17 8 Acetal Formation Some reactions of aldehydes








- Slides: 34

17. 8 Acetal Formation

Some reactions of aldehydes and ketones progress beyond the nucleophilic addition stage Acetal formation Imine formation Enamine formation Compounds related to imines The Wittig reaction

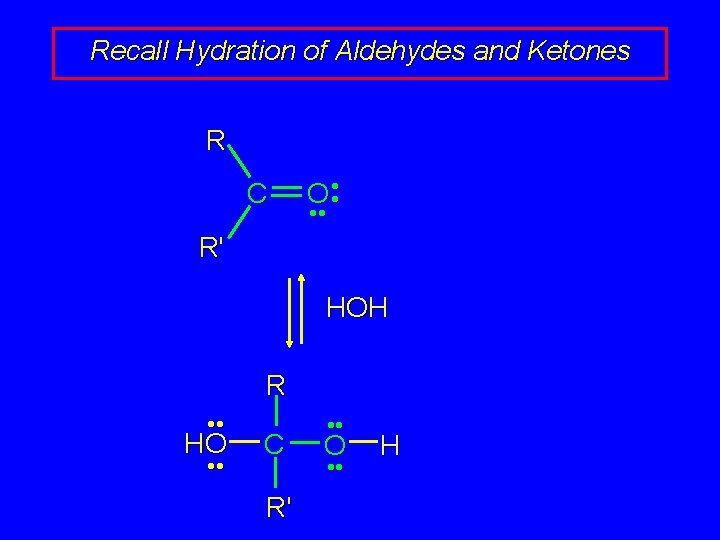
Recall Hydration of Aldehydes and Ketones R C O • • R' HOH R • • HO • • C R' • • O • • H

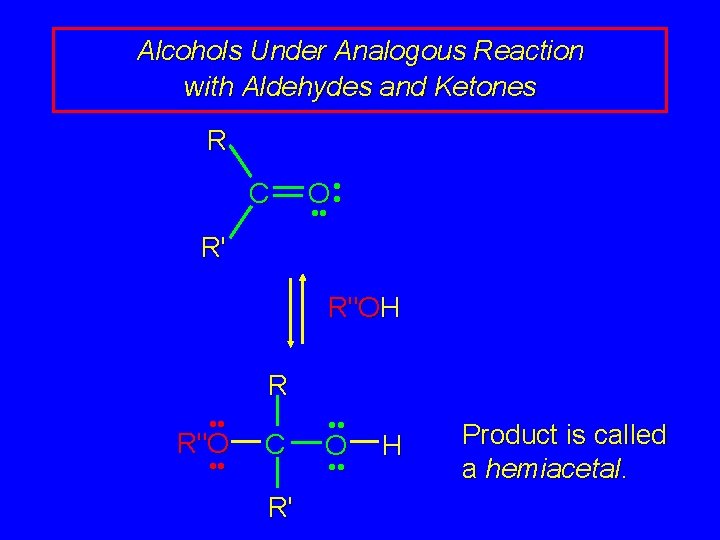
Alcohols Under Analogous Reaction with Aldehydes and Ketones R C O • • R' R"OH R • • R"O • • C R' • • O • • H Product is called a hemiacetal.

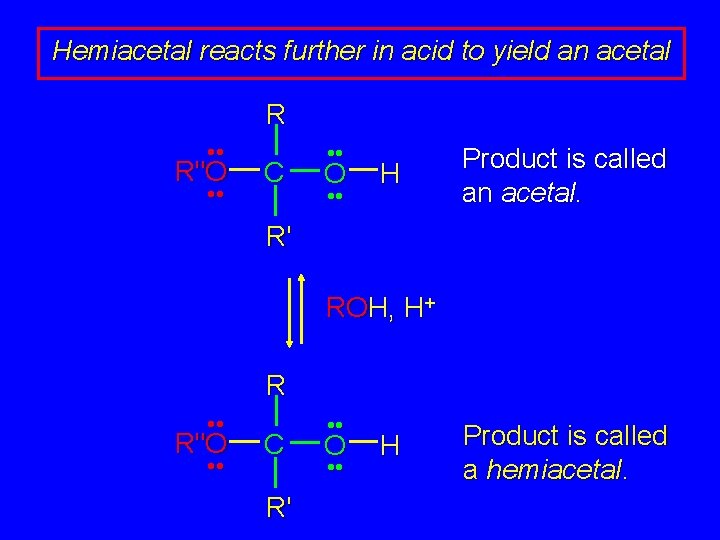
Hemiacetal reacts further in acid to yield an acetal R • • R"O • • C • • O • • H Product is called an acetal. R' ROH, H+ R • • R"O • • C R' • • O • • H Product is called a hemiacetal.

