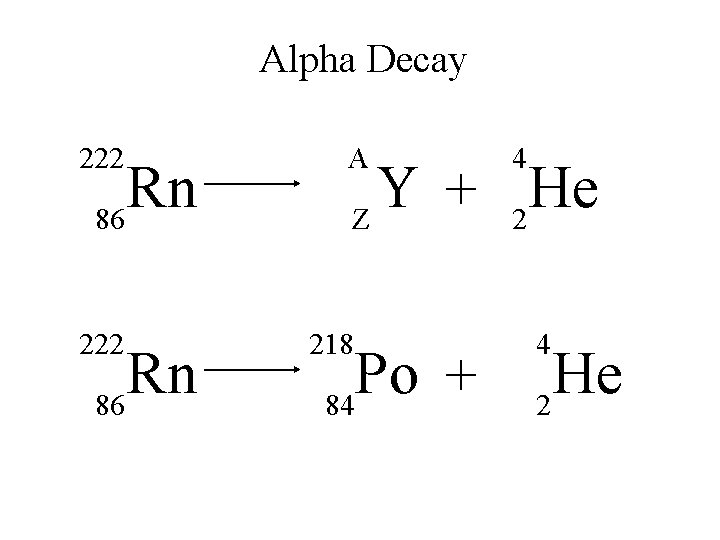
Alpha Decay 222 Rn 86 A 4 Y He + Z 2 218 Po + 84 4 He 2

