16870 W Bernardo Drive Suite 390 San Diego

16870 W. Bernardo Drive, Suite 390 San Diego, CA 92127 Phone: 858 -618 -1400 Facsimile: 858 -618 -1441 www. aegisthera. com “Increasing transmucosal absorption and improving protein therapeutic viability with alkylsaccharides” BIOCOM – February 13, 2013 Edward T. Maggio, Ph. D CEO, Aegis Therapeutics © 2013 Aegis Therapeutics, Therapeutics LLCLLC

Aegis’ Company Background Ø Located in San Diego Ø We out-license advanced intranasal & transmucosal delivery and stabilization technologies for therapeutic proteins, peptides, and small molecule drugs (Aegis’ Intravail® & Pro. Tek® technologies) Ø Patented technology from the UAB medical school - exclusive license; additional patents recently issued and pending worldwide Ø Multiple product-specific licenses issued to date: ~30 research licenses for ~34 API’s; 8 full commercialization licenses with more conversions in negotiations © 2013 Aegis Therapeutics LLC 2
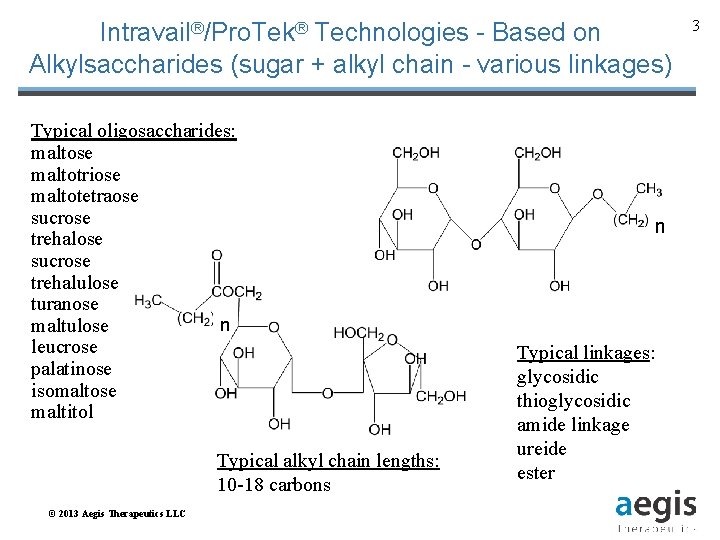
Intravail®/Pro. Tek® Technologies - Based on Alkylsaccharides (sugar + alkyl chain - various linkages) Typical oligosaccharides: maltose maltotriose maltotetraose sucrose trehalulose turanose n maltulose leucrose palatinose isomaltose maltitol Typical alkyl chain lengths: 10 -18 carbons © 2013 Aegis Therapeutics LLC n Typical linkages: glycosidic thioglycosidic amide linkage ureide ester 3
Multiple Modes of Transmucosal Delivery for Macromolecular Drugs Ø Nasal Metered spray pumps Ø Oral (gastrointestinal) Gelcaps Tablets Ø Oral cavity (buccal, sublingual) Flash dissolve Edible films © 2013 Aegis Therapeutics LLC 4
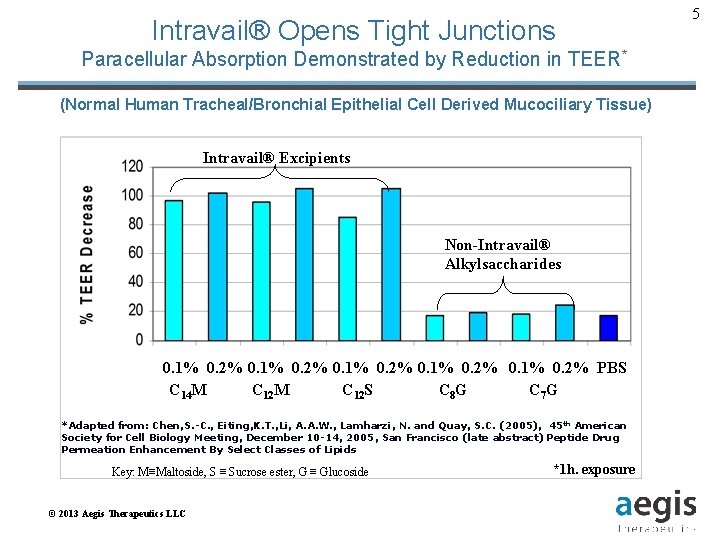