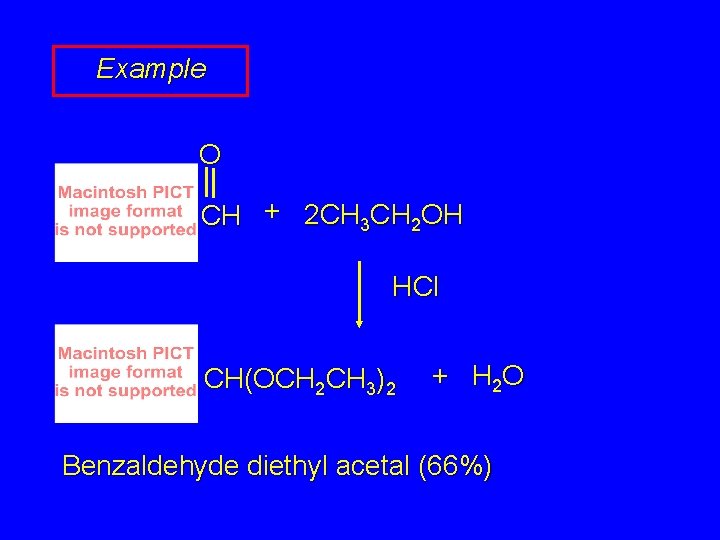
Example O CH + 2 CH 3 CH 2 OH HCl CH(OCH 2 CH 3)2 + H 2 O Benzaldehyde diethyl acetal (66%)

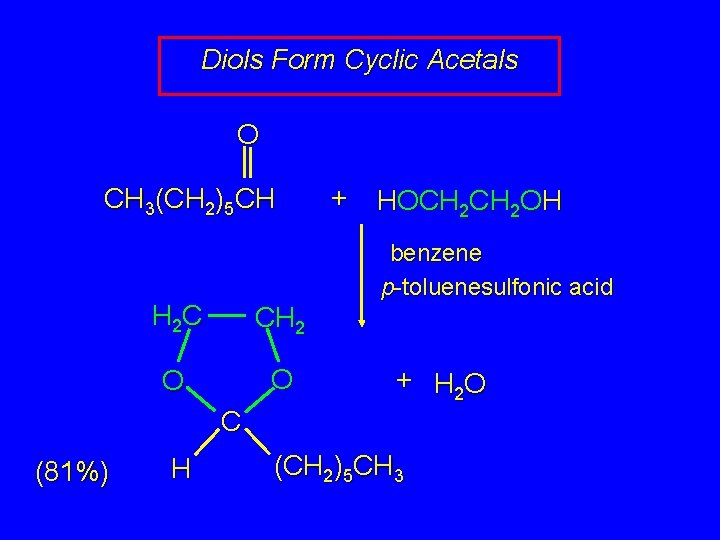
Diols Form Cyclic Acetals O CH 3(CH 2)5 CH + HOCH 2 OH benzene p-toluenesulfonic acid H 2 C CH 2 O O + H 2 O C (81%) H (CH 2)5 CH 3

In general: Position of equilibrium is usually unfavorable for acetal formation from ketones. Important exception: Cyclic acetals can be prepared from ketones.

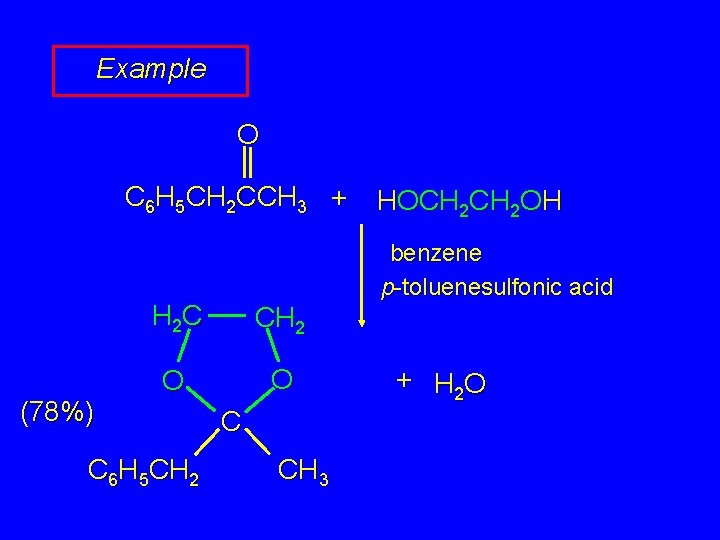
Example O C 6 H 5 CH 2 CCH 3 + (78%) benzene p-toluenesulfonic acid H 2 C CH 2 O O C 6 H 5 CH 2 HOCH 2 OH C CH 3 + H 2 O

