17 3 Weak Acids Weak Bases Titration of

17. 3 Weak Acids Weak Bases Titration of Weak Acid with Strong Base Titration of Base Acid with Strong Acid Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Weak Acids Weak Bases Titration November 20
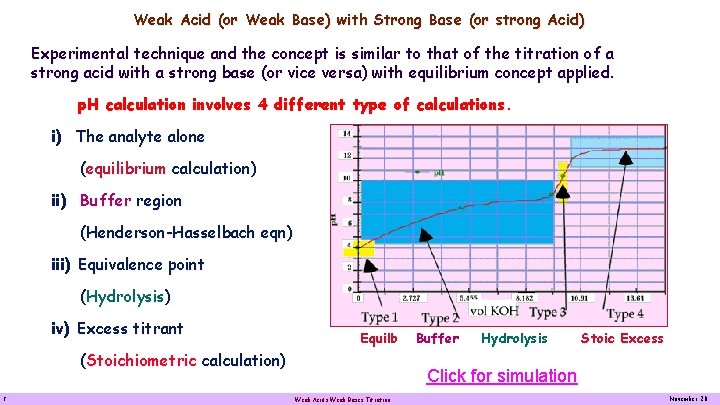
Weak Acid (or Weak Base) with Strong Base (or strong Acid) Experimental technique and the concept is similar to that of the titration of a strong acid with a strong base (or vice versa) with equilibrium concept applied. p. H calculation involves 4 different type of calculations i) The analyte alone (equilibrium calculation) ii) Buffer region (Henderson-Hasselbach eqn) iii) Equivalence point (Hydrolysis) iv) Excess titrant Equilb (Stoichiometric calculation) 2 Buffer Hydrolysis Stoic Excess Click for simulation Weak Acids Weak Bases Titration November 20
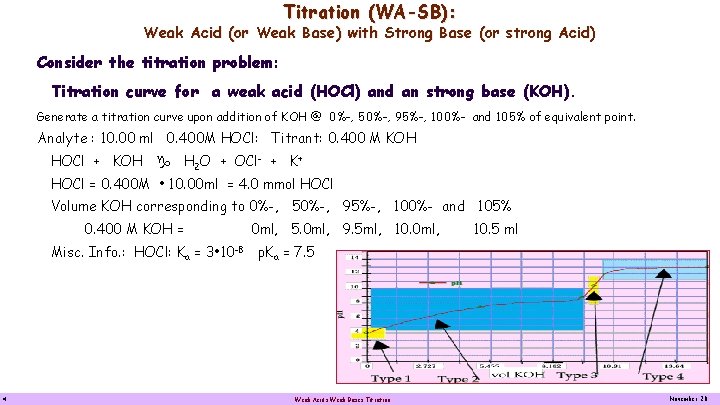
Titration (WA-SB): Weak Acid (or Weak Base) with Strong Base (or strong Acid) Consider the titration problem: Titration curve for a weak acid (HOCl) and an strong base (KOH). Generate a titration curve upon addition of KOH @ 0%-, 50%-, 95%-, 100%- and 105% of equivalent point. Analyte : 10. 00 ml 0. 400 M HOCl: Titrant: 0. 400 M KOH HOCl + KOH H 2 O + OCl- + K+ HOCl = 0. 400 M • 10. 00 ml = 4. 0 mmol HOCl Volume KOH corresponding to 0%-, 50%-, 95%-, 100%- and 105% 0. 400 M KOH = 0 ml, 5. 0 ml, 9. 5 ml, 10. 0 ml, 10. 5 ml Misc. Info. : HOCl: Ka = 3 • 10 -8 p. Ka = 7. 5 4 Weak Acids Weak Bases Titration November 20
