18 Ethers and Epoxides Thiols and Sulfides Based

18: Ethers and Epoxides; Thiols and Sulfides Based on Mc. Murry’s Organic Chemistry, 6 th edition © 2003 Ronald Kluger Department of Chemistry University of Toronto Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003

Ethers and Their Relatives n An ether has two organic groups (alkyl, aryl, or vinyl) bonded to the same oxygen atom, R–O–R n Diethyl ether is used industrially as a solvent n Tetrahydrofuran (THF) is a solvent that is a cyclic ether n Thiols (R–S–H) and sulfides (R–S–R ) are sulfur (for oxygen) analogs of alcohols and ethers Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 2

18. 1 Naming Ethers n Simple ethers are named by identifying the two organic substituents and adding the word ether n If other functional groups are present, the ether part is considered an alkoxy substituent Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 3

18. 2 Structure, Properties, and Sources of Ethers n R–O–R ~ tetrahedral bond angle (112° in dimethyl ether) n Oxygen is sp 3 -hybridized n Oxygen atom gives ethers a slight dipole moment n Diethyl ether prepared industrially by sulfuric acid– catalyzed dehydration of ethanol – also with other primary alcohols Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 4

18. 3 The Williamson Ether Synthesis n Reaction of metal alkoxides and primary alkyl halides and tosylates n Best method for the preparation of ethers n Alkoxides prepared by reaction of an alcohol with a strong base such as sodium hydride, Na. H Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 5
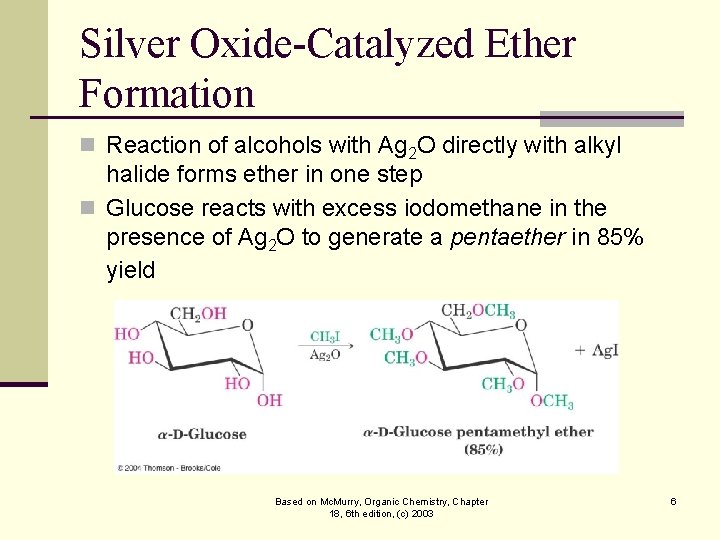
Silver Oxide-Catalyzed Ether Formation n Reaction of alcohols with Ag 2 O directly with alkyl halide forms ether in one step n Glucose reacts with excess iodomethane in the presence of Ag 2 O to generate a pentaether in 85% yield Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 6
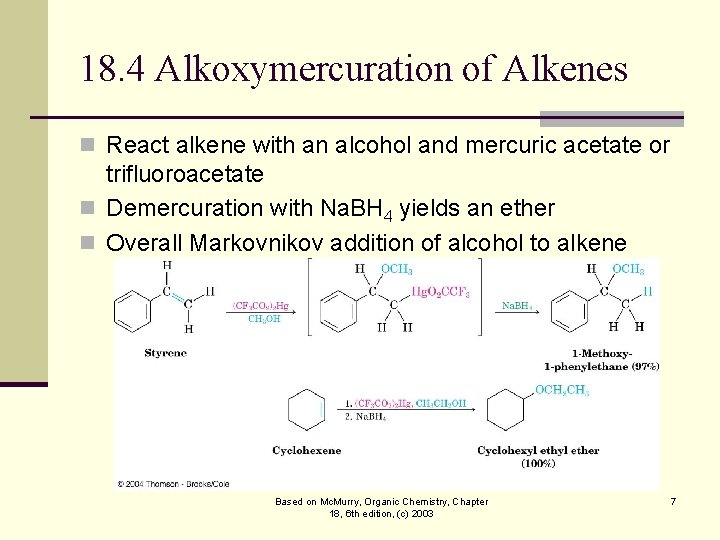
18. 4 Alkoxymercuration of Alkenes n React alkene with an alcohol and mercuric acetate or trifluoroacetate n Demercuration with Na. BH 4 yields an ether n Overall Markovnikov addition of alcohol to alkene Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 7