Intravail® Opens Tight Junctions Paracellular Absorption Demonstrated by Reduction in TEER* (Normal Human Tracheal/Bronchial Epithelial Cell Derived Mucociliary Tissue) Intravail® Excipients Non-Intravail® Alkylsaccharides 0. 1% 0. 2% PBS C 14 M C 12 S C 8 G C 7 G *Adapted from: Chen, S. -C. , Eiting, K. T. , Li, A. A. W. , Lamharzi, N. and Quay, S. C. (2005), 45 th American Society for Cell Biology Meeting, December 10 -14, 2005, San Francisco (late abstract) Peptide Drug Permeation Enhancement By Select Classes of Lipids Key: M≡Maltoside, S ≡ Sucrose ester, G ≡ Glucoside © 2013 Aegis Therapeutics LLC *1 h. exposure 5
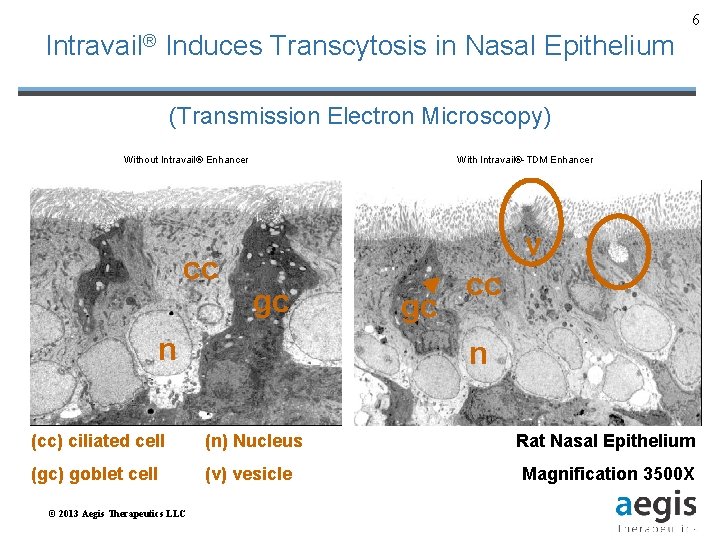
6 Intravail® Induces Transcytosis in Nasal Epithelium (Transmission Electron Microscopy) Without Intravail® Enhancer cc With Intravail®-TDM Enhancer v gc n n (cc) ciliated cell (n) Nucleus (gc) goblet cell (v) vesicle © 2013 Aegis Therapeutics LLC gc cc Rat Nasal Epithelium Magnification 3500 X
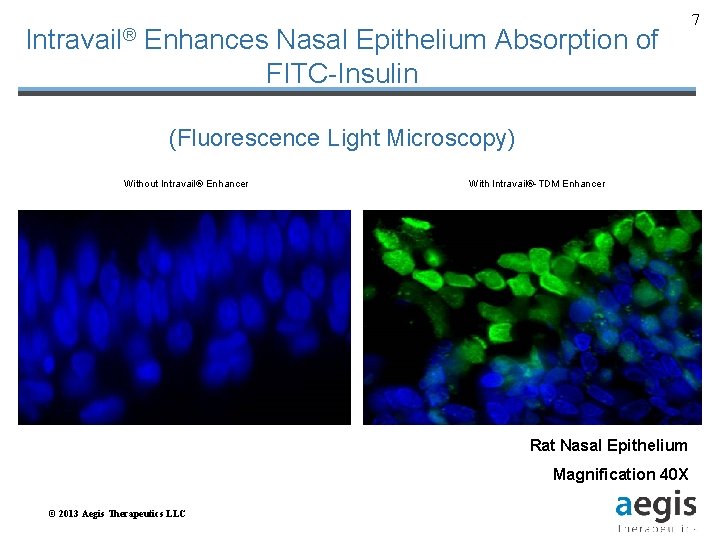
Intravail® Enhances Nasal Epithelium Absorption of FITC-Insulin (Fluorescence Light Microscopy) Without Intravail® Enhancer With Intravail®-TDM Enhancer Rat Nasal Epithelium Magnification 40 X © 2013 Aegis Therapeutics LLC 7
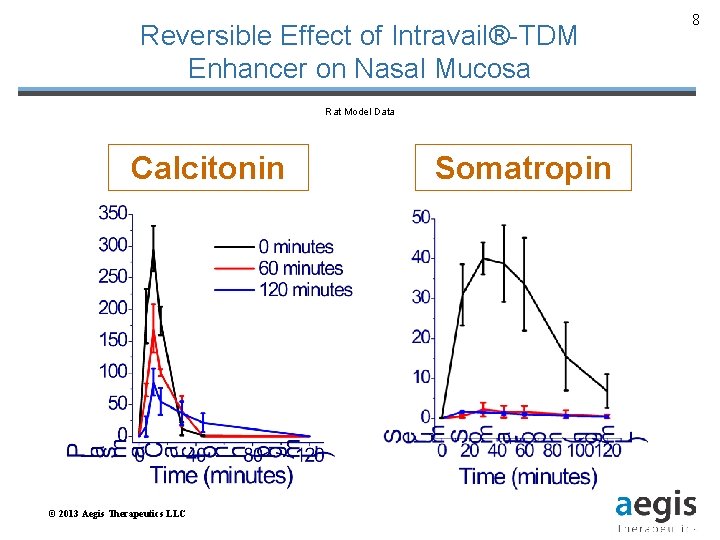
Reversible Effect of Intravail®-TDM Enhancer on Nasal Mucosa Rat Model Data Calcitonin © 2013 Aegis Therapeutics LLC Somatropin 8
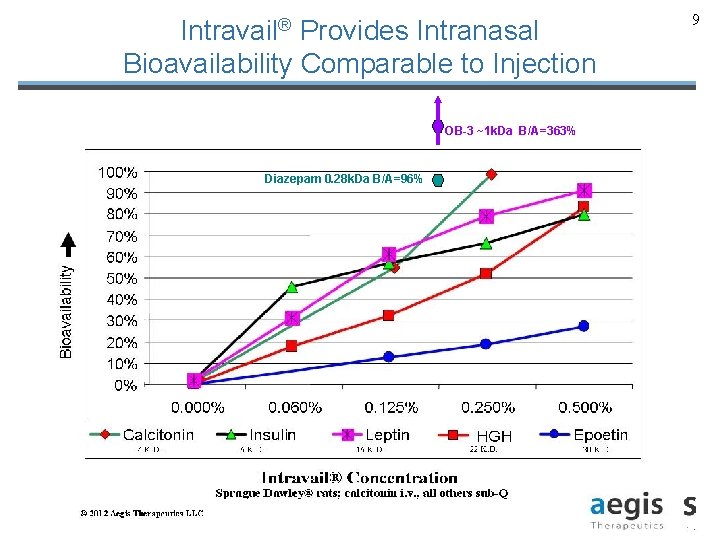
Intravail® Provides Intranasal Bioavailability Comparable to Injection OB-3 ~1 k. Da B/A=363% Diazepam 0. 28 k. Da B/A=96% © 2013 Aegis Therapeutics LLC 9
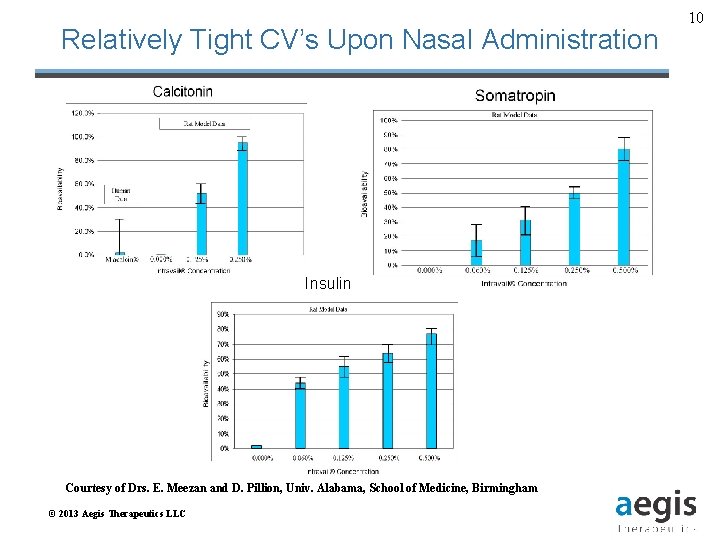