Type i: Weak Acid with Strong Base 0 % Type 1: Calculation EQUILBRIUM 5 Weak Acids Weak Bases Titration November 20
Titration (4 ii): Weak Acid (or Weak Base) with Strong Base (or strong Acid) 50 % Type 2: Calculation BUFFER, Henderson- Hasselbalch Eqn 6 Weak Acids Weak Bases Titration November 20
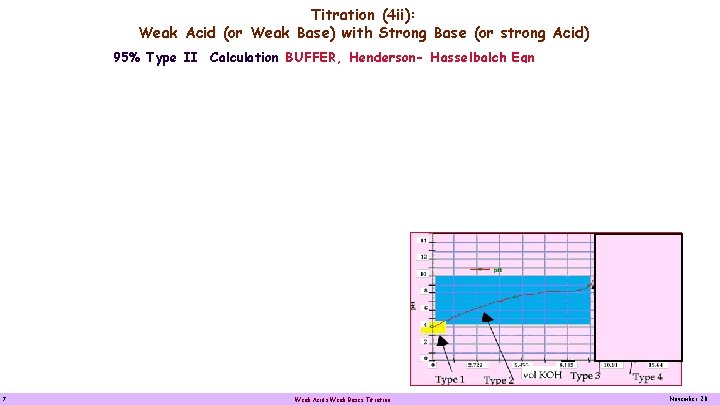
Titration (4 ii): Weak Acid (or Weak Base) with Strong Base (or strong Acid) 95% Type II Calculation BUFFER, Henderson- Hasselbalch Eqn 7 Weak Acids Weak Bases Titration November 20
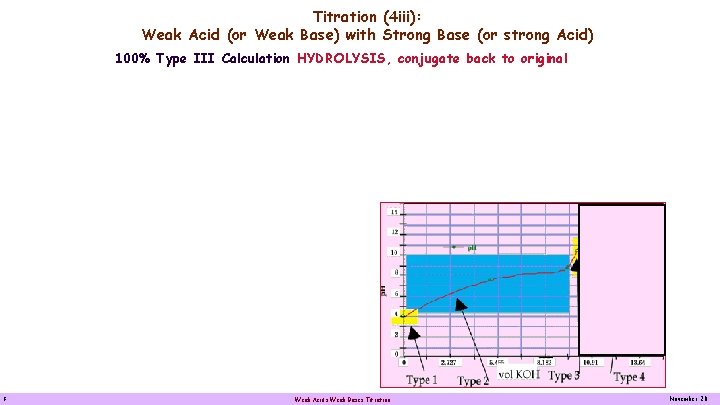
Titration (4 iii): Weak Acid (or Weak Base) with Strong Base (or strong Acid) 100% Type III Calculation HYDROLYSIS, conjugate back to original 8 Weak Acids Weak Bases Titration November 20
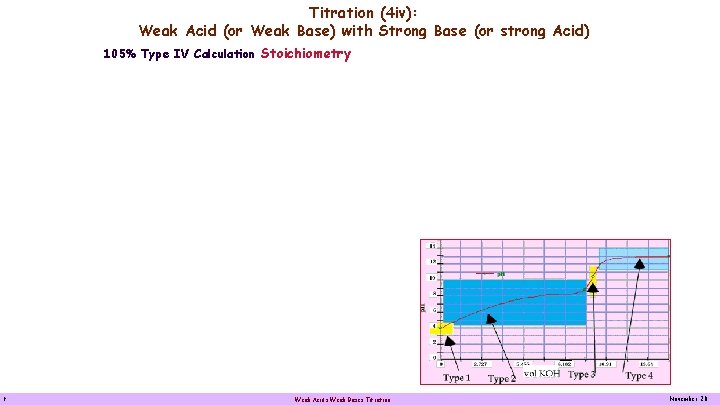
Titration (4 iv): Weak Acid (or Weak Base) with Strong Base (or strong Acid) 105% Type IV Calculation Stoichiometry 9 Weak Acids Weak Bases Titration November 20
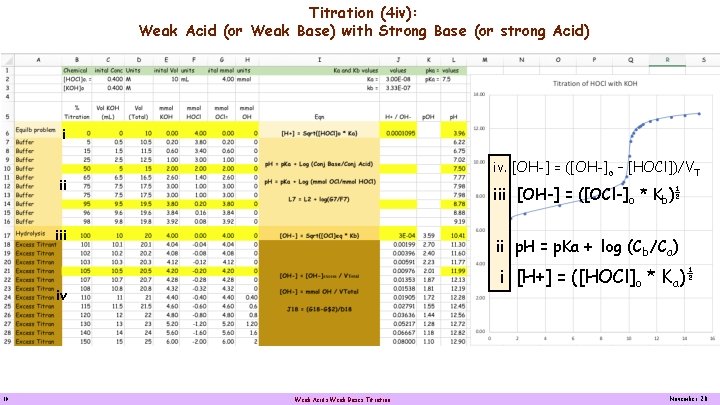
Titration (4 iv): Weak Acid (or Weak Base) with Strong Base (or strong Acid) i iv. [OH-] = ([OH-]o – [HOCl])/VT ii iii [OH-] = ([OCl-]o * Kb)½ iii ii p. H = p. Ka + log (Cb/Ca) i [H+] = ([HOCl]o * Ka)½ iv 10 Weak Acids Weak Bases Titration November 20

Titration (4 iv): Weak Acid (or Weak Base) with Strong Base (or strong Acid) [OH-] = ([OH-]o – [HOCl])/VT [OH-] = ([OCl-]o * Kb)½ p. H = p. Ka + log (Cb/Ca) [H+] = ([HOCl]o * Ka)½ 11 Weak Acids Weak Bases Titration November 20
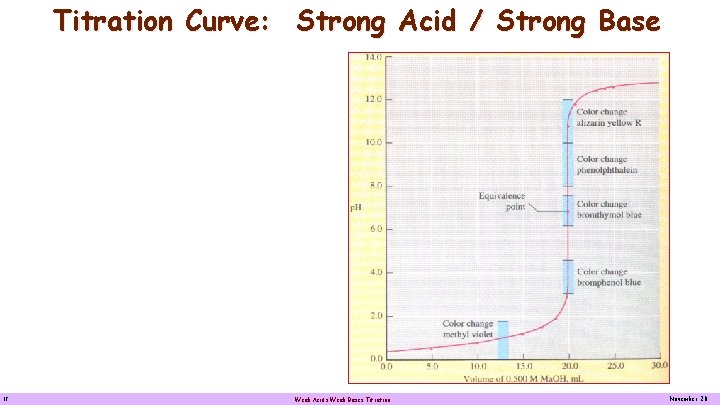
