18 5 Amides Tylenol an aspirin substitute contains

18. 5 Amides Tylenol, an aspirin substitute, contains acetaminophen. Acetaminophen is an amide; it acts to reduce fever and pain; however, it has little anti -inflammatory effect. Learning Goal Draw the amide product from amidation, and give the IUPAC and common names. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
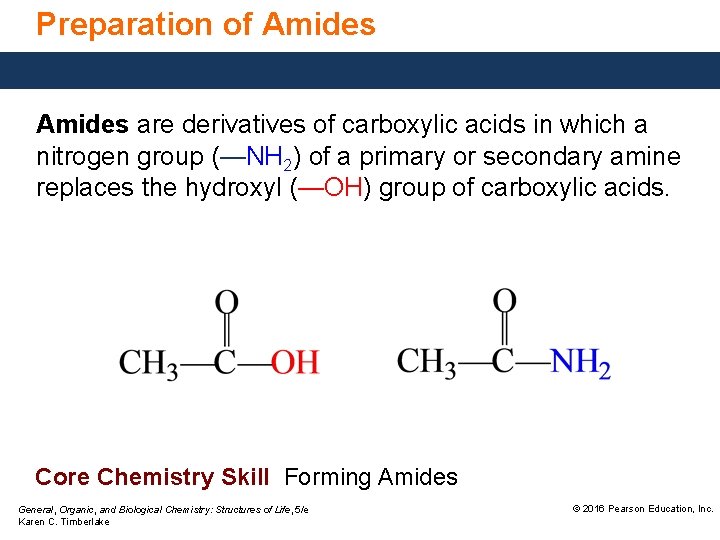
Preparation of Amides are derivatives of carboxylic acids in which a nitrogen group (—NH 2) of a primary or secondary amine replaces the hydroxyl (—OH) group of carboxylic acids. Core Chemistry Skill Forming Amides General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
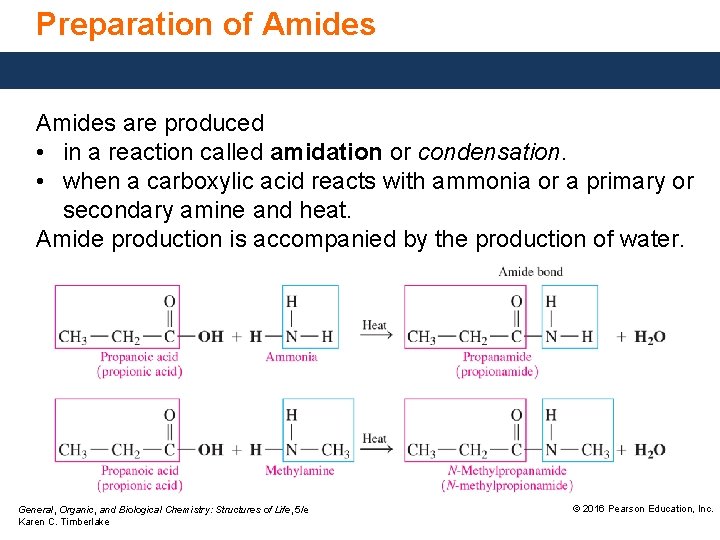
Preparation of Amides are produced • in a reaction called amidation or condensation. • when a carboxylic acid reacts with ammonia or a primary or secondary amine and heat. Amide production is accompanied by the production of water. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
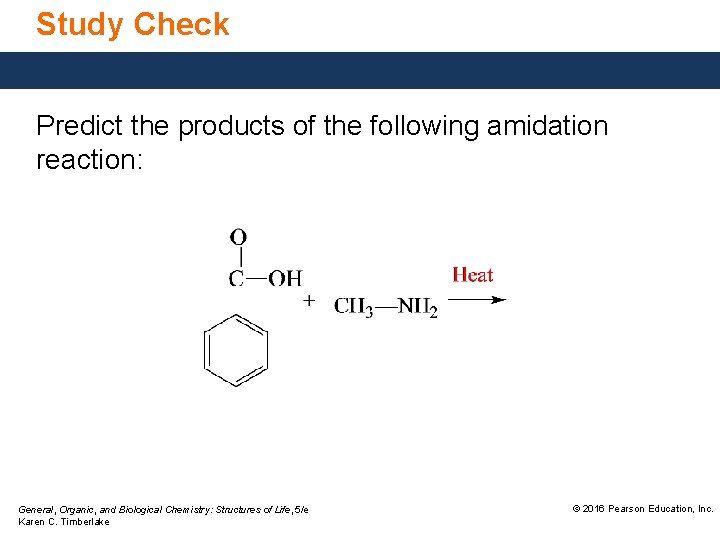
Study Check Predict the products of the following amidation reaction: General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
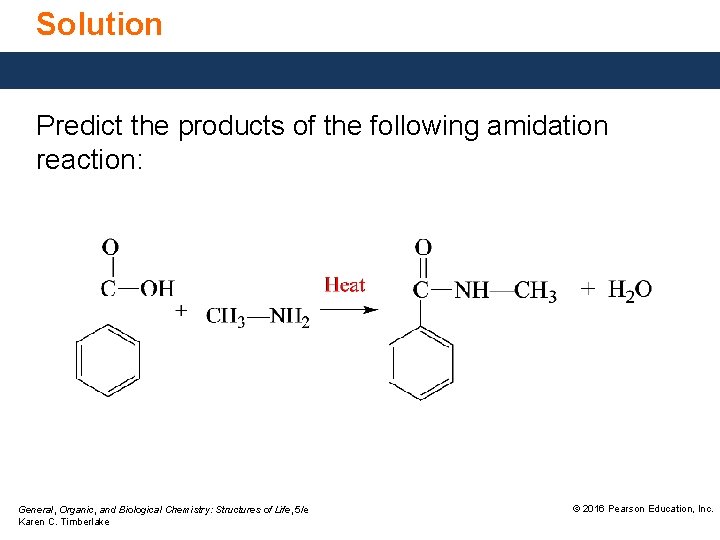