Relatively Tight CV’s Upon Nasal Administration Insulin Courtesy of Drs. E. Meezan and D. Pillion, Univ. Alabama, School of Medicine, Birmingham © 2013 Aegis Therapeutics LLC 10
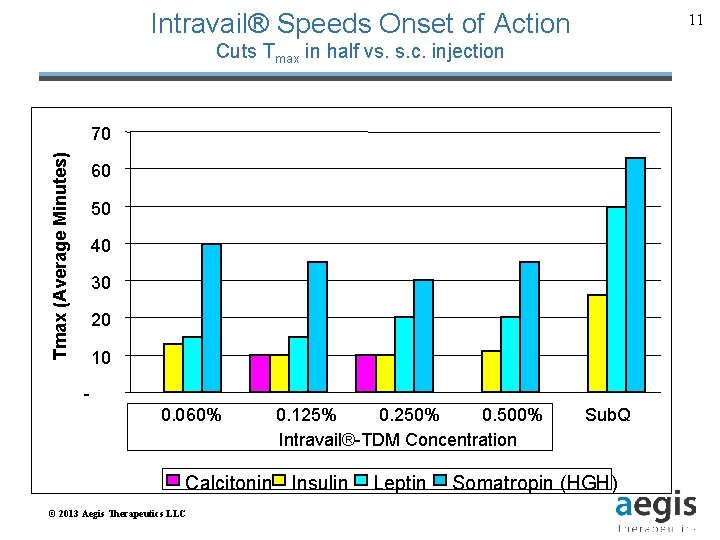
Intravail® Speeds Onset of Action 11 Cuts Tmax in half vs. s. c. injection Tmax (Average Minutes) 70 60 50 40 30 20 10 0. 060% 0. 125% 0. 250% 0. 500% Intravail™ Dose Intravail®-TDM Concentration Sub. Q Calcitonin Insulin Leptin Somatropin (HGH) © 2013 Aegis Therapeutics LLC
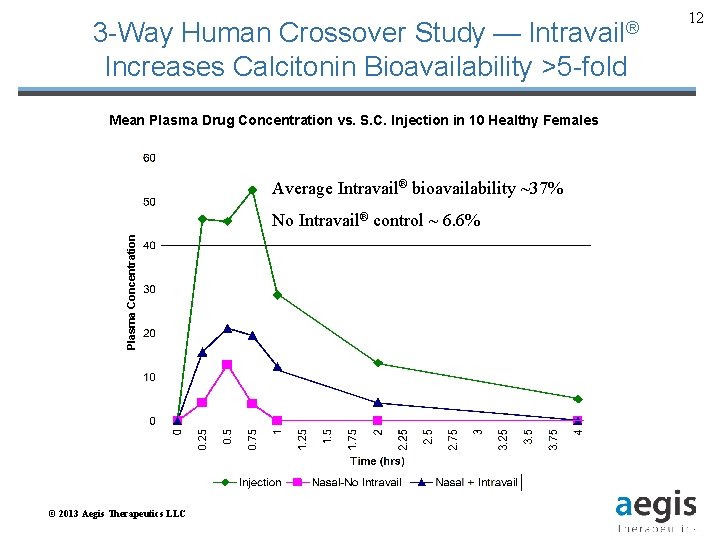
3 -Way Human Crossover Study — Intravail® Increases Calcitonin Bioavailability >5 -fold Mean Plasma Drug Concentration vs. S. C. Injection in 10 Healthy Females Average Intravail® bioavailability ~37% Plasma Concentration No Intravail® control ~ 6. 6% © 2013 Aegis Therapeutics LLC 12
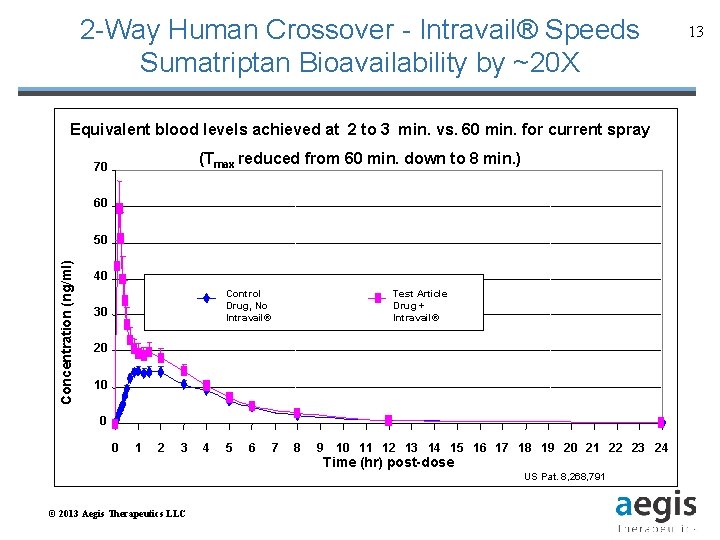
2 -Way Human Crossover - Intravail® Speeds Sumatriptan Bioavailability by ~20 X Equivalent blood levels achieved to 3 min. Sumatriptan vs. 60 min. for current Mean ± S. D. Plasma Profilesat for 2 Intravail® Nasal Spray spray (Tmax reduced from 60 min. down to 8 min. ) 70 60 Concentration (ng/ml) 50 40 Control Reference Test Article A Formulation Drug, No Intravail® 30 Drug + Intravail® 20 10 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Time (hr) post-dose US Pat. 8, 268, 791 © 2013 Aegis Therapeutics LLC 13

Intravail® Nasal Diazepam Provides Superior PK Compared to Diastat® Rectal Gel Intravail® Nasal Diazepam (Licensed by Neurelis Therapeutics, San Diego, CA) Ø Bioavailability 96% vs. i. v. injection (vs. Diastat ~90%) Ø Tmax 1. 5 hr (same as Diastat rectal gel) Ø Cmax 272 ng/m. L (Diastat rectal gel ~262 ng/m. L) Ø Greater convenience/patient acceptance Source: Neurelis Therapeutics, Phase I Study © 2013 Aegis Therapeutics LLC 14