Alpha Decay 230 Th 90 A 4 226 4 Y He + Z 2 Ra He + 88 2
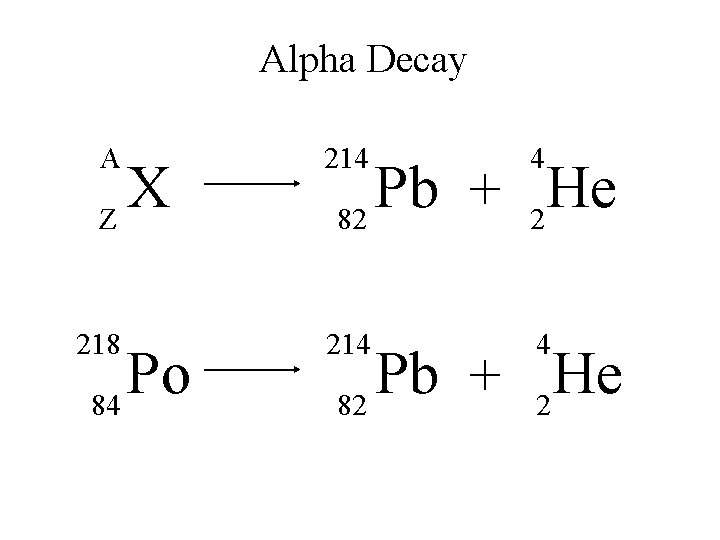
Alpha Decay A 214 4 218 214 4 X Z Po 84 Pb He + 82 2
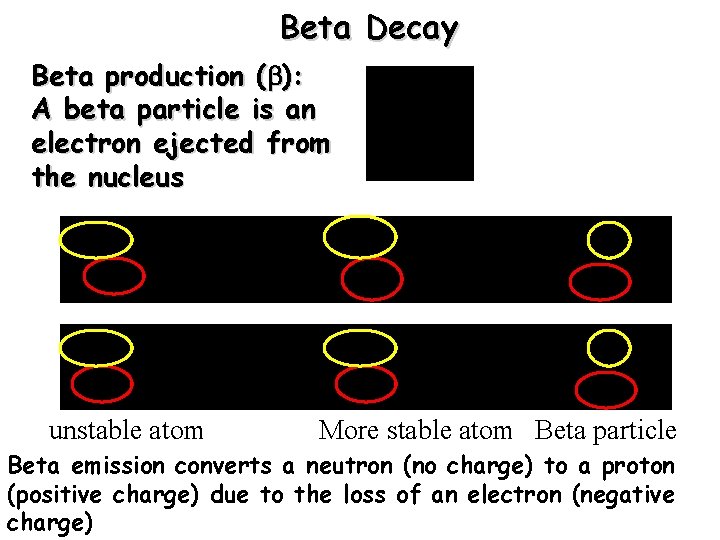
Beta Decay Beta production ( ): A beta particle is an electron ejected from the nucleus unstable atom More stable atom Beta particle Beta emission converts a neutron (no charge) to a proton (positive charge) due to the loss of an electron (negative charge)
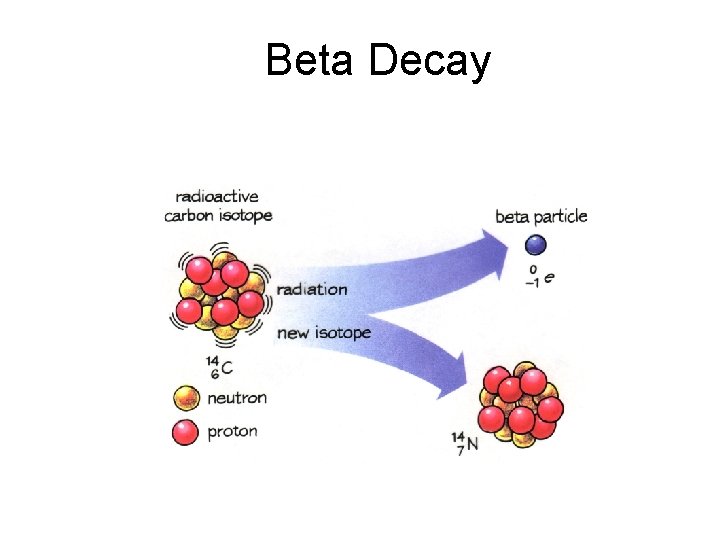
Beta Decay
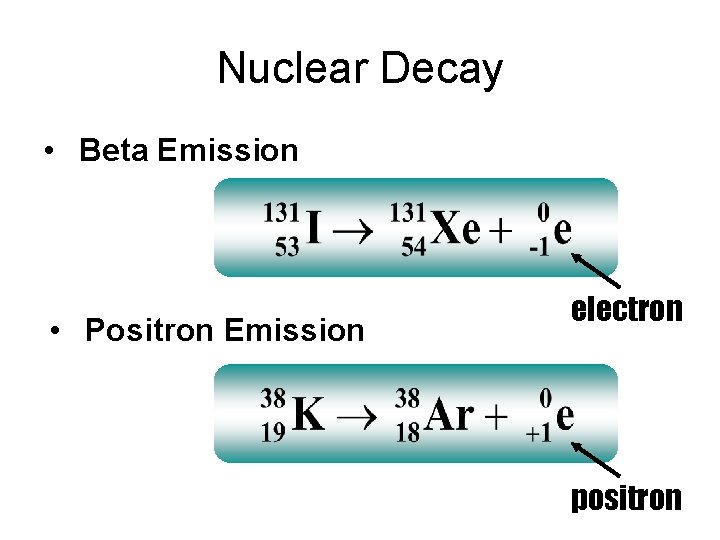
Nuclear Decay • Beta Emission • Positron Emission electron positron
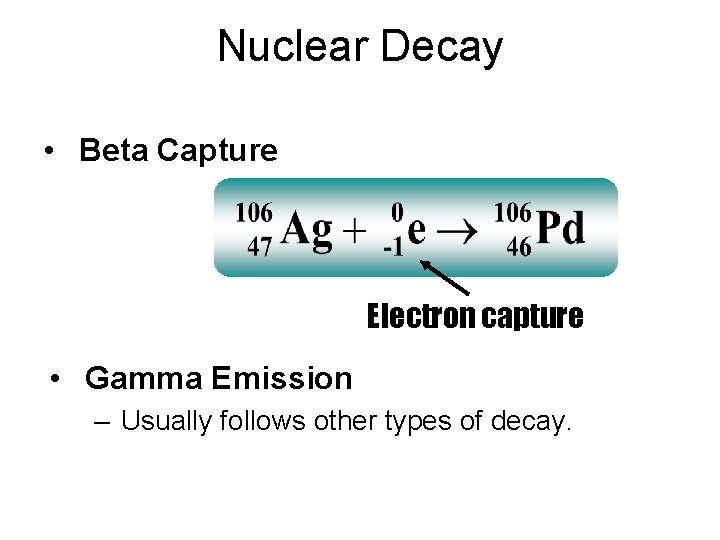
Nuclear Decay • Beta Capture Electron capture • Gamma Emission – Usually follows other types of decay.
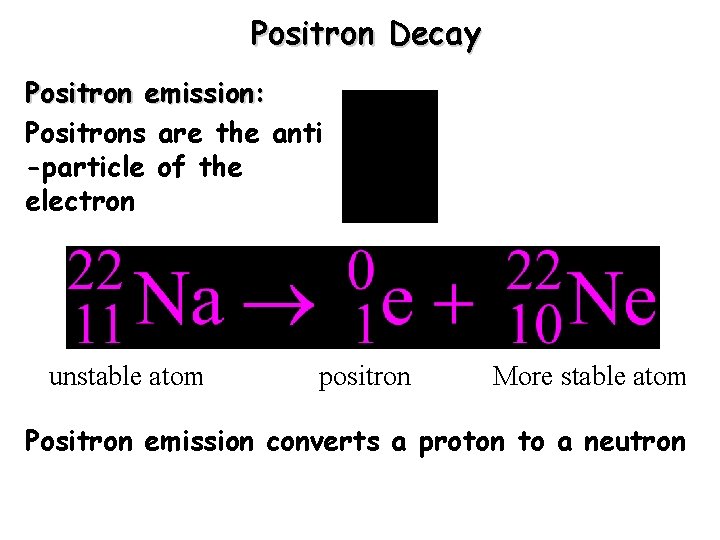
Positron Decay Positron emission: Positrons are the anti -particle of the electron unstable atom positron More stable atom Positron emission converts a proton to a neutron
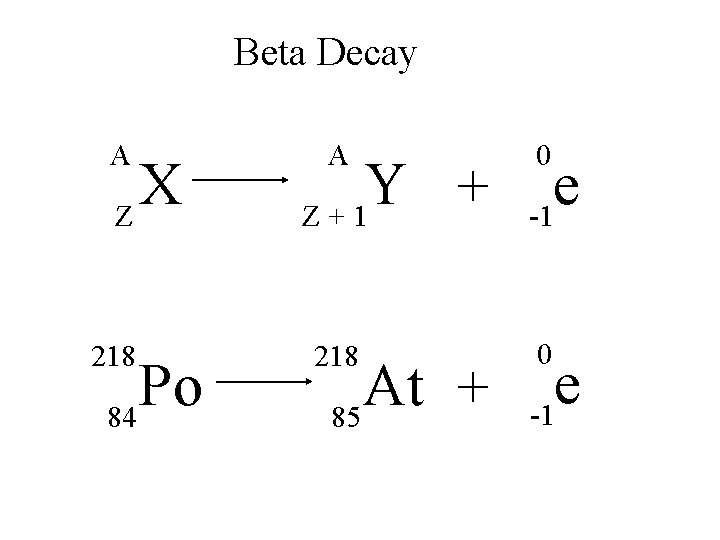
Beta Decay A X Z 218 Po 84 A Y + Z+1 218 At + 85 0 e -1
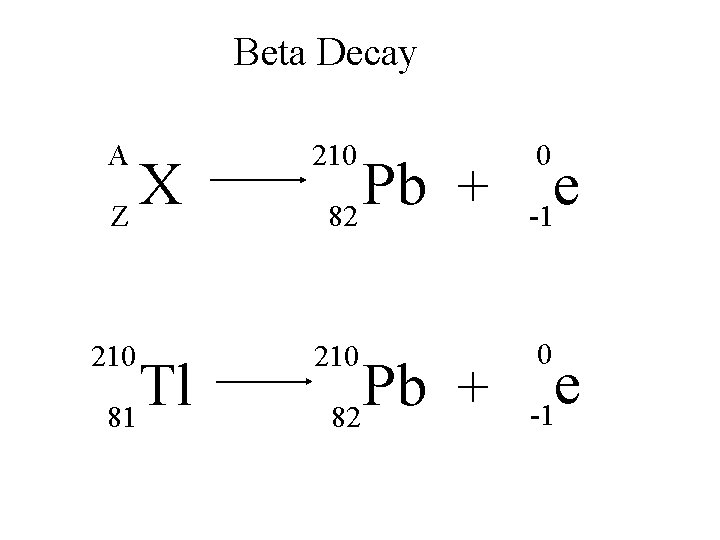
Beta Decay A 210 e -1 210 0 X Z Tl 81 Pb + 82 0 e -1