Solution Predict the products of the following amidation reaction: General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Naming Amides • In both the common and IUPAC names, amides are named by dropping the oic acid (IUPAC) or ic acid (common) from the carboxylic acid name and adding the suffix amide. • Alkyl groups attached to the nitrogen of an amide are named with the prefix N followed by the alkyl name. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Naming Amides General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
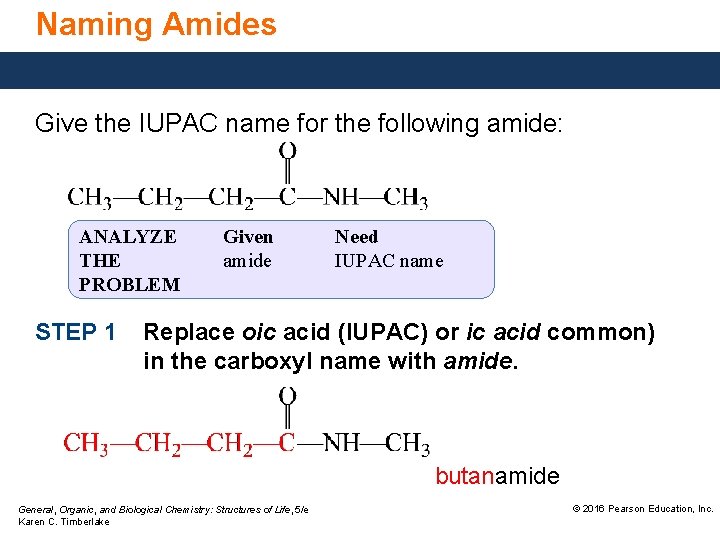
Naming Amides Give the IUPAC name for the following amide: ANALYZE THE PROBLEM STEP 1 Given amide Need IUPAC name Replace oic acid (IUPAC) or ic acid common) in the carboxyl name with amide. butanamide General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Naming Amides Give the IUPAC name for the following amide: STEP 2 Name each substituent on the N atom using the prefix N and the alkyl name. N-methylbutanamide General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
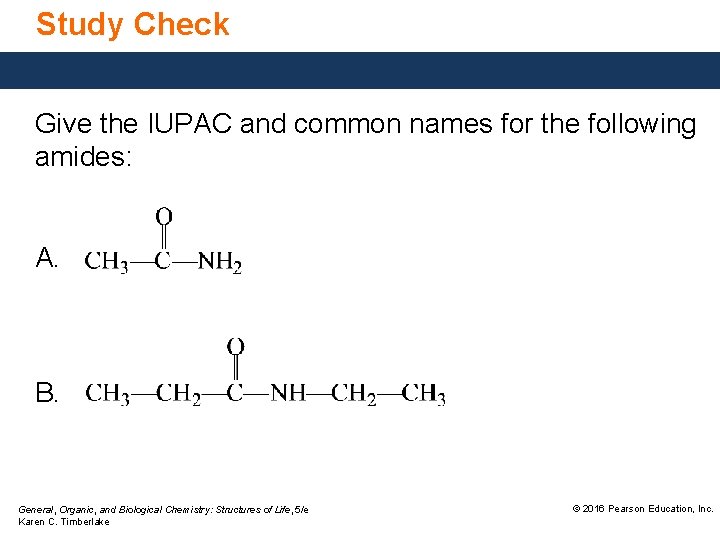
Study Check Give the IUPAC and common names for the following amides: A. B. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
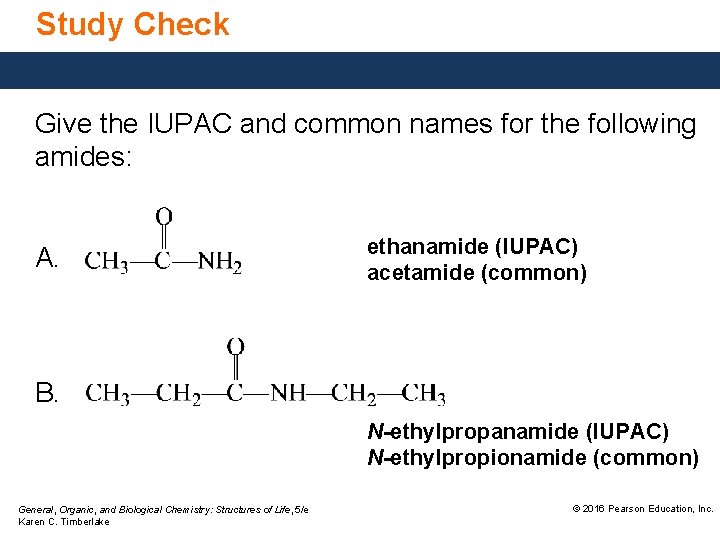