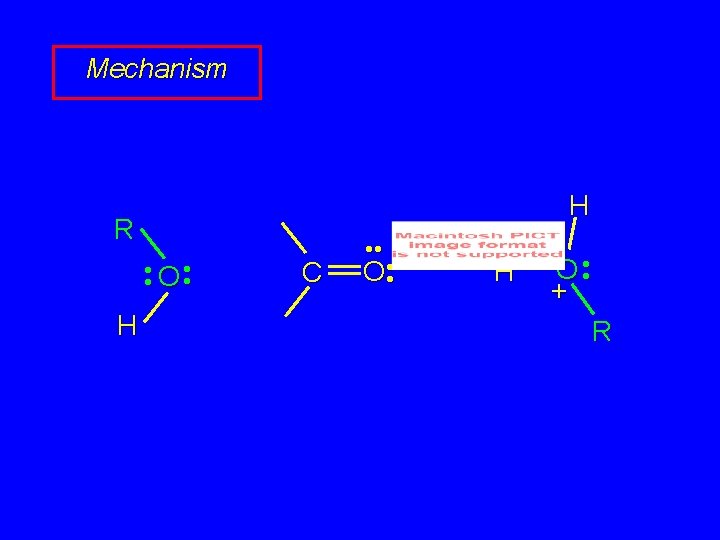
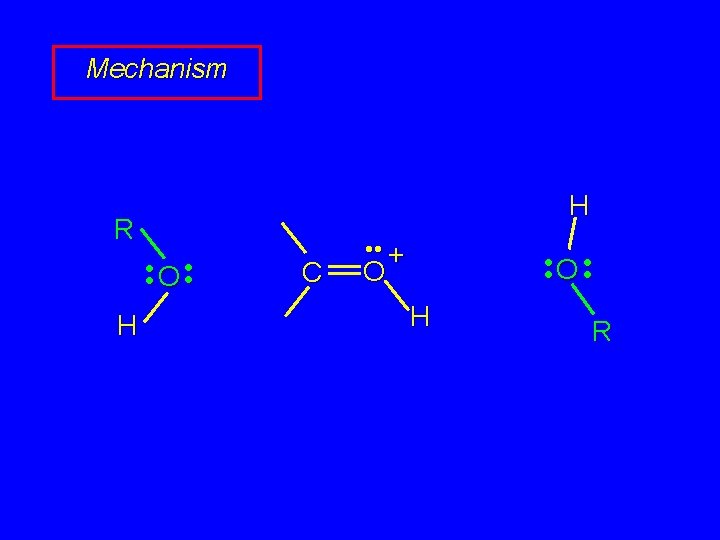
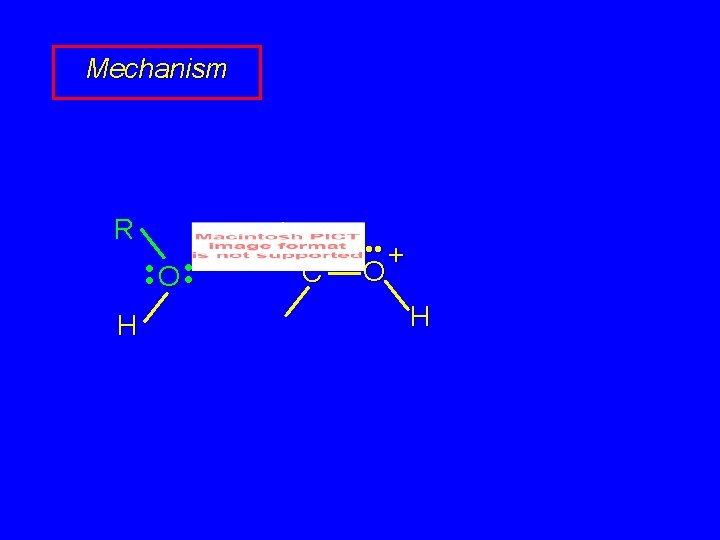
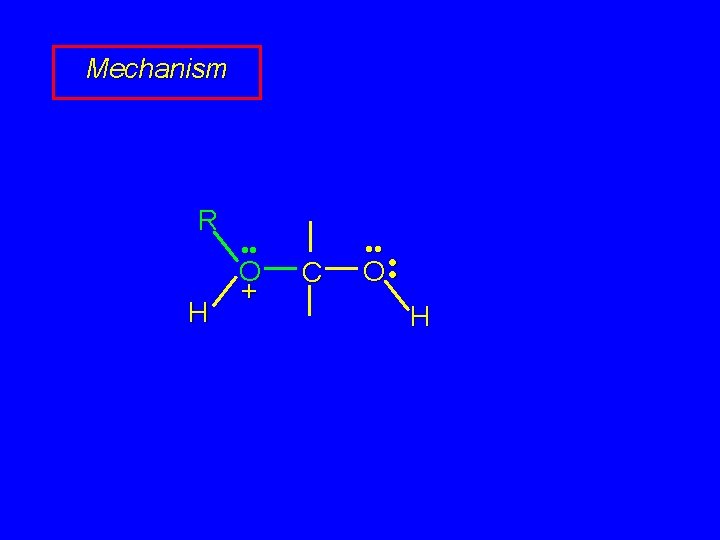
Mechanism of Acetal Formation First stage is analogous to hydration and leads to hemiacetal acid-catalyzed nucleophilic addition of alcohol to C=O