Beta Decay 210 A 210 Bi 83 Y + Z Po + 84 0 e -1
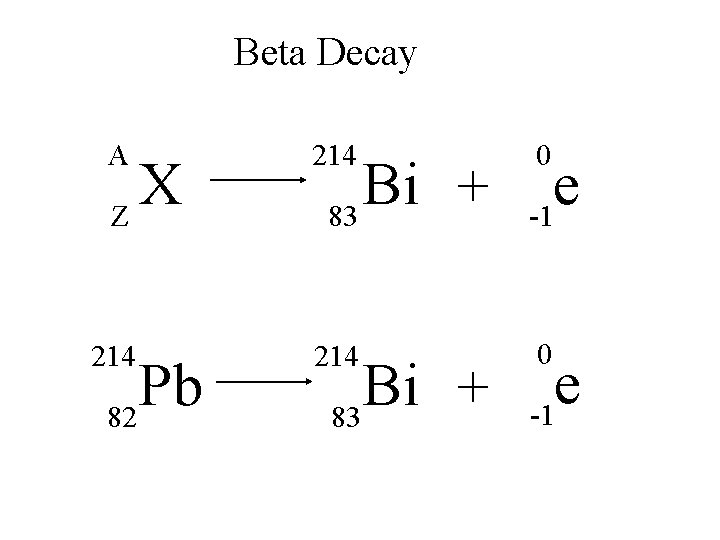
Beta Decay A 214 e -1 214 0 X Z Pb 82 Bi + 83 0 e -1

