18 e Rule Electronic structures of many organometallic



- Slides: 8

18 e Rule § Electronic structures of many organometallic compounds are based on a total valence electron count of 18 on the central metal atom. Sidgwick coined the term ‘inert gas rule’ for indication of stability of 18 e compounds. Electron Counting Methods 1) Neutral Ligand / Covalent Method: All ligands are treated as neutral and are categorized according to how many electrons they are considered to donate. 2) Donor Pair/ Ionic Method: Ligands are considered to donate electrons in pairs, resulting in the need to treat some ligands as neutral and others as charged. Electron Counts for [MXa. Lb]c+ § L: A neutral ligand, which can be a lone-pair donor, such as CO or NH 3, a π-bond donor, such as C 2 H 4, or a σ-bond donor such as H 2, which are all 2 e ligands on both models. § X: Ligands such as H, Cl, or Me, which are 1 e X ligands on the covalent model and 2 e X− ligands on the ionic model. 1

18 e Rule § N: Number of electrons in neutral M atom. § a and b are the numbers of ligands. § c is the net ionic charge. Covalent Model § Number of electrons on metal = N § Number of electrons in ligands = a + 2 b § Total electron count = (N) + (a + 2 b) − c Ionic Model § Oxidation state of metal ion = (c + a) § Number of electrons on metal = N − a − c § Number of electrons in ligands = 2 a + 2 b § Total electron count = (N − a − c) + (2 a + 2 b) = N + a + 2 b − c 2

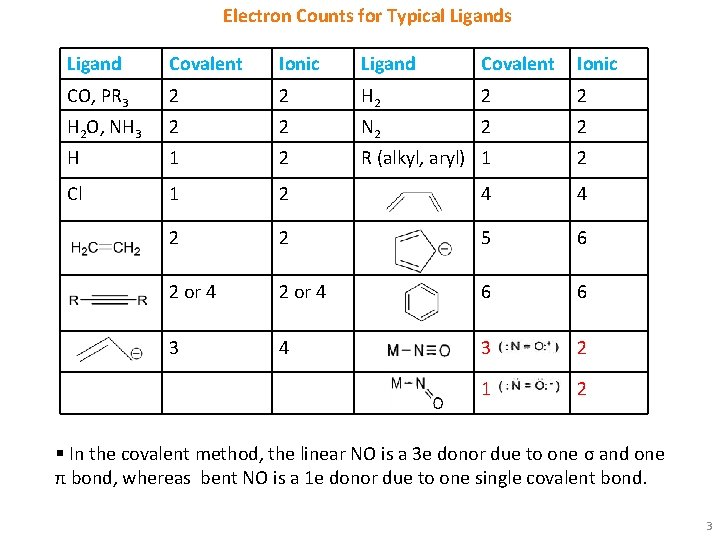
Electron Counts for Typical Ligands Ligand Covalent Ionic CO, PR 3 2 2 H 2 O, NH 3 2 2 N 2 2 2 H 1 2 R (alkyl, aryl) 1 2 Cl 1 2 4 4 2 2 5 6 2 or 4 6 6 3 4 3 2 1 2 § In the covalent method, the linear NO is a 3 e donor due to one σ and one π bond, whereas bent NO is a 1 e donor due to one single covalent bond. 3

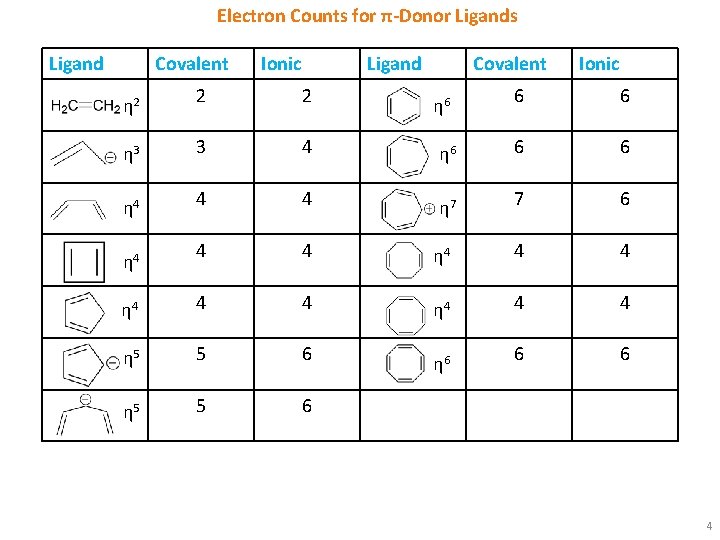
Electron Counts for π-Donor Ligands Ligand Covalent Ionic Ligand 2 2 η 3 3 4 η 4 4 Covalent Ionic 6 6 η 6 6 6 4 η 7 7 6 4 4 η 4 4 4 η 5 5 6 6 6 η 5 5 6 η 2 η 4 η 6 4