Mechanism R • • O • • H C • • + O H

Mechanism R H • • O + C • • O • • H



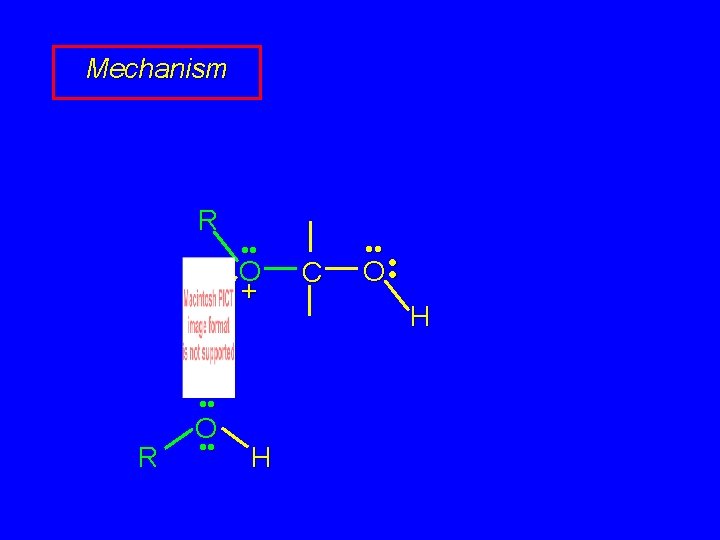
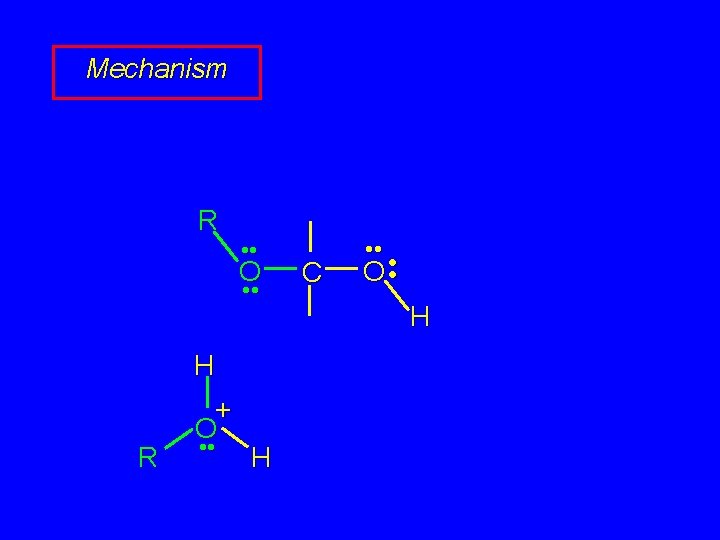
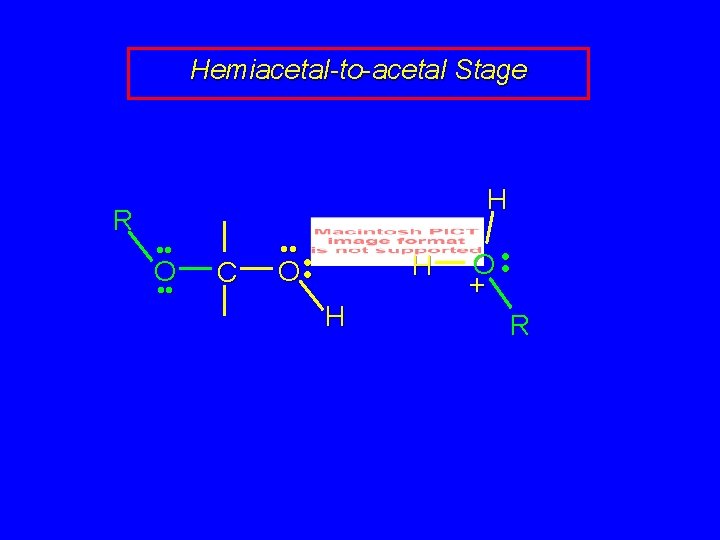
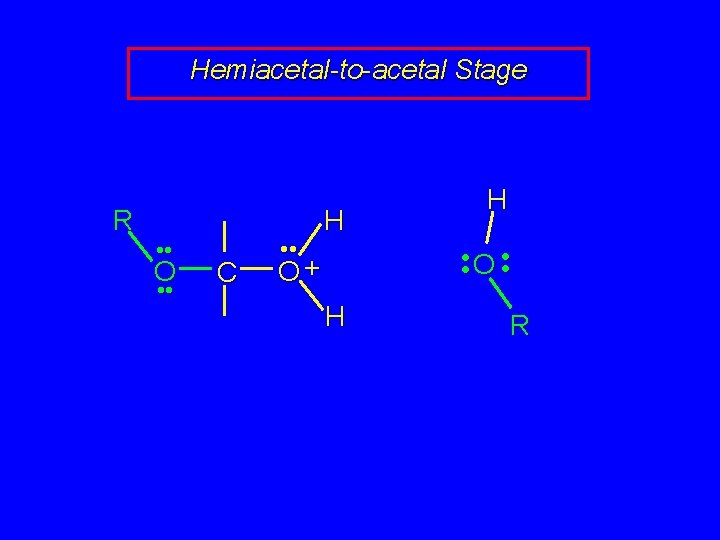