Multiple Modes of Transmucosal Delivery for Macromolecular Drugs Ø Nasal Metered spray pumps Ø Oral (gastrointestinal) Gelcaps Tablets Ø Oral cavity (buccal, sublingual) Flash dissolve Edible films © 2013 Aegis Therapeutics LLC 15
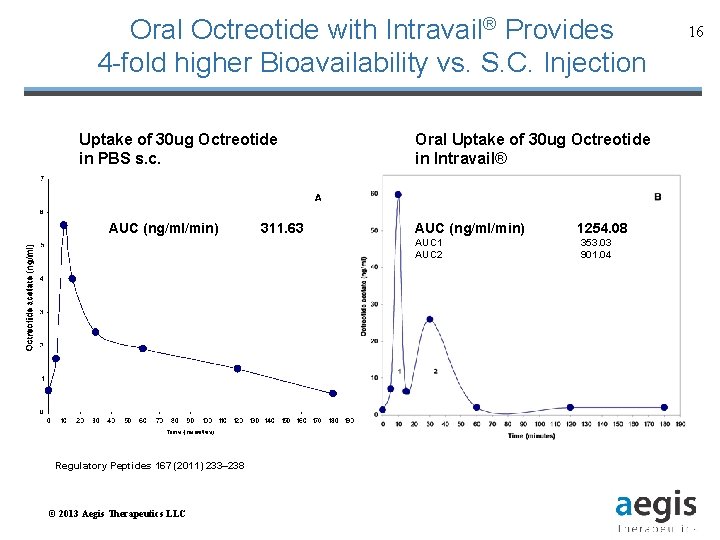
Oral Octreotide with Intravail® Provides 4 -fold higher Bioavailability vs. S. C. Injection Uptake of 30 ug Octreotide in PBS s. c. AUC (ng/ml/min) 311. 63 Oral Uptake of 30 ug Octreotide in Intravail® AUC (ng/ml/min) AUC 1 AUC 2 Regulatory Peptides 167 (2011) 233– 238 © 2013 Aegis Therapeutics LLC 1254. 08 353. 03 901. 04 16
![Oral Delivery of [D-leu-4]OB 3 Anti-Obesity Peptide 17 in Rodents (~1 k. Da MW) Oral Delivery of [D-leu-4]OB 3 Anti-Obesity Peptide 17 in Rodents (~1 k. Da MW)](http://slidetodoc.com/presentation_image_h/d289032e3f9c411ca6e3db50b4861f8d/image-17.jpg)
Oral Delivery of [D-leu-4]OB 3 Anti-Obesity Peptide 17 in Rodents (~1 k. Da MW) Serum Peptide Conc. (ng/m. L) 10000 With Intravail® 8000 552, 710 ng*min/m. L ~56% oral bioavailability 6000 4000 2000 No Intravail® 137, 585 ng*min/m. L 0 0 20 40 Lee et al. Regulatory Peptides 160 (2010) 129– 132 © 2013 Aegis Therapeutics LLC 60 Time (min. ) 80 100 120
![Oral Delivery of Exenatide & [D-leu-4]OB 3 – Synergy in Weight Loss & Glucose Oral Delivery of Exenatide & [D-leu-4]OB 3 – Synergy in Weight Loss & Glucose](http://slidetodoc.com/presentation_image_h/d289032e3f9c411ca6e3db50b4861f8d/image-18.jpg)