18. 5 Reactions of Ethers: Acidic Cleavage n Ethers are generally unreactive n Strong acid will cleave an ether at elevated temperature n HI, HBr produce an alkyl halide from less hindered component by SN 2 (tertiary ethers undergo SN 1) Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 8
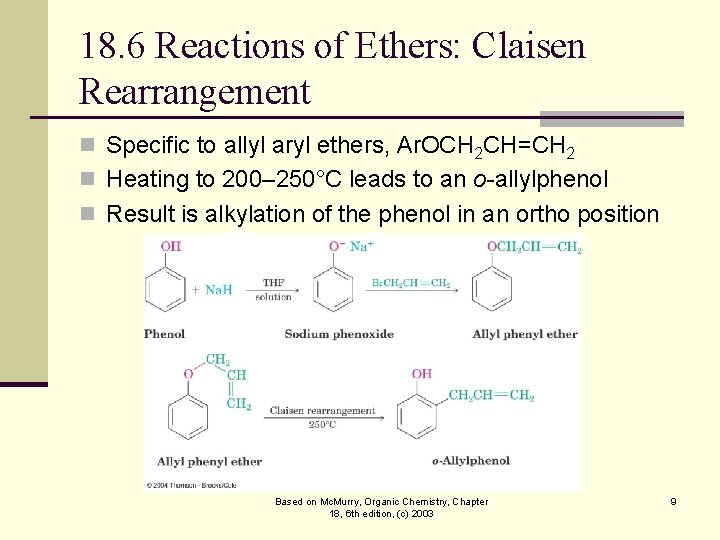
18. 6 Reactions of Ethers: Claisen Rearrangement n Specific to allyl aryl ethers, Ar. OCH 2 CH=CH 2 n Heating to 200– 250°C leads to an o-allylphenol n Result is alkylation of the phenol in an ortho position Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 9
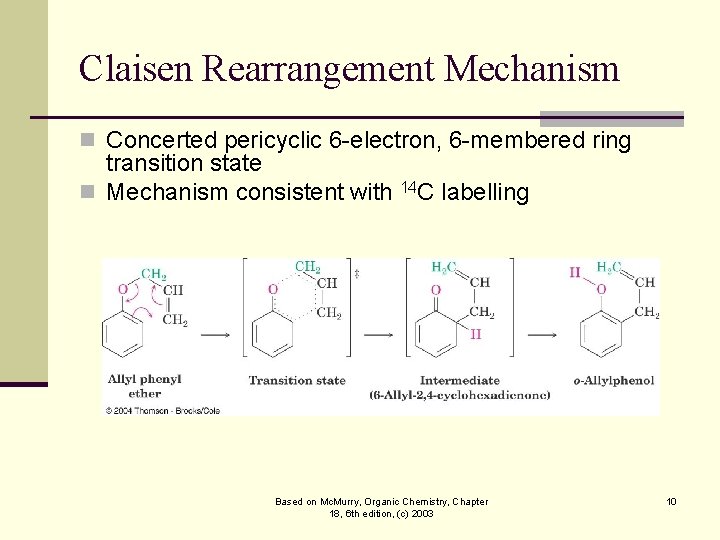
Claisen Rearrangement Mechanism n Concerted pericyclic 6 -electron, 6 -membered ring transition state n Mechanism consistent with 14 C labelling Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 10
