17 2 Balancing Equations Antoine Laurent Lavoisier established


17. 2 Balancing Equations ØAntoine Laurent Lavoisier, established an important principal based on his experiments with chemical reactions. ØHe stated that the total mass of the products of a reaction is equal to the total mass of the reactants. ØThe law of conservation of mass holds true for even a burning mass of wood.

17. 2 Balancing Equations ØThe combined mass of the burning wood and oxygen is converted into carbon dioxide and water.

17. 2 Conservation of mass ØLavoisier showed that a closed system must be used when studying chemical reactions. ØWhen chemicals are reacted in a closed container, you can show that the mass before and after the reaction is the same.

17. 2 Formula mass ØThe sum of the atomic mass values of the atoms in a chemical formula is called the formula mass.

17. 2 Avogadro’s Number ØThe Avogadro number was named in honor of Amedeo Avogadro who discovered that a mole of any gas under the same conditions has the same number of molecules. ØJohann Josef Loschmidt, a German physicist, named and discovered the Avogadro number. ØLoschmidt realized that a mole of any substance—be it a gas, liquid, or solid— contains 6. 02 x 1023 atoms or molecules.

17. 2 Molar Mass ØThe mass (in grams) of one mole of a compound is called its molar mass.

Solving Problems What is the molar mass of one mole of Ca. CO 3? 1. Looking for: Ø … molar mass of Ca. CO 3 2. Given Ø … chemical formula 3. Relationships: Ø no. amu in formula = molar mass in grams

Solving Problems 4. Solution Formula mass Ca. C 03 = 100. 19 g 1 mole Ca. C 03 = 100. 19 g Ca. CO 3
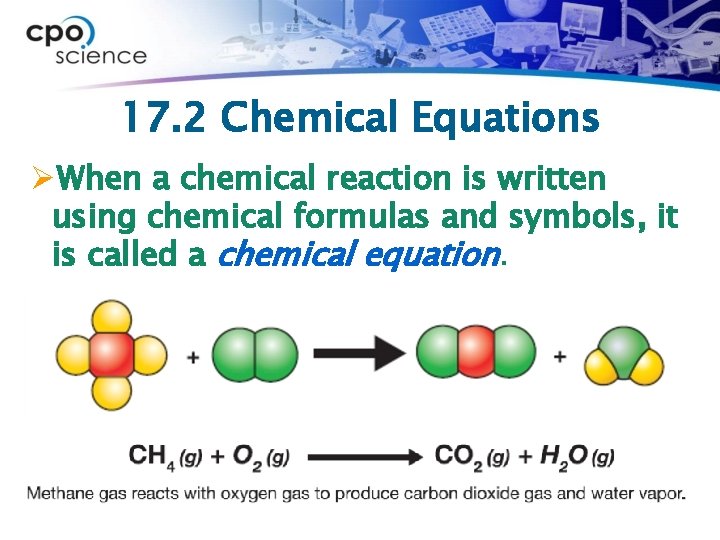
17. 2 Chemical Equations ØWhen a chemical reaction is written using chemical formulas and symbols, it is called a chemical equation.

17. 2 Chemical equations ØAn arrow is always included between reactants and products. ØIt means “to produce” or “to yield. ” to produce Reactants Products “Methane combines with oxygen gas to produce carbon dioxide gas and water vapor. ”

17. 2 Numbers in equations

17. 2 Balancing equations ØThe law conservation of mass is applied by balancing the number and type of atoms on either side of the equation.