Oral Delivery of Exenatide & [D-leu-4]OB 3 – Synergy in Weight Loss & Glucose Control Leinung MC & Grasso P (2012) Regulatory Peptides 179(1 -3): 33 -8. © 2013 Aegis Therapeutics LLC 18
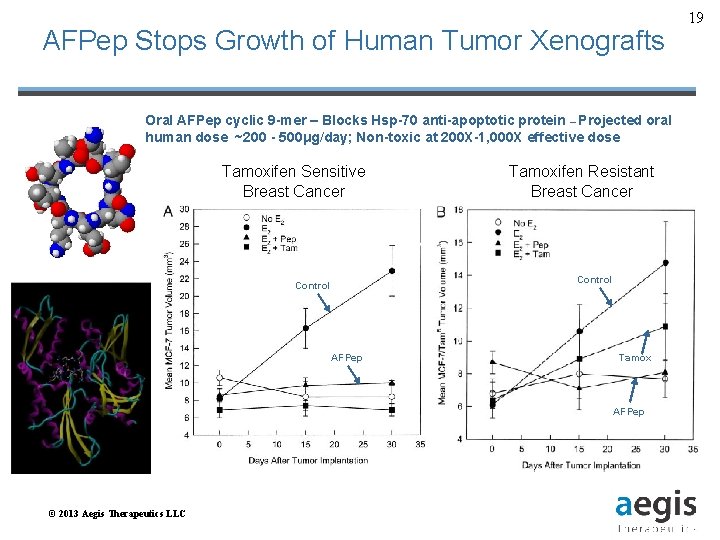
AFPep Stops Growth of Human Tumor Xenografts Oral AFPep cyclic 9 -mer – Blocks Hsp-70 anti-apoptotic protein – Projected oral human dose ~200 - 500μg/day; Non-toxic at 200 X-1, 000 X effective dose Tamoxifen Sensitive Breast Cancer Tamoxifen Resistant Breast Cancer Control AFPep Tamox AFPep © 2013 Aegis Therapeutics LLC 19
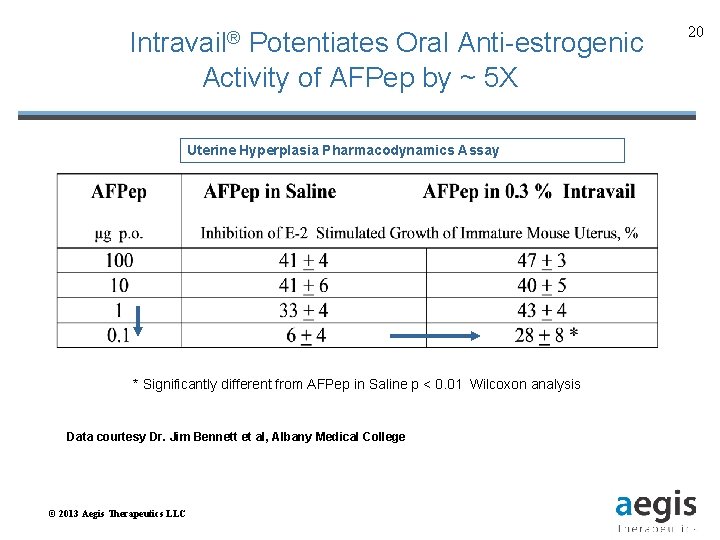
Intravail® Potentiates Oral Anti-estrogenic Activity of AFPep by ~ 5 X Uterine Hyperplasia Pharmacodynamics Assay * Significantly different from AFPep in Saline p < 0. 01 Wilcoxon analysis Data courtesy Dr. Jim Bennett et al, Albany Medical College © 2013 Aegis Therapeutics LLC 20

21 Intravail® Oral Antisense Drug Absorption in Primates Ø Ø Ø Ø Conducted by Aegis-licensee Six canulated monkeys for IJ administration Antisense oligonucleotide - MW ~ 7 KDa Absorption without Intravail® - not detectable Absorption with Intravail® - up to 18% Eight-fold reduction in required excipient volume No decapitation of intestinal pili Presently optimizing the formulation © 2013 Aegis Therapeutics LLC

Multiple Modes of Transmucosal Delivery for Macromolecular Drugs Ø Nasal Metered spray pumps Ø Oral (gastrointestinal) Gelcaps Tablets Ø Oral cavity (buccal, sublingual) Flash dissolve Edible films © 2013 Aegis Therapeutics LLC 22
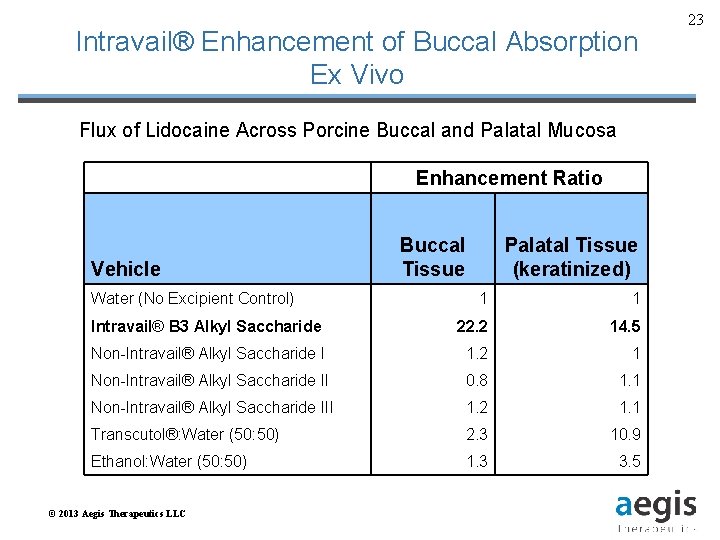