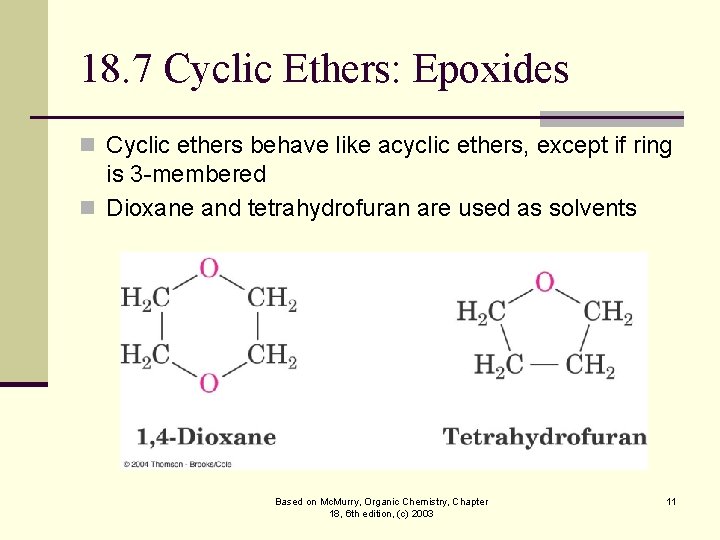
18. 7 Cyclic Ethers: Epoxides n Cyclic ethers behave like acyclic ethers, except if ring is 3 -membered n Dioxane and tetrahydrofuran are used as solvents Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 11
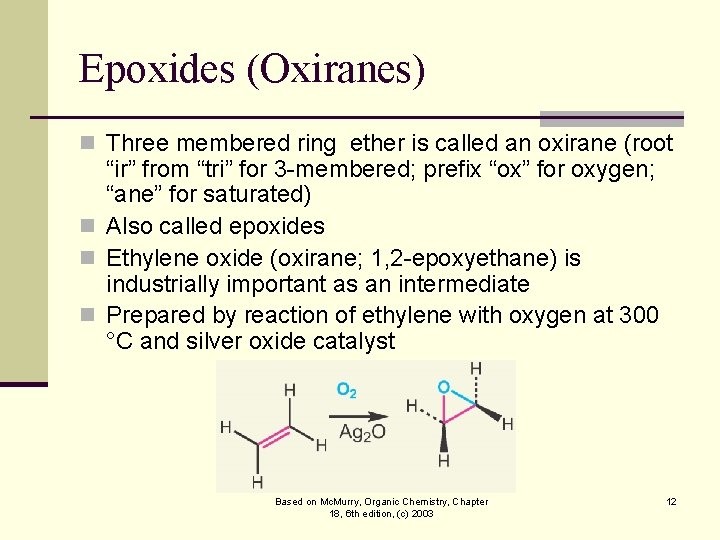
Epoxides (Oxiranes) n Three membered ring ether is called an oxirane (root “ir” from “tri” for 3 -membered; prefix “ox” for oxygen; “ane” for saturated) n Also called epoxides n Ethylene oxide (oxirane; 1, 2 -epoxyethane) is industrially important as an intermediate n Prepared by reaction of ethylene with oxygen at 300 °C and silver oxide catalyst Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 12
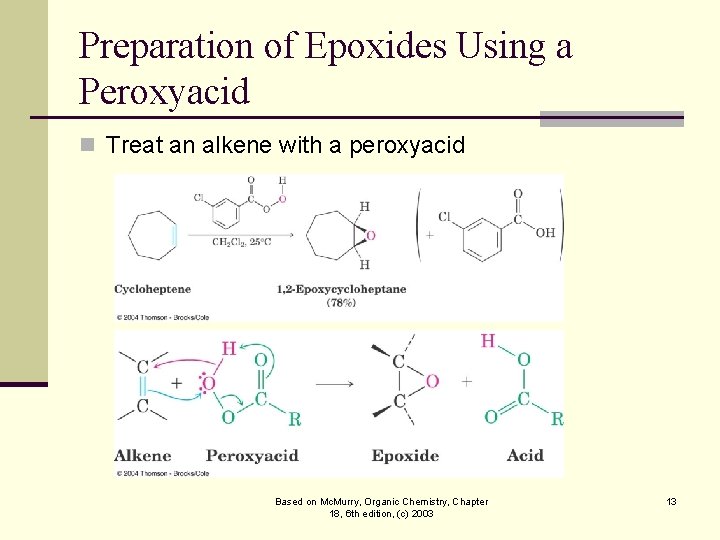
Preparation of Epoxides Using a Peroxyacid n Treat an alkene with a peroxyacid Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 13