Gamma Ray Production Gamma ray production ( ): • Gamma rays are high energy photons produced in association with other forms of decay. Gamma rays are massless and do not, by themselves, change the nucleus

Gamma Decay When atoms decay by emitting a or particles to form a new atom, the nuclei of the new atom formed may still have too much energy to be completely stable. This excess energy is emitted as gamma rays (gamma ray photons have energies of ~ 1 x 10 -12 J).

Complete and balance the following reactions a. 7 1 H Li + 3 1 b. 3 1 c. 14 6 C 14 7 d. 26 12 Mg + 1 0 H + 2 1 4 He 2 H 1 0 + ________ n N + ____ n 0 +1 e + ____ For (e): Polonium – 214 goes through beta particle decay e. 214 Po + 84 0 e ____ -1

Half-Life • HALF-LIFE is the time that it takes for 1/2 a sample to decompose. • The rate of a nuclear transformation depends only on the “reactant” concentration.

Kinetics of Radioactive Decay For each duration (half-life), one half of the substance decomposes. For example: Ra-234 has a half-life of 3. 6 days If you start with 50 grams of Ra-234 After 3. 6 days > 25 grams After 7. 2 days > 12. 5 grams After 10. 8 days > 6. 25 grams

Half-life • The half-life (t 1/2) of a radioactive nuclide is the time required for one-half the nuclei in a sample of the nuclide to decay. For 1000 mg of I -131 to decay … … to 500 mg takes 8 days. It takes another 8 days for 500 mg to decay to 250 mg … … and 8 more days for 250 mg to decay to 125 mg.

Half-Life Is Characteristic of the Radioisotope

Radiocarbon Dating Radioactive C-14 is formed in the upper atmosphere by nuclear reactions initiated by neutrons in cosmic radiation 14 N + 1 n ---> 14 C + 1 H o The C-14 is oxidized to CO 2, which circulates through the biosphere. When a plant dies, the C-14 is not replenished. But the C-14 continues to decay with t 1/2 = 5730 years. Activity of a sample can be used to date the sample.

Learning Check! The half life of I-123 is 13 hr. How much of a 64 mg sample of I-123 is left after 39 hours?