Study Check Give the IUPAC and common names for the following amides: A. ethanamide (IUPAC) acetamide (common) B. N-ethylpropanamide (IUPAC) N-ethylpropionamide (common) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Physical Properties of Amides • • • Amides do not have the properties of bases seen in amines. Only methanamide is a liquid at room temperature, while other amides are solids. Primary (1°) amides have the highest melting points because the —NH 2 group can form the most hydrogen bonds. Melting points of the secondary (2°) amides are lower because they form fewer hydrogen bonds. Tertiary (3°) amides cannot form hydrogen bonds; they have the lowest melting points (see Table 18. 3). General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
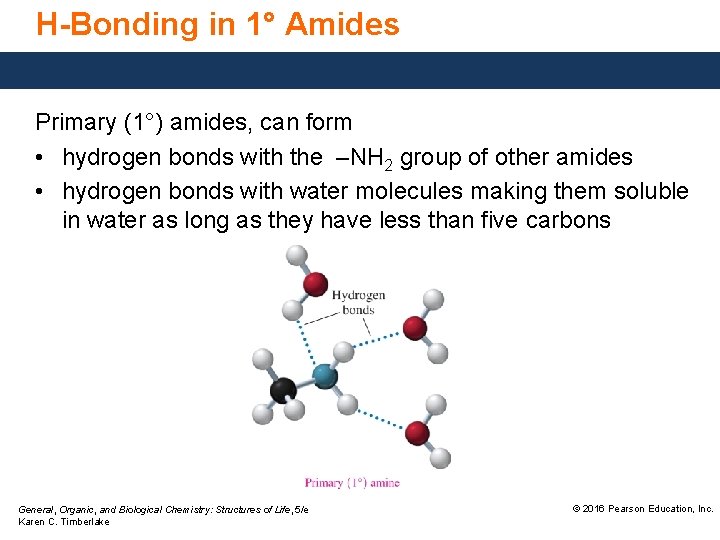
H-Bonding in 1° Amides Primary (1°) amides, can form • hydrogen bonds with the –NH 2 group of other amides • hydrogen bonds with water molecules making them soluble in water as long as they have less than five carbons General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

H-Bonding in 2° Amides Secondary (2°) amides • have lower melting points because they form fewer hydrogen bonds with other amides. • form hydrogen bonds with water molecules, making them soluble in water as long as they have fewer than five carbons. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