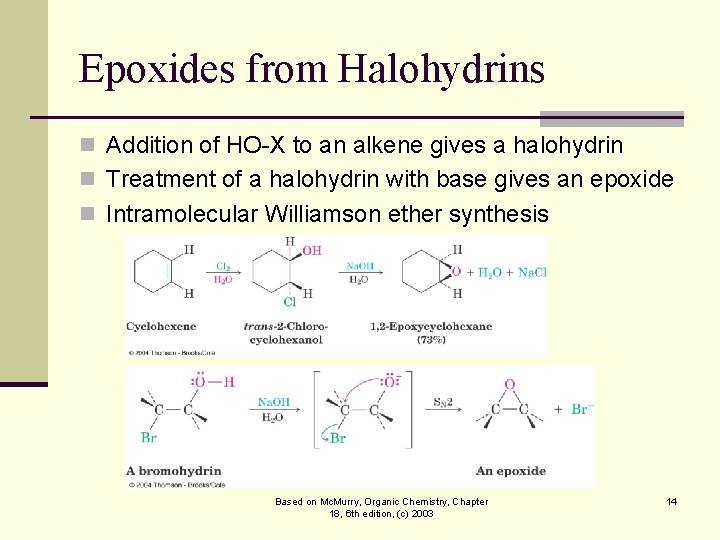
Epoxides from Halohydrins n Addition of HO-X to an alkene gives a halohydrin n Treatment of a halohydrin with base gives an epoxide n Intramolecular Williamson ether synthesis Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 14
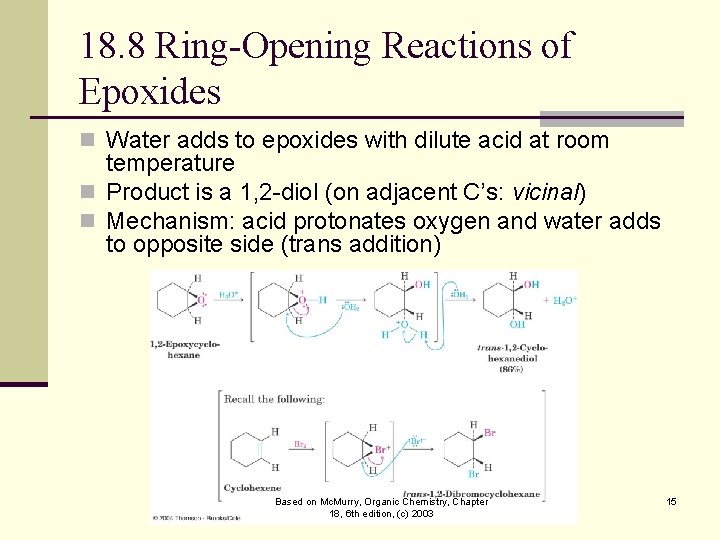
18. 8 Ring-Opening Reactions of Epoxides n Water adds to epoxides with dilute acid at room temperature n Product is a 1, 2 -diol (on adjacent C’s: vicinal) n Mechanism: acid protonates oxygen and water adds to opposite side (trans addition) Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 15
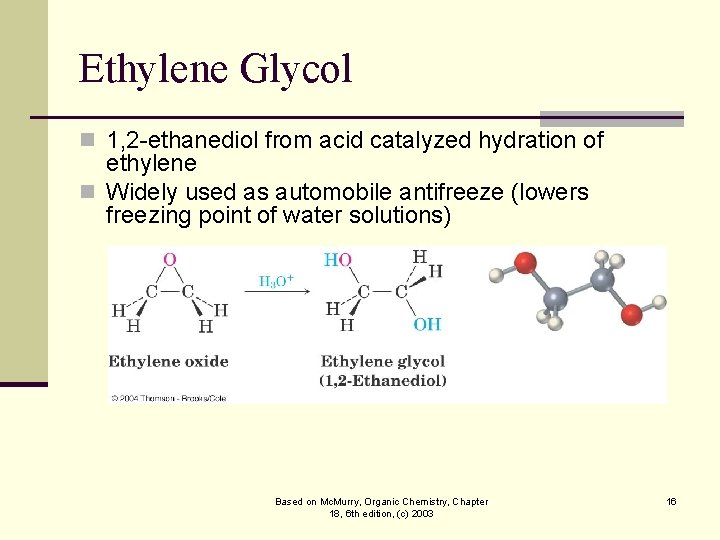
Ethylene Glycol n 1, 2 -ethanediol from acid catalyzed hydration of ethylene n Widely used as automobile antifreeze (lowers freezing point of water solutions) Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 16