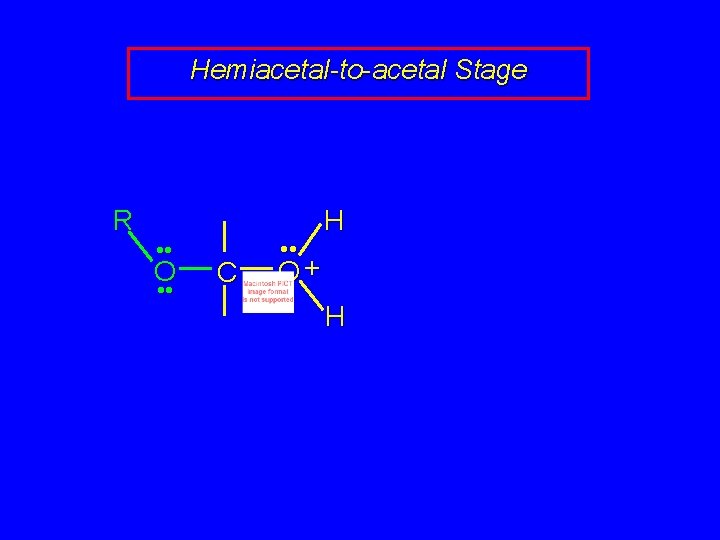
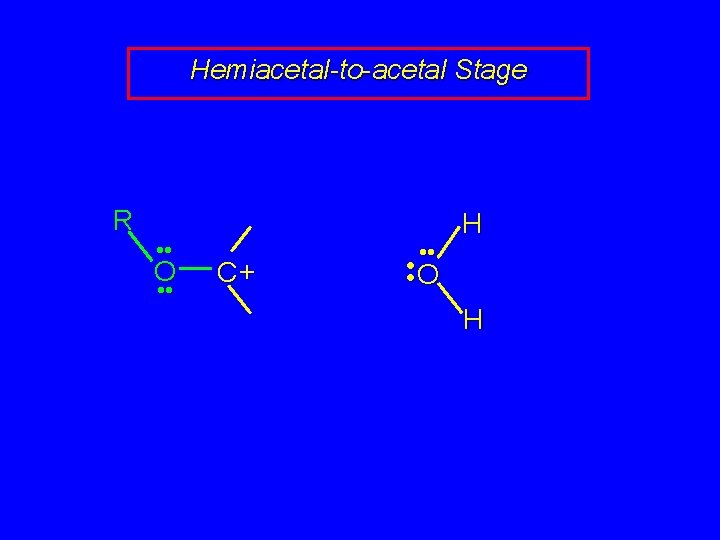
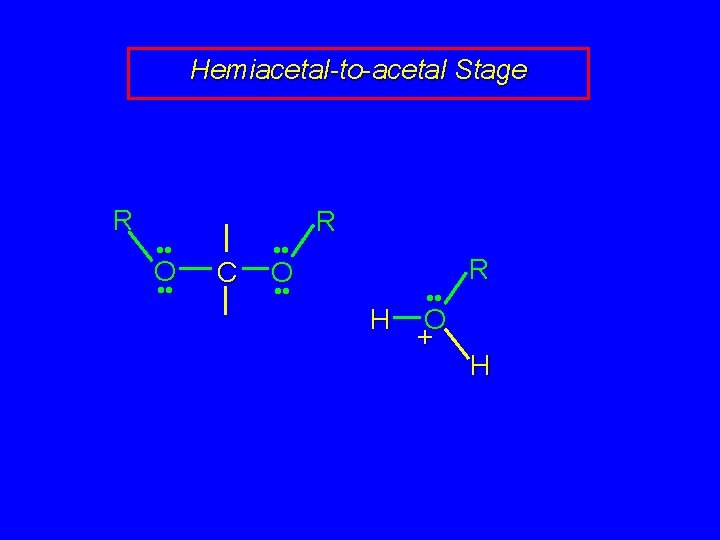
Mechanism of Acetal Formation Second stage is hemiacetal-to-acetal conversion involves carbocation chemistry

