17 December 2021 Why do we need metals

17 December 2021 Why do we need metals? LO: To recognise the differences in common metals and how we extract them Starter: Name five things that are made from metal? What useful properties do metals have? Extension: Are all metals the same? Explain your answer with examples Connect Key Words
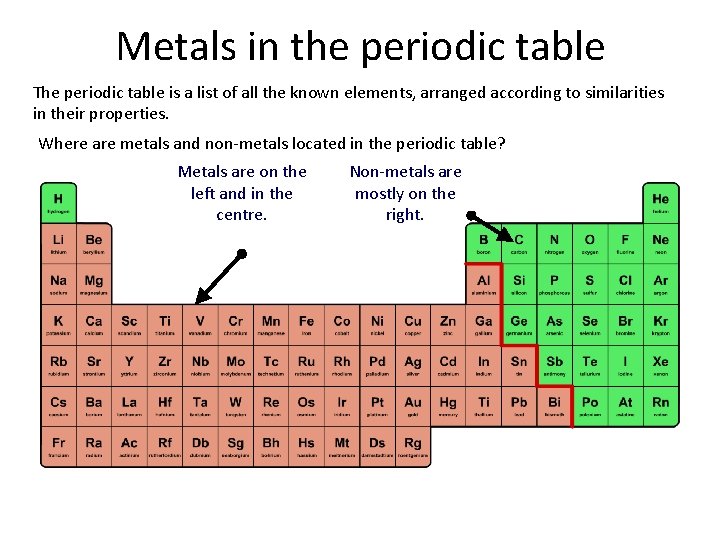
Metals in the periodic table The periodic table is a list of all the known elements, arranged according to similarities in their properties. Where are metals and non-metals located in the periodic table? Metals are on the left and in the centre. Non-metals are mostly on the right.

Typical properties of metals What are the properties of metals? l solid l strong l malleable (easily shaped) l conduct heat and electricity l dense l shiny l sonorous (make a ringing sound) l ductile (can be pulled into wires) l high melting and boiling points. Do all metals have all these properties?

Properties and uses For each of the properties come up with one use of metals. e. g. Strong, used for building • • • solid strong malleable (easily shaped) conduct heat and electricity dense shiny sonorous (make a ringing sound) ductile (can be pulled into wires) high melting and boiling points.

Where do metals come from? Some unreactive metals, like gold, silver and copper, are found in the Earth’s crust as pure substances. Metals that are found in a pure form are said to occur native. Most metals are actually found combined with other elements, as compounds in ores. These metals must be extracted from their ores before they can be made useful. Highly reactive metals, such as titanium, require complicated extraction which can increase the cost of the pure metal.

The reactivity series and extraction The reactivity of a metal determines how it is extracted. potassium sodium calcium magnesium Metals above carbon in the reactivity series must be extracted using electrolysis. increasing reactivity aluminium (carbon) zinc iron Metals less reactive than carbon can be extracted from their ores by reduction. lead (hydrogen) copper silver gold platinum Copper, silver, gold and platinum can occur native and do not need to be extracted. Sometimes copper needs to be extracted from an ore.

Extracting iron Metals that are less reactive than carbon can often be extracted from their ores by heating with carbon. Certain metals, such as iron, can only be reduced using carbon if they are heated to very high temperatures. Iron is extracted by this method in a blast furnace. Copper can also be extracted by heating copper-rich ores in a furnace (smelting).

Demo of the blast furnace

What is electrolysis? Electrolysis is a process that uses electricity to separate the elements in a compound. Electrolysis is expensive and so it is only used to extract reactive metals that cannot be extracted in other ways. Aluminium is a reactive metal that is found in the ore bauxite combined with oxygen as aluminium oxide (Al 2 O 3). Electrolysis breaks down the Al 2 O 3 into aluminium and oxygen. As the aluminium loses oxygen, reduction takes place. aluminium oxide aluminium + oxygen

Practical: electrolysis of copper sulfate • Carefully connect the circuit as show (6 V max) • Watch the negative electrode carefully. Discuss what is happening with your partner
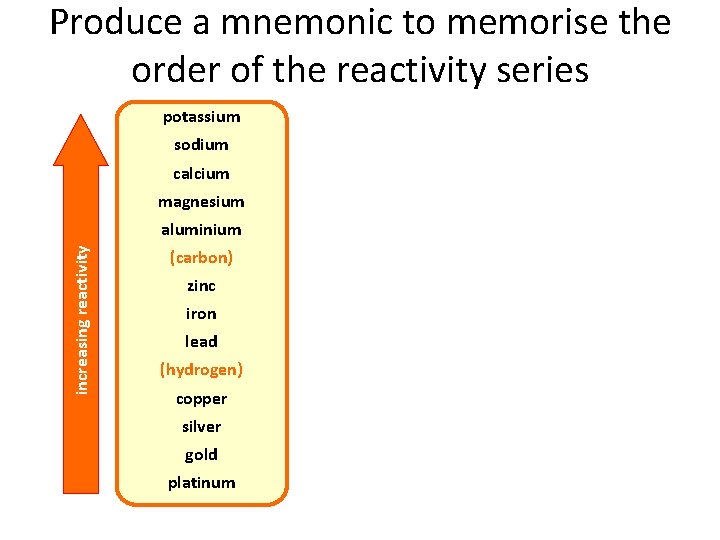
Produce a mnemonic to memorise the order of the reactivity series potassium sodium calcium magnesium increasing reactivity aluminium (carbon) zinc iron lead (hydrogen) copper silver gold platinum
- Slides: 11