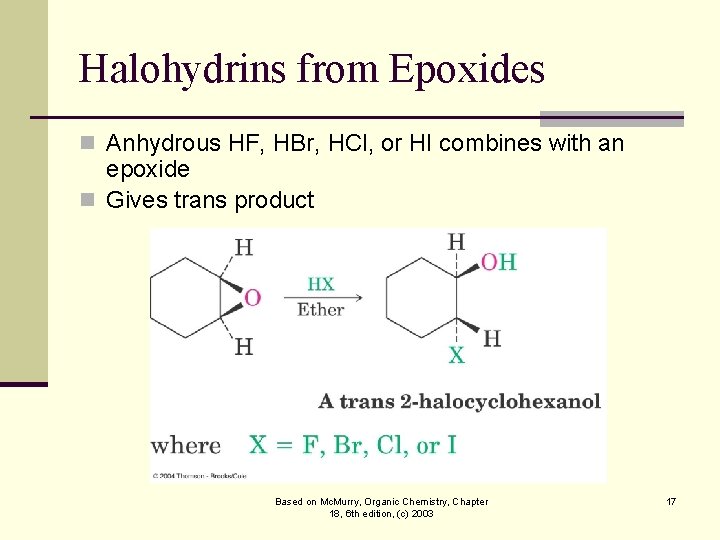
Halohydrins from Epoxides n Anhydrous HF, HBr, HCl, or HI combines with an epoxide n Gives trans product Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 17
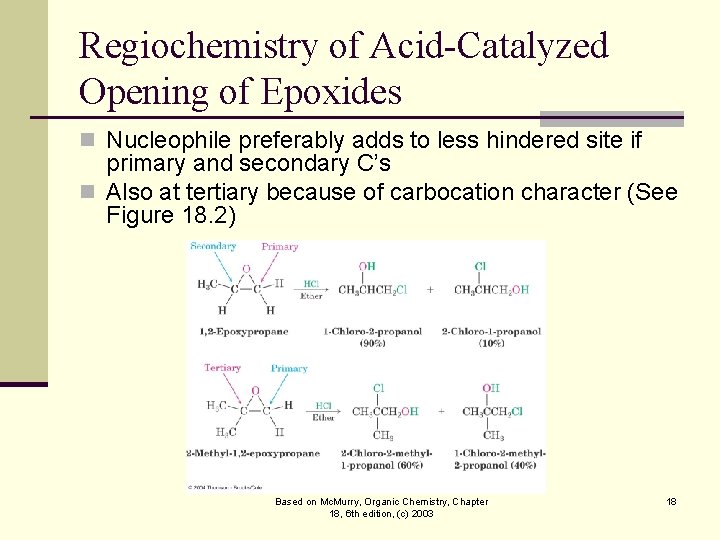
Regiochemistry of Acid-Catalyzed Opening of Epoxides n Nucleophile preferably adds to less hindered site if primary and secondary C’s n Also at tertiary because of carbocation character (See Figure 18. 2) Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 18
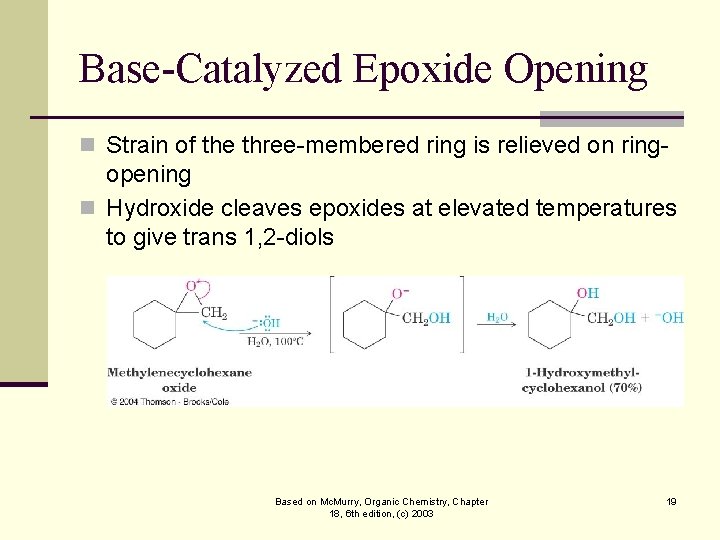
Base-Catalyzed Epoxide Opening n Strain of the three-membered ring is relieved on ring- opening n Hydroxide cleaves epoxides at elevated temperatures to give trans 1, 2 -diols Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 19