Titration Curve: Strong Acid / Strong Base Titration of 20. 0 m. L 0. 500 M of 0. 100 M HCl with. Titration 0. 500 M Na. OH with 0. 100 M Na. OH. A strong acid-strong base titration curve, showing how the p. H increases as 0. 5000 M Na. OH is added to 20. 00 m. L of 0. 5000 M HCl. The equivalence point p. H is 7. 00. The steep portion of the curve includes the transition intervals of bromothymol blue, phenolphthalein and bromophenol blue. 12 Weak Acids Weak Bases Titration November 20
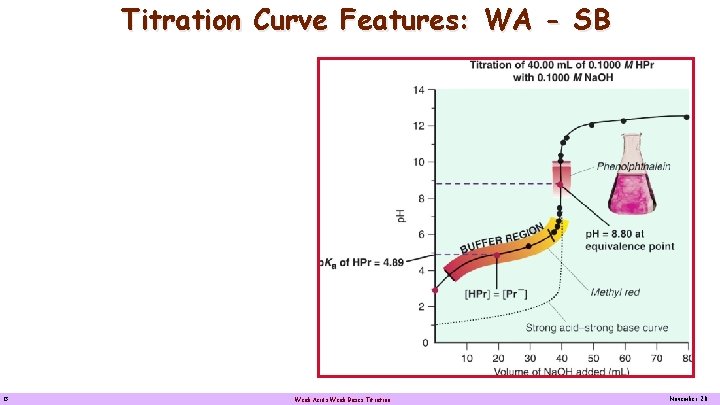
Titration Curve Features: WA - SB Titration of. 40. 00 m. L 0. 1000 M HCH 3 CH 2 OOO with 0. 1000 M Na. OH Weak acid - Strong base Titration curve for a weak acid by a strong base: 40. 00 m. L of 0. 1000 M CH 3 CH 2 OOOH by 0. 1000 M Na. OH. When exactly one-half the acid is neutralized, [CH 3 CH 2 COOH] = [CH 3 CH 2 COO-] and p. H = p. Ka = 8. 80 The equivalent point is above 7. 00 because the solution contains the weak base CH 3 CH 2 COO-. Phenolphthalein is a suitable indicator for this titration but Methyl red is not because its color changes over a large volume range. 13 Weak Acids Weak Bases Titration November 20
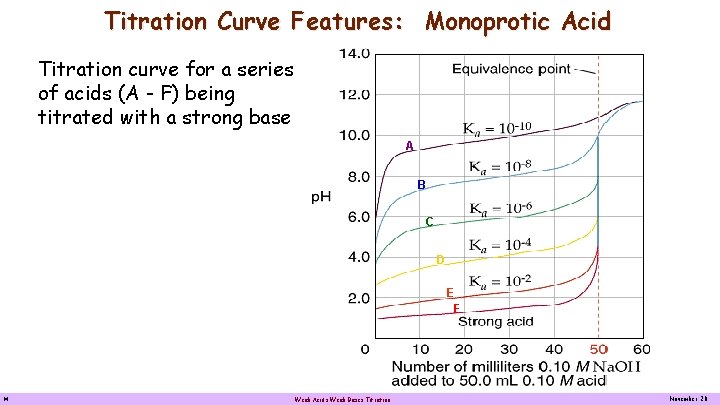
Titration Curve Features: Monoprotic Acid Titration curve for a series of acids (A - F) being titrated with a strong base A Acid F is the strongest acid, Acid E is the next strongest acid followed by acid D, acid C, Acid B and acid A. Acid A is the weakest among the weak acid. The Ka’s of each acid is determined by reading the p. H half way to the equivalent volume for each acid. 14 B C D E F Weak Acids Weak Bases Titration November 20