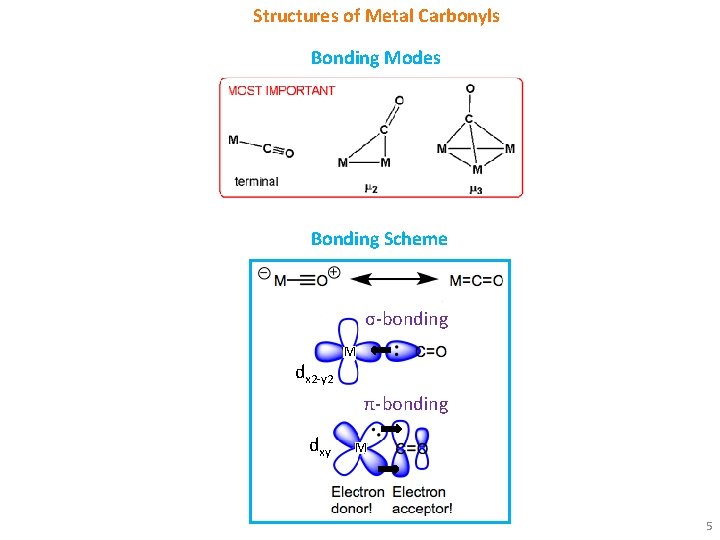
Structures of Metal Carbonyls Bonding Modes Bonding Scheme σ-bonding dx 2 -y 2 M π-bonding dxy M 5

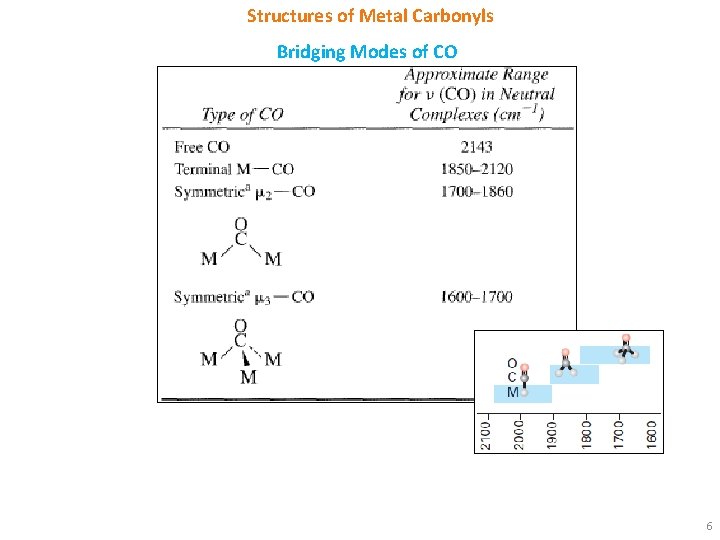
Structures of Metal Carbonyls Bridging Modes of CO 6

Structures of Metal Carbonyls § Both σ donation (which donates electron density from a bonding orbital on CO) and π acceptance (which places electron density in C-O antibonding orbitals) would be expected to weaken the C-O bond and to decrease the energy necessary to stretch that bond. § The more negative the charge on the organometallic species, the greater the tendency of the metal to donate electrons to the π* orbitals of CO and the lower the energy of the C-O stretching vibrations. § Order of C-O bond length: Ti(CO)62 - > V(CO)6 - > Cr(CO)6 > Mn(CO)6+ > Fe(CO)62+ 7

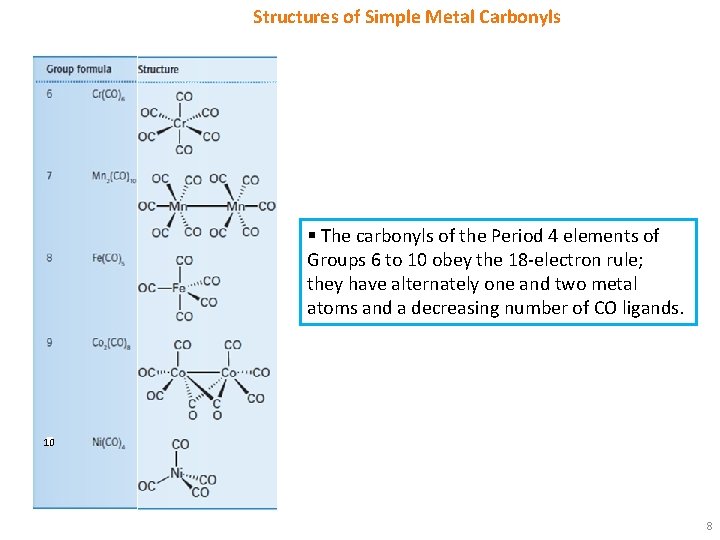
Structures of Simple Metal Carbonyls § The carbonyls of the Period 4 elements of Groups 6 to 10 obey the 18 -electron rule; they have alternately one and two metal atoms and a decreasing number of CO ligands. 10 8