Intravail® Enhancement of Buccal Absorption Ex Vivo Flux of Lidocaine Across Porcine Buccal and Palatal Mucosa Vehicle Water (No Excipient Control) Enhancement Ratio Buccal Tissue Palatal Tissue (keratinized) 1 1 Intravail® B 3 Alkyl Saccharide 22. 2 14. 5 Non-Intravail® Alkyl Saccharide I 1. 2 1 Non-Intravail® Alkyl Saccharide II 0. 8 1. 1 Non-Intravail® Alkyl Saccharide III 1. 2 1. 1 Transcutol®: Water (50: 50) 2. 3 10. 9 Ethanol: Water (50: 50) 1. 3 3. 5 © 2013 Aegis Therapeutics LLC 23
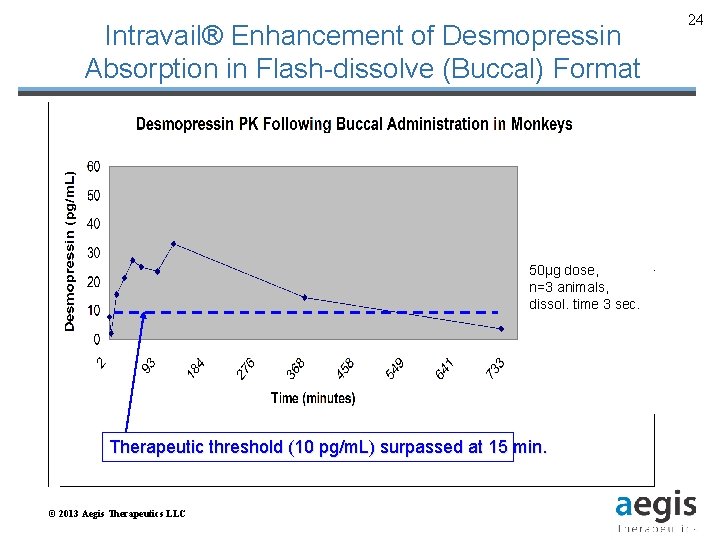
Intravail® Enhancement of Desmopressin Absorption in Flash-dissolve (Buccal) Format 50μg dose, n=3 animals, dissol. time 3 sec. Therapeutic threshold (10 pg/m. L) surpassed at 15 min. © 2013 Aegis Therapeutics LLC 24
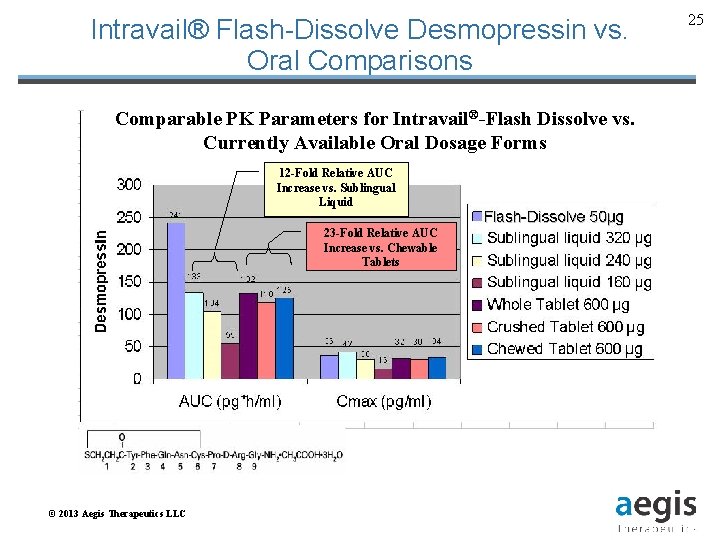
Intravail® Flash-Dissolve Desmopressin vs. Oral Comparisons Comparable PK Parameters for Intravail®-Flash Dissolve vs. Currently Available Oral Dosage Forms 12 -Fold Relative AUC Increase vs. Sublingual Liquid 23 -Fold Relative AUC Increase vs. Chewable Tablets © 2013 Aegis Therapeutics LLC 25

26 Protein Aggregation and Immunogenicity Ø Defining the Problem Ø Current Formulation Practice Ø Need for Alternatives Ø Regulatory Impact Fibrils of a linear octapeptide CD 4 blocker form within 10 min. of aqueous dissolution (electron micrograph). © 2013 Aegis Therapeutics LLC

27 Problems Caused by Protein Aggregation Ø Ø Ø Loss of efficacy Altered PK Increased immunogenicity Anaphylaxis or skin sensitization Reduced shelf-life Manufacturing yield losses due to membrane or column fouling or precipitation Ø m. Ab’s particularly prone to aggregation © 2013 Aegis Therapeutics LLC