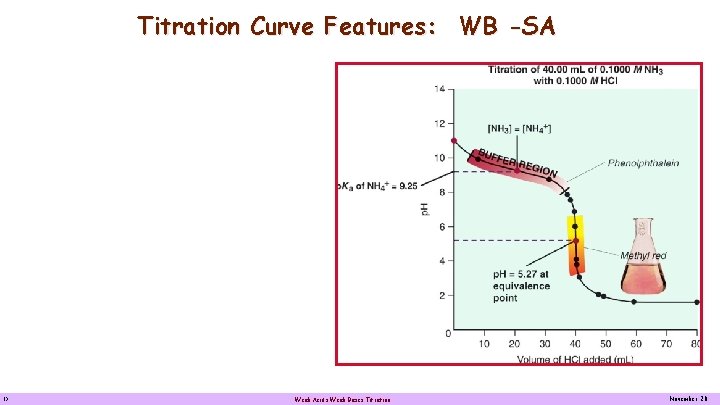
Titration Curve Features: WB -SA Titration of 0. 1000 M NH 3 with 0. 1000 M HCl Weak base - Strong acid A weak base-strong acid titration curve, showing how the p. H decreases as 0. 1000 M HCl is added to 40. 00 m. L of 0. 1000 M NH 3. When exactly one-half the base is neutralized, [NH 3] = [NH 4+] and p. OH = p. Kb of NH 3 (4. 76) or the p. H = 14 - p. Kb of NH 3 (9. 24). Note that this p. H value (9. 24) is actually the pka of NH 4+, the conjugate of NH 3. Note that the equivalent point is below 7. 00 because the solution contains the weak acid NH 4+. Methyl red is a suitable indicator for this titration but phenolphthalein is not because its color changes over a large volume range. 15 Weak Acids Weak Bases Titration November 20
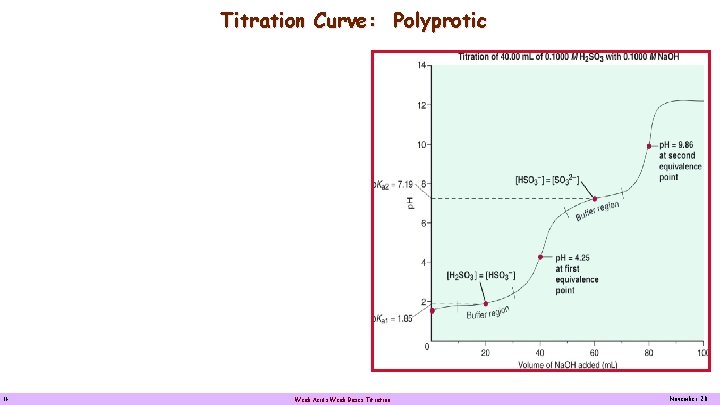
Titration Curve: Polyprotic Titration Curve of 0. 100 M H 2 SO 3 with 0. 100 M Na. OH Curve for the titration of a weak polyprotic acid. Titrating 40. 00 m. L of 0. 1000 M H 2 SO 3 with 0. 1000 M Na. OH leads to a curve with two buffer regions and two equivalence points. Because the Ka values are separated by several orders of magnitude, in effect the titration curve looks like two weak acid-strong base curves attached. The p. H of the first equivalence point is below 7 because the solution contains HSO 3 -, which is a stronger acid than it is a base. Ka of HSO 3 - = 6. 5 • 10 -8; Kb of HSO 3 - = 7. 1 • 10 -13 16 Weak Acids Weak Bases Titration November 20
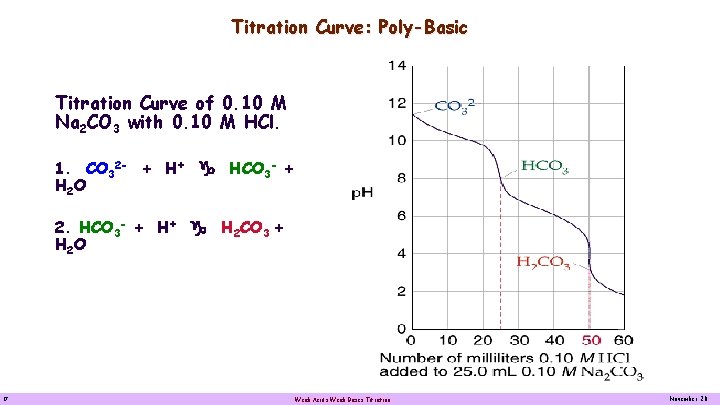