Hemiacetal-to-acetal Stage R H • • O • • C • • O • • H H O • • + R

Hemiacetal-to-acetal Stage R • • O • • C • • H H • • O • • O+ H R

Hemiacetal-to-acetal Stage R • • O • • C • • H O+ H

Hemiacetal-to-acetal Stage R • • O • • C+ • • H • • O H

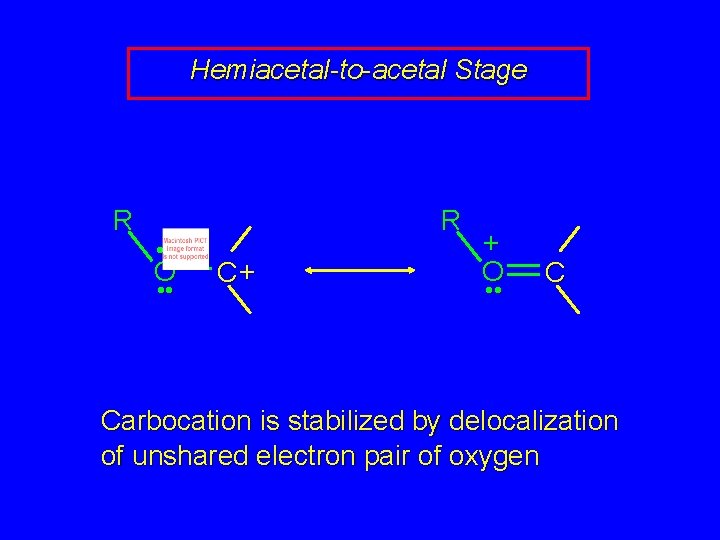
Hemiacetal-to-acetal Stage R • • O • • R C+ + O • • C Carbocation is stabilized by delocalization of unshared electron pair of oxygen

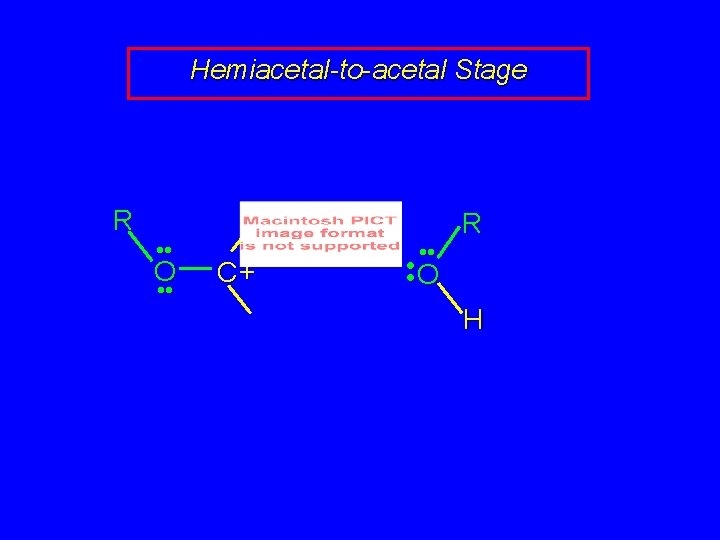
Hemiacetal-to-acetal Stage R • • O • • C+ • • R • • O H

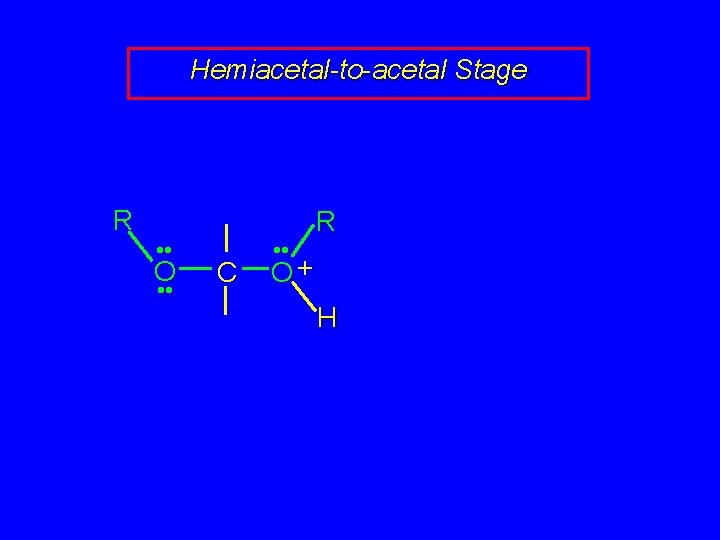
Hemiacetal-to-acetal Stage R • • O • • C • • R O+ H