Nuclear Medicine • Radioisotope Tracers – absorbed by specific organs and used to diagnose diseases • Radiation Treatment – larger doses are used to kill cancerous cells in targeted organs – internal or external radiation source Radiation treatment using -rays from cobalt-60.
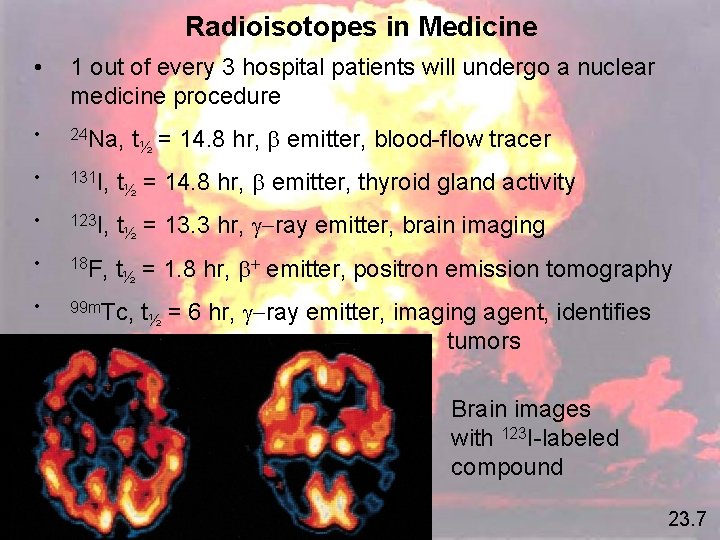
Radioisotopes in Medicine • 1 out of every 3 hospital patients will undergo a nuclear medicine procedure • 24 Na, t • 131 I, t ½ = 14. 8 hr, emitter, thyroid gland activity • 123 I, t ½ = 13. 3 hr, g-ray emitter, brain imaging • 18 F, t • 99 m. Tc, t ½ = 14. 8 hr, emitter, blood-flow tracer ½ = 1. 8 hr, + emitter, positron emission tomography ½ = 6 hr, g-ray emitter, imaging agent, identifies tumors Brain images with 123 I-labeled compound 23. 7

Food Irradiation • Food can be irradiated with rays from 60 Co or 137 Cs. • Irradiated milk has a shelf life of 3 mo. without refrigeration. • USDA has approved irradiation of meats and eggs.

Transmutations • The changing of one element to another is called transmutation • This occurs whenever there is an alpha decay or a beta decay • Remember, that for a gamma decay, the nucleus just changes internal energy levels, but doesn’t change the identity of nucleons

Synthetic Elements • Transuranium Elements – elements with atomic #s above 92 – synthetically produced in nuclear reactors and accelerators – most decay very rapidly

Energy and Mass Nuclear changes occur with small but measurable losses of mass. The lost mass is called the mass defect, and is converted to energy according to Einstein’s equation: E = mc 2 m = mass defect E = change in energy c = speed of light (3. 0 Because c 2 is so large, even small amounts of mass are converted to enormous amount of energy. x 108 m/s)