17. 2 Balancing equations Ø Counting atoms is necessary to balance an equation. How many carbon atoms? How many hydrogen atoms? How many oxygen atoms?
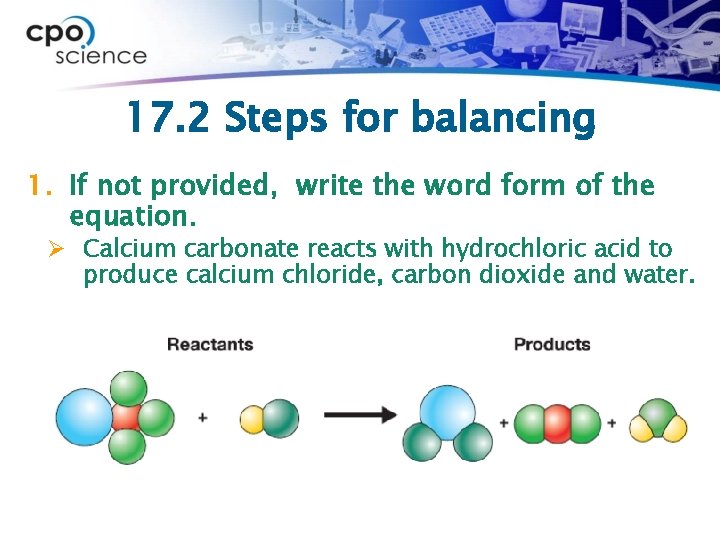
17. 2 Steps for balancing 1. If not provided, write the word form of the equation. Ø Calcium carbonate reacts with hydrochloric acid to produce calcium chloride, carbon dioxide and water.
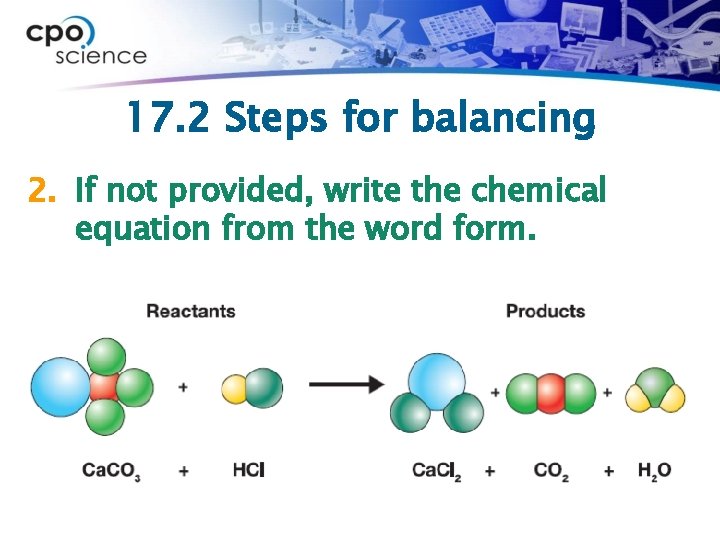
17. 2 Steps for balancing 2. If not provided, write the chemical equation from the word form.

17. 2 Steps for balancing 3. Count the number of each type of atom on both sides.
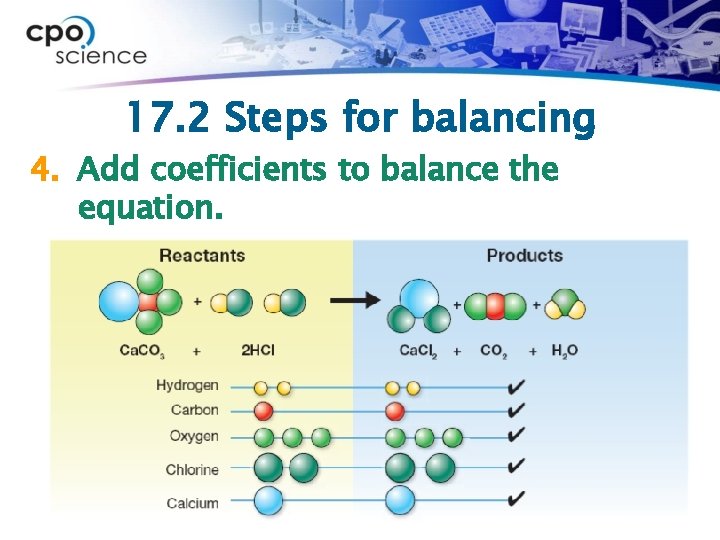
17. 2 Steps for balancing 4. Add coefficients to balance the equation.

Solving Problems Ø In this reaction, chalcocite (a mineral) reacts with oxygen in the presence of heat. The products are a type of copper oxide and sulfur dioxide. Balance this equation: Cu 2 S + O 2 → Cu 2 O + SO 2

Solving Problems 1. Looking for: Ø …the coefficients for each molecule 2. Given Ø … chemical formulas which show types and no. of atoms

Solving Problems 3. Relationships Ø Coefficients can be added in front of any chemical formula in a chemical equation. Ø When a coefficient is added in front of a chemical formula, all atoms in that formula are multiplied by that number. Ø Use common denominators to help choose coefficients to try.

Solving Problems 4. Solution- Trial and error
- Slides: 22