Surfactants Counteract Protein Aggregation Ø Surfactants prevent or reduce aggregation Ø This results in stabilized bioactivity and reduced immunogenicity Ø Polysorbates (i. e. PS-20 and PS-80) are the most commonly used anti-aggregation surfactants Ø Highly effective at preventing aggregation Ø Well-accepted excipients - long track record of use © 2013 Aegis Therapeutics LLC 28
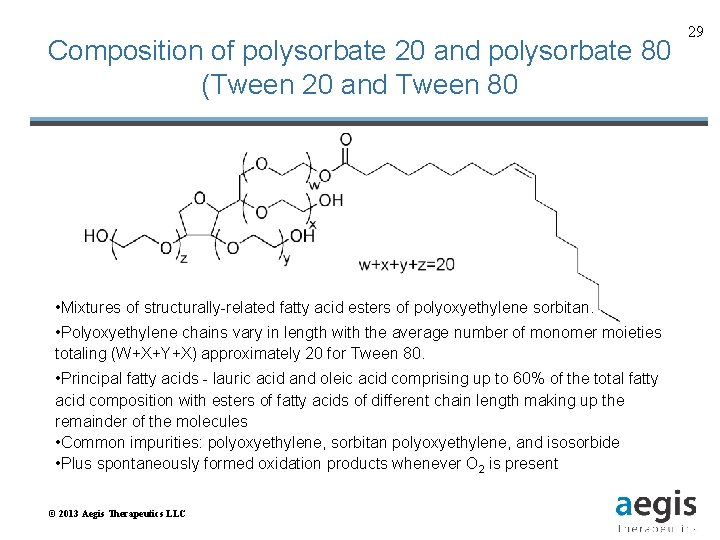
Composition of polysorbate 20 and polysorbate 80 (Tween 20 and Tween 80 • Mixtures of structurally-related fatty acid esters of polyoxyethylene sorbitan. • Polyoxyethylene chains vary in length with the average number of monomer moieties totaling (W+X+Y+X) approximately 20 for Tween 80. • Principal fatty acids - lauric acid and oleic acid comprising up to 60% of the total fatty acid composition with esters of fatty acids of different chain length making up the remainder of the molecules • Common impurities: polyoxyethylene, sorbitan polyoxyethylene, and isosorbide • Plus spontaneously formed oxidation products whenever O 2 is present © 2013 Aegis Therapeutics LLC 29
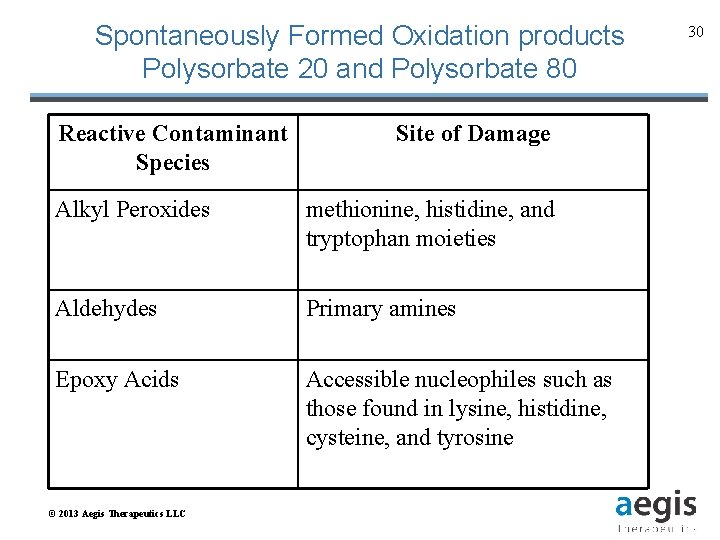
Spontaneously Formed Oxidation products Polysorbate 20 and Polysorbate 80 Reactive Contaminant Species Site of Damage Alkyl Peroxides methionine, histidine, and tryptophan moieties Aldehydes Primary amines Epoxy Acids Accessible nucleophiles such as those found in lysine, histidine, cysteine, and tyrosine © 2013 Aegis Therapeutics LLC 30
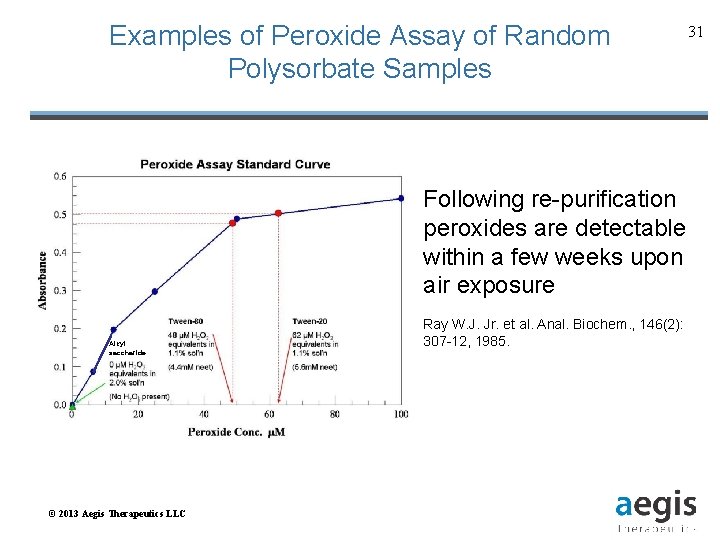
Examples of Peroxide Assay of Random Polysorbate Samples 31 Following re-purification peroxides are detectable within a few weeks upon air exposure Alkyl saccharide © 2013 Aegis Therapeutics LLC Ray W. J. Jr. et al. Anal. Biochem. , 146(2): 307 -12, 1985.