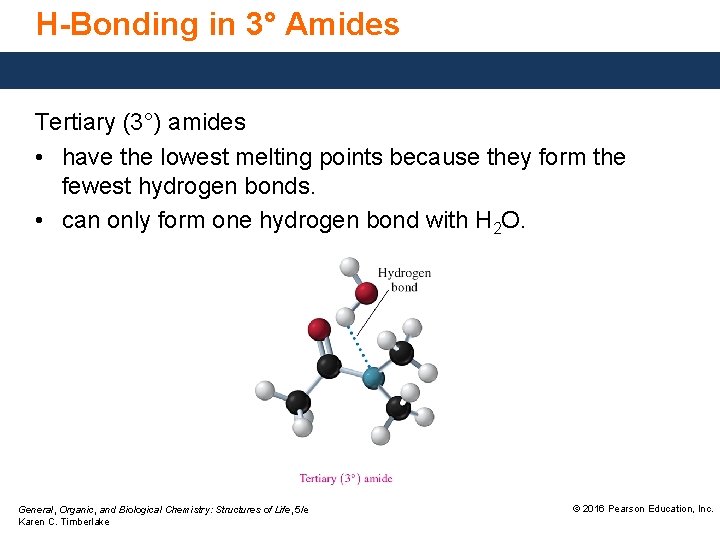
H-Bonding in 3° Amides Tertiary (3°) amides • have the lowest melting points because they form the fewest hydrogen bonds. • can only form one hydrogen bond with H 2 O. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
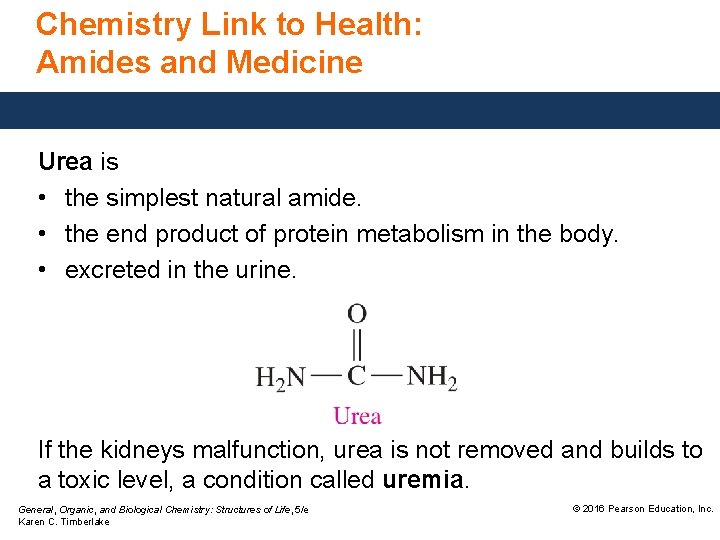
Chemistry Link to Health: Amides and Medicine Urea is • the simplest natural amide. • the end product of protein metabolism in the body. • excreted in the urine. If the kidneys malfunction, urea is not removed and builds to a toxic level, a condition called uremia. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
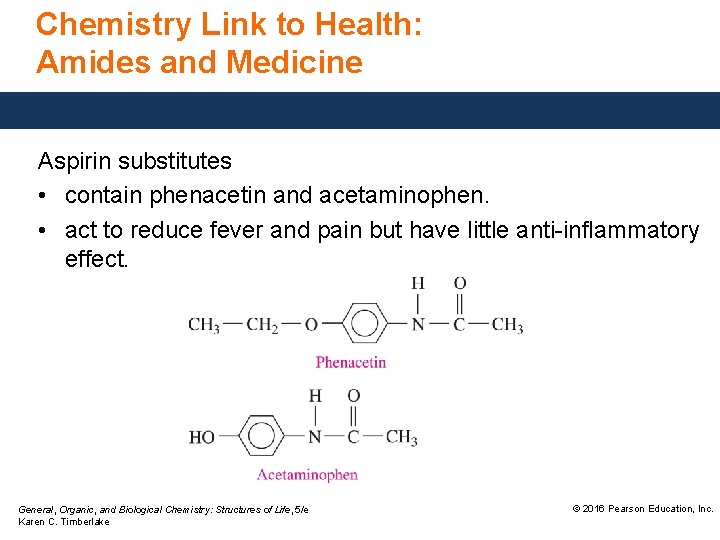
Chemistry Link to Health: Amides and Medicine Aspirin substitutes • contain phenacetin and acetaminophen. • act to reduce fever and pain but have little anti-inflammatory effect. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
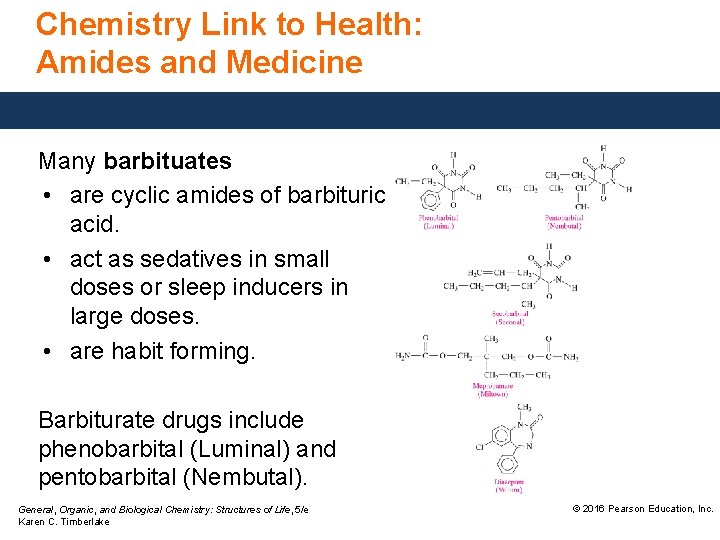
Chemistry Link to Health: Amides and Medicine Many barbituates • are cyclic amides of barbituric acid. • act as sedatives in small doses or sleep inducers in large doses. • are habit forming. Barbiturate drugs include phenobarbital (Luminal) and pentobarbital (Nembutal). General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 18