Nuclear Binding Energy • The energy released in forming a nucleus from its protons and neutrons is called the nuclear binding energy. • Although the energy is released, binding energy is expressed as a positive quantity. • Alternatively, nuclear binding energy is the quantity of energy necessary to separate a nucleus into individual protons and neutrons.
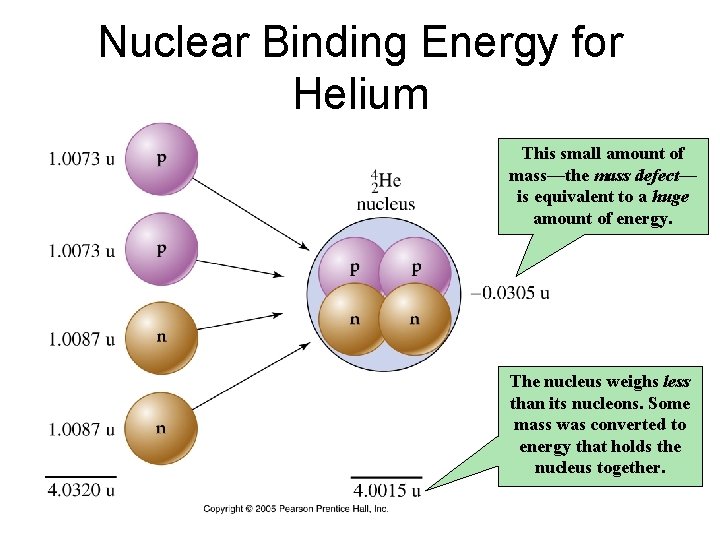
Nuclear Binding Energy for Helium This small amount of mass—the mass defect— is equivalent to a huge amount of energy. The nucleus weighs less than its nucleons. Some mass was converted to energy that holds the nucleus together.
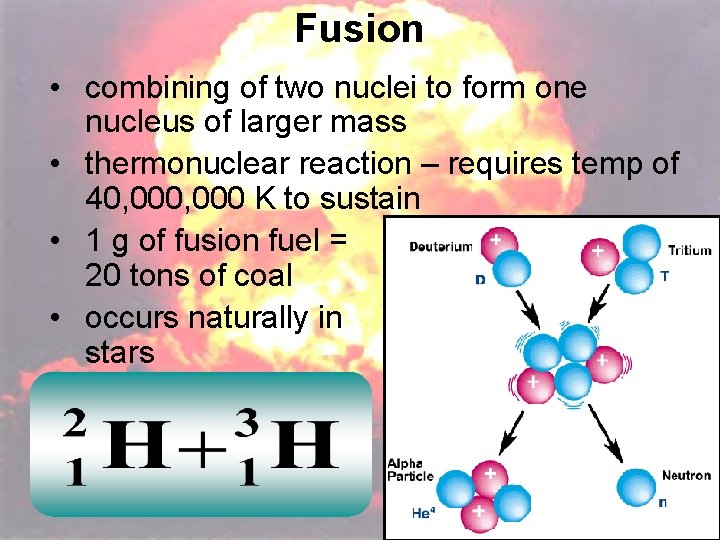
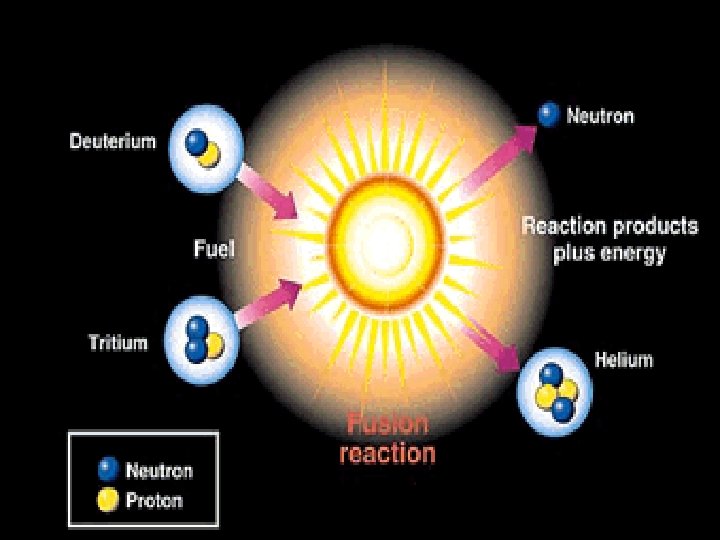
Fusion • combining of two nuclei to form one nucleus of larger mass • thermonuclear reaction – requires temp of 40, 000 K to sustain • 1 g of fusion fuel = 20 tons of coal • occurs naturally in stars
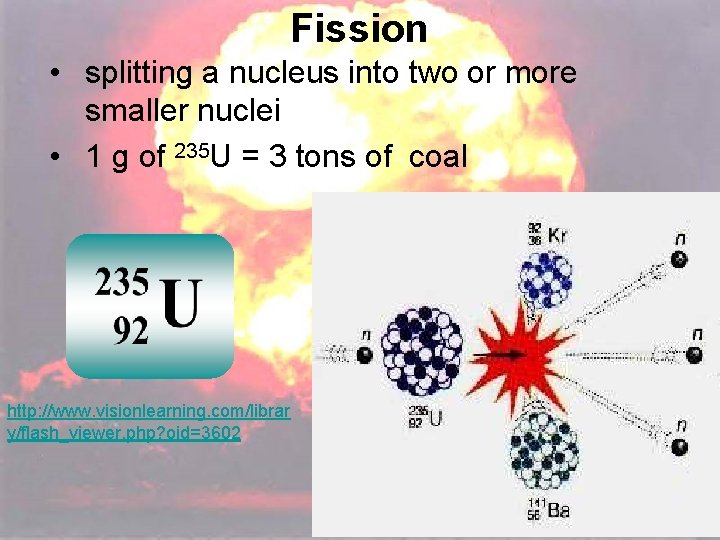
Fission • splitting a nucleus into two or more smaller nuclei • 1 g of 235 U = 3 tons of coal http: //www. visionlearning. com/librar y/flash_viewer. php? oid=3602

Fission vs. Fusion F I S S I O N • 235 U is limited • danger of meltdown • toxic waste • thermal pollution F U S I O N • fuel is abundant • no danger of meltdown • no toxic waste • not yet sustainable

Nuclear Weapons • Atomic Bomb (Fission Bomb) – chemical explosion is used to form a critical mass of 235 U or 239 Pu – fission develops into an uncontrolled chain reaction • Hydrogen Bomb – chemical explosion fission fusion – fusion increases the fission rate – more powerful than the atomic bomb http: //www. visionlearning. com/library/flash_viewer. php? oi d=3602