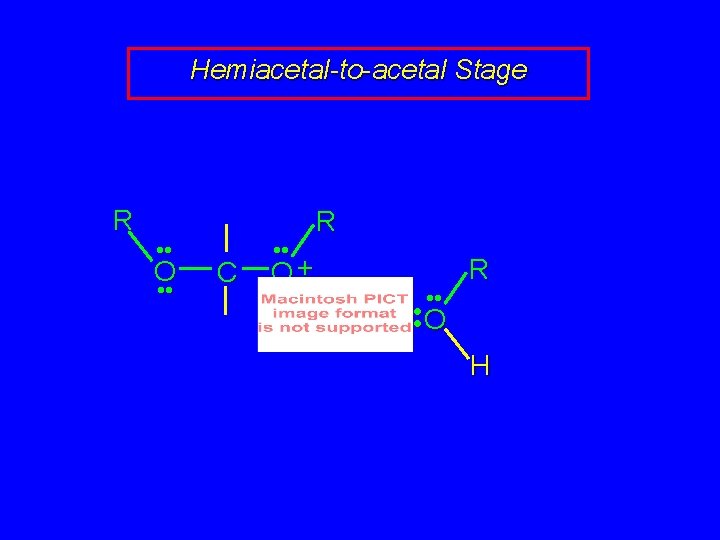
Hemiacetal-to-acetal Stage R • • O • • C • • R O+ H • • R • • O H

Hemiacetal-to-acetal Stage R • • O • • C • • R O • • H • • O + R H

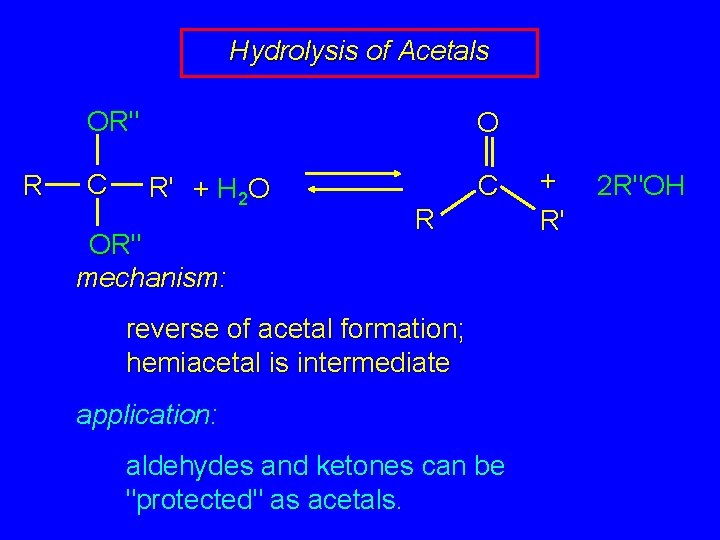
Hydrolysis of Acetals OR" R C O R' + H 2 O OR" mechanism: C R reverse of acetal formation; hemiacetal is intermediate application: aldehydes and ketones can be "protected" as acetals. + R' 2 R"OH

17. 9 Acetals as Protecting Groups

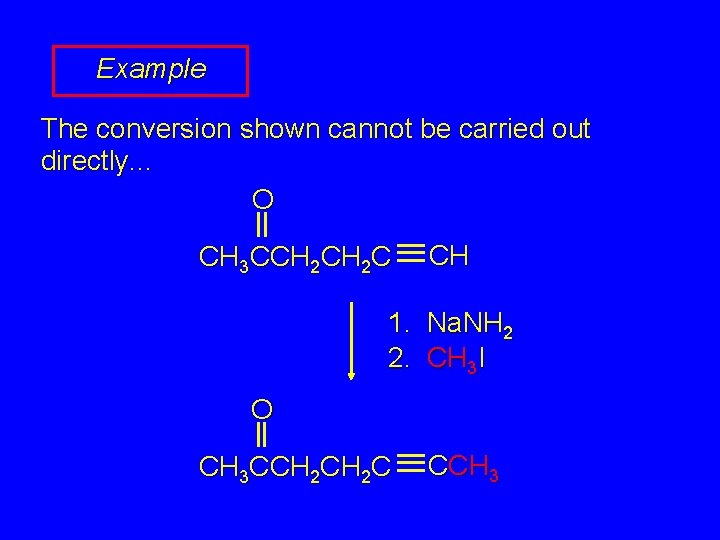
Example The conversion shown cannot be carried out directly. . . O CH 3 CCH 2 C CH 1. Na. NH 2 2. CH 3 I O CH 3 CCH 2 C CCH 3

because the carbonyl group and the carbanion are incompatible functional groups. O CH 3 CCH 2 C C: –