Addition of Grignards to Ethylene Oxide n Adds –CH 2 OH to the Grignard reagent’s hydrocarbon chain n Acyclic and other larger ring ethers do not react Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 20

18. 9 Crown Ethers n Large rings consisting repeating (-OCH 2 -) or similar units n Named as x-crown-y n x is the total number of atoms in the ring n y is the number of oxygen atoms n 18 -crown-6 ether: 18 -membered ring containing 6 oxygens atoms n Central cavity is electronegative and attracts cations Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 21

Uses of Crown Ethers n Complexes between crown ethers and ionic salts are soluble in nonpolar organic solvents n Creates reagents that are free of water that have useful properties n Inorganic salts dissolve in organic solvents leaving the anion unassociated, enhancing reactivity Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 22

18. 10 Thiols and Sulfides n Thiols (RSH), are sulfur analogs of alcohols n n Named with the suffix -thiol SH group is called “mercapto group” (“capturer of mercury”) Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 23

Sulfides n Sulfides (RSR ), are sulfur analogs of ethers n Named by rules used for ethers, with sulfide in place of ether for simple compounds and alkylthio in place of alkoxy Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 24

Thiols: Formation and Reaction n From alkyl halides by displacement with a sulfur nucleophile such as SH n The alkylthiol product can undergo further reaction with the alkyl halide to give a symmetrical sulfide, giving a poorer yield of the thiol Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 25

Using Thiourea to Form Alkylthiols n Thiols can undergo further reaction with the alkyl halide to give dialkyl sulfides n For a pure alkylthiol use thiourea (NH 2(C=S)NH 2) as the nucleophile n This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 26

Oxidation of Thiols to Disulfides n Reaction of an alkyl thiol (RSH) with bromine or iodine gives a disulfide (RSSR) n The thiol is oxidized in the process and the halogen is reduced Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 27

Sulfides n Thiolates (RS ) are formed by the reaction of a thiol with a base n Thiolates react with primary or secondary alkyl halide to give sulfides (RSR’) n Thiolates are excellent nucleophiles and react with many electrophiles Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 28

Sulfides as Nucleophiles n Sulfur compounds are more nucleophilic than their oxygen-compound analogs n 3 p electrons valence electrons (on S) are less tightly held than 2 p electrons (on O) n Sulfides react with primary alkyl halides (SN 2) to give trialkylsulfonium salts (R 3 S+) Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 29

Oxidation of Sulfides n Sulfides are easily oxidized with H 2 O 2 to the sulfoxide (R 2 SO) n Oxidation of a sulfoxide with a peroxyacid yields a sulfone (R 2 SO 2) n Dimethyl sulfoxide (DMSO) is often used as a polar aprotic solvent Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 30

18. 11 Spectroscopy of Ethers n Infrared: C–O single-bond stretching 1050 to 1150 cm 1 overlaps many other absorptions. n Proton NMR: H on a C next to ether O are shifted downfield to 3. 4 to 4. 5 n The 1 H NMR spectrum of dipropyl ether shows these signals at 3. 4 n In epoxides, these H’s absorb at 2. 5 to 3. 5 d in their 1 H NMR spectra n Carbon NMR: C’s in ethers exhibit a downfield shift to 50 to 80 Based on Mc. Murry, Organic Chemistry, Chapter 18, 6 th edition, (c) 2003 31
- Slides: 31