Nuclear Fission & POWER • Currently about 103 nuclear power plants in the U. S. and about 435 worldwide. • 17% of the world’s energy comes from nuclear.
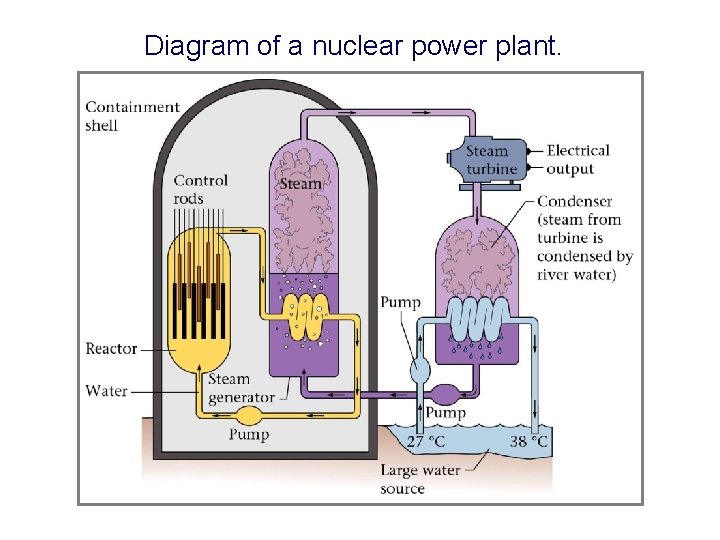
Diagram of a nuclear power plant.
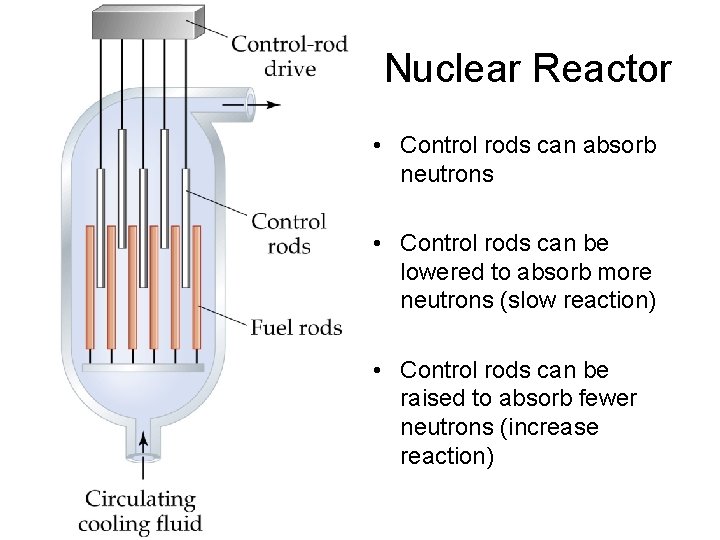
Nuclear Reactor • Control rods can absorb neutrons • Control rods can be lowered to absorb more neutrons (slow reaction) • Control rods can be raised to absorb fewer neutrons (increase reaction)
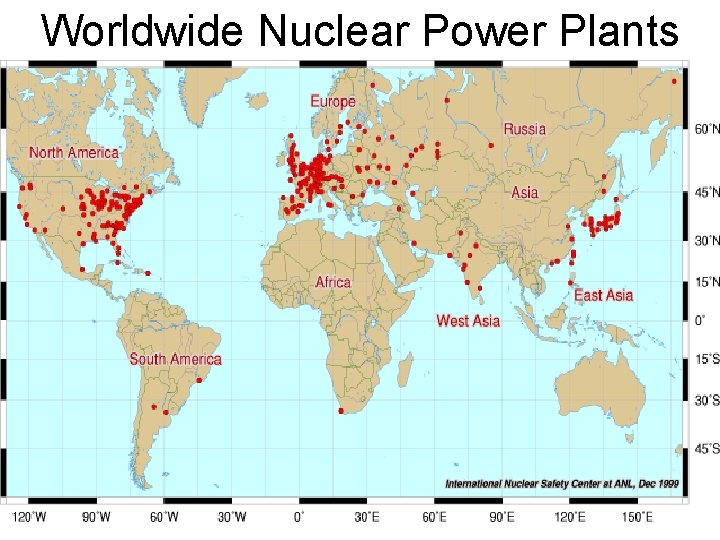
Worldwide Nuclear Power Plants
- Slides: 70