Strategy 1) protect C=O 2) alkylate 3) restore C=O

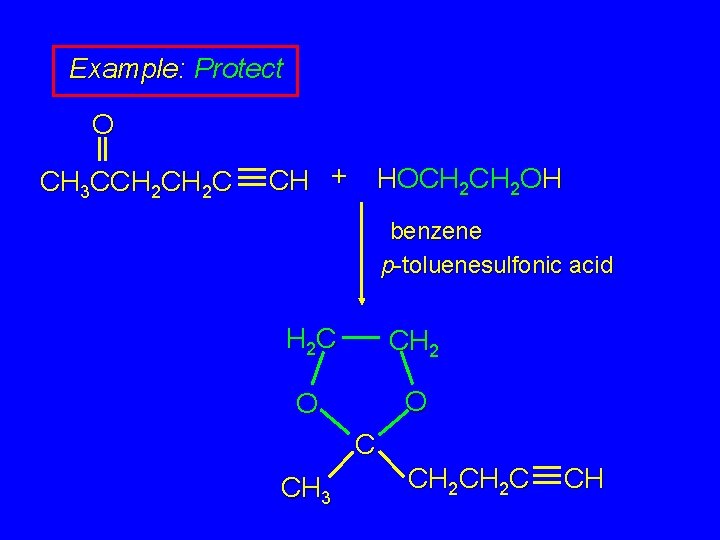
Example: Protect O CH 3 CCH 2 C CH + HOCH 2 OH benzene p-toluenesulfonic acid H 2 C CH 2 O O C CH 3 CH 2 C CH

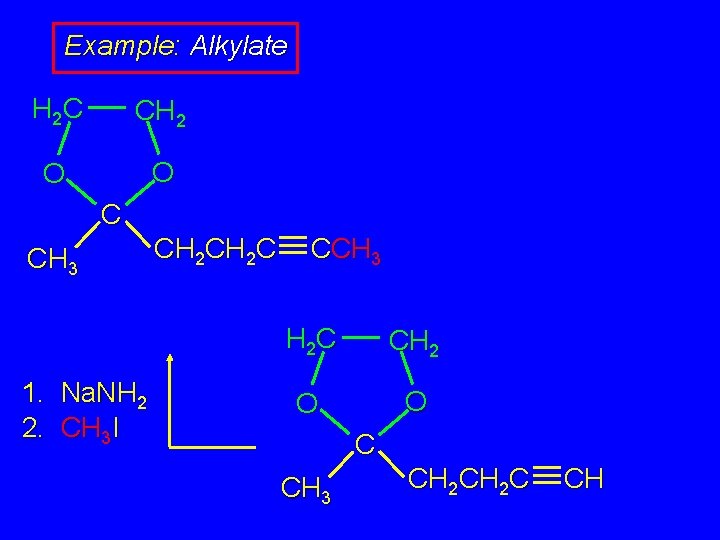
Example: Alkylate H 2 C CH 2 O O C CH 3 1. Na. NH 2 2. CH 3 I CH 2 C CCH 3 H 2 C CH 2 O O C CH 3 CH 2 C CH

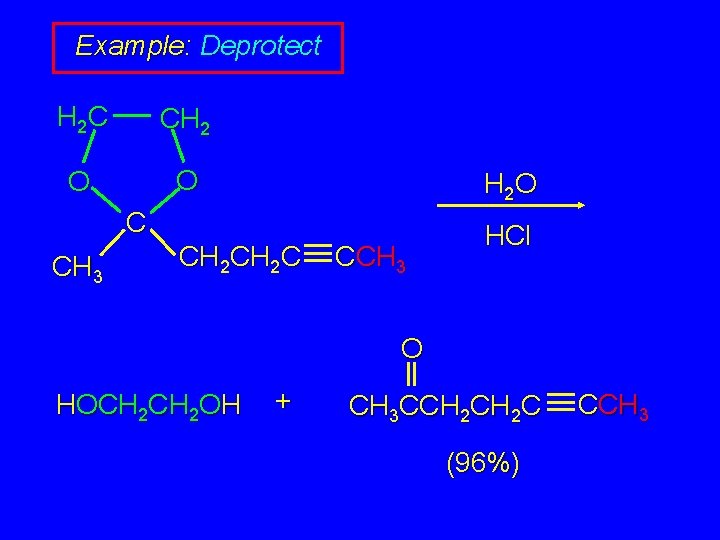
Example: Deprotect H 2 C CH 2 O O H 2 O C CH 3 CH 2 C CCH 3 HCl O HOCH 2 OH + CH 3 CCH 2 C (96%) CCH 3