Titration Curve: Poly-Basic Titration Curve of 0. 10 M Na 2 CO 3 with 0. 10 M HCl. 1. CO 32 - + H+ g HCO 3 - + H 2 O 2. HCO 3 - + H+ g H 2 CO 3 + H 2 O 17 Weak Acids Weak Bases Titration November 20
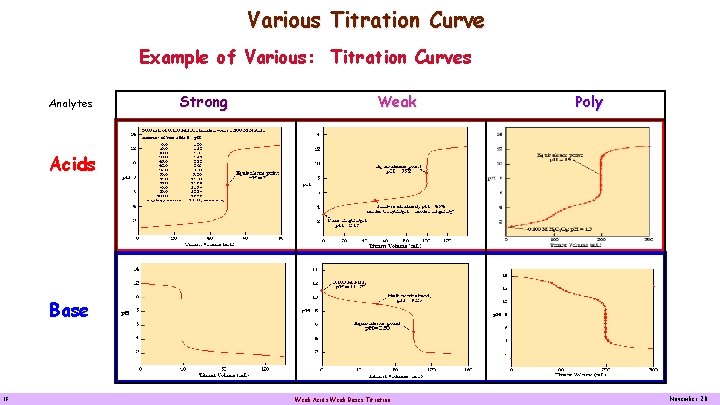
Various Titration Curve Example of Various: Titration Curves Analytes Strong Weak Poly Acids Base 18 Weak Acids Weak Bases Titration November 20

Summary There are two main type of titration problems. The strategy to solve them are: 1) SA-SB: Strong acid being titrated with a strong base (or vice versa). This is a stoichiometry type of problem. The p. H at the equivalent point = 7. 0. 2 a) Weak acid being titrated with a strong base. In this type of problem, there are four sub-problems that must be solved. a) At 0% titrant, the problem is an equilibrium type. The Ka of the weak acid will determine the extent of ionization and therefore the p. H. b) At 1 -99% titrant, the problem is a buffer type. Use the Henderson-Hasselbach Equation to solve for the p. H c) At equivalence point, this is now a hydrolysis problem. Recall that the conjugate base of the weak acid now reacts with water to produce OH-. The solution will be basic at the equivalent point. d) Pass the equivalence point, this is now a stoichiometry problem. The excess titrant base will dictate the p. H of the solution. 2 b) Weak base being titrated with a strong acid. This is the same type of problem as (2 a) above except in this case a weak base is being neutralized by the strong acid titrant. 19 Weak Acids Weak Bases Titration November 20
- Slides: 18