Problems with Polysorbates 32 Ø Underlying chemical structures spontaneously oxidize to peroxides, epoxy-acids and aldehydes Ø These reactive elements increase immunogenicity Ø Non-immunologic hypersensitivity and anaphylactoid reactions have also been attributed to poylsorbates in biotherapeutics Ø Growing immunogenicity concerns by regulatory authorities, physicians, and patients creates a need to find non-reactive alternative surfactants © 2013 Aegis Therapeutics LLC

Pro. Tek® Alkyl Saccharide Excipients - Possible Alternatives to Polysorbates Ø Prevent aggregation Ø Reduce immunogenicity Ø Increase shelf-life Ø Reduce denaturation due to elevated temperatures and shear force Ø Increase reproducibility of lyophilizates upon reconstitution Ø Extend patent franchise of existing biotherapeutics through formulation “Compositions of Matter” © 2013 Aegis Therapeutics LLC Provided Courtesy of Aegis Therapeutics, LLC 33
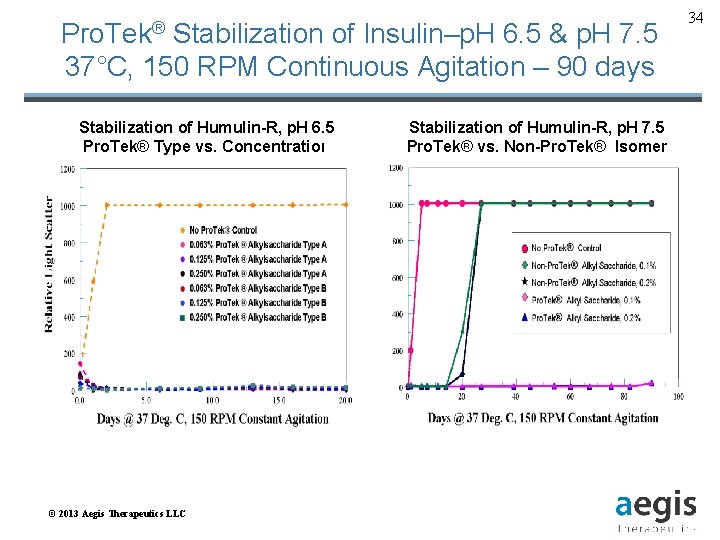
Pro. Tek® Stabilization of Insulin–p. H 6. 5 & p. H 7. 5 37°C, 150 RPM Continuous Agitation – 90 days Stabilization of Humulin-R, p. H 6. 5 Pro. Tek® Type vs. Concentration © 2013 Aegis Therapeutics LLC Stabilization of Humulin-R, p. H 7. 5 Pro. Tek® vs. Non-Pro. Tek® Isomer 34
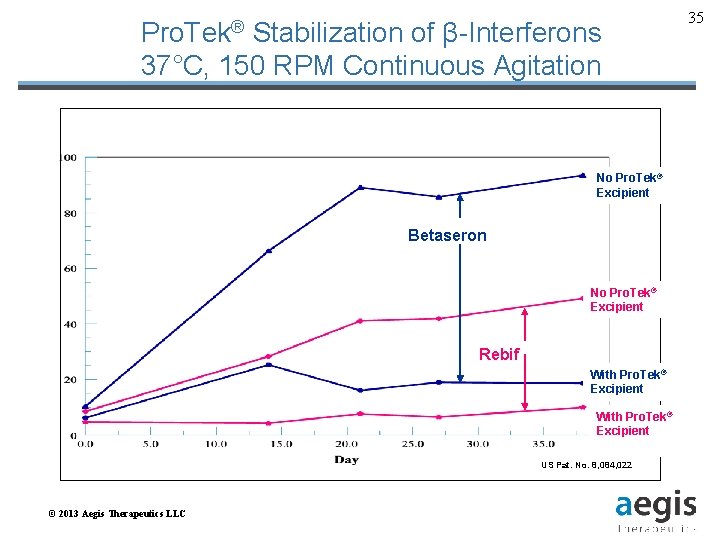
Pro. Tek® Stabilization of β-Interferons 37°C, 150 RPM Continuous Agitation No Pro. Tek® Excipient Betaseron No Pro. Tek® Excipient Rebif With Pro. Tek® Excipient US Pat. No. 8, 084, 022 Patent Pending © 2013 Aegis Therapeutics LLC Provided Courtesy of Aegis Therapeutics, LLC 35
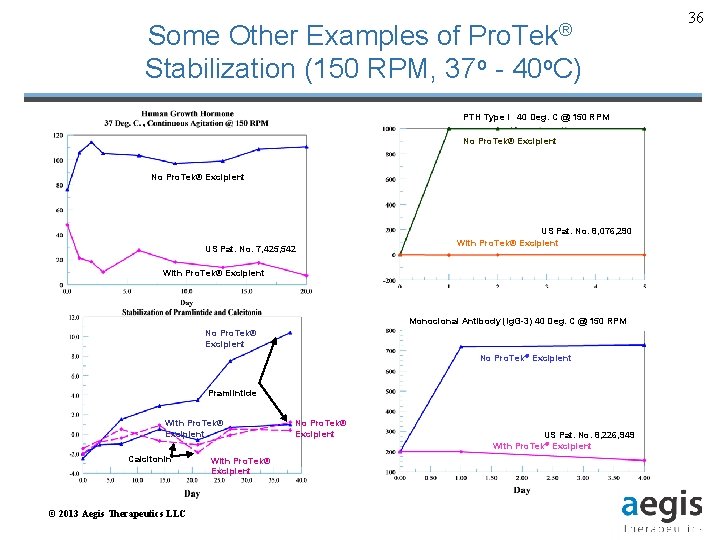
Some Other Examples of Pro. Tek® Stabilization (150 RPM, 37 o - 40 o. C) PTH Type I 40 Deg. C @ 150 RPM No Pro. Tek® Excipient US Pat. No. 7, 425, 542 US Pat. No. 8, 076, 290 With Pro. Tek® Excipient Monoclonal Antibody (Ig. G-3) 40 Deg. C @ 150 RPM No Pro. Tek® Excipient Pramlintide With Pro. Tek® Excipient Calcitonin © 2013 Aegis Therapeutics LLC With Pro. Tek® Excipient No Pro. Tek® Excipient US Pat. No. 8, 226, 949 With Pro. Tek® Excipient 36

Intravail®/Pro. Tek® Safety Profile Ø Safe, odorless, tasteless, non-toxic, non-mutagenic, and nonirritating Ø Chemically synthesized molecules - metabolize to CO 2 & H 2 O Ø Nonionic, non-denaturing, and pharmaceutically compatible with virtually any protein, peptide or drug Ø Non-sensitizing and non-irritating to the rabbit eye @ 25% concentration Ø Mild surfactants used in personal care and food products (NOEL ~ 20 g/kg bw); designated as “GRAS” by FDA Ø W. H. O. ADI ~ 15, 000 X the daily intake (~41 yrs of daily use) © 2013 Aegis Therapeutics LLC 37

Intravail® Summary 38 Ø Allows non-invasive delivery of large and small molecule drugs (enhanced mucosal absorption) Ø Unmatched intranasal bioavailability (up to ~30 k. D) Ø Rapid onset of action Ø Avoidance of gastric hydrolysis & “first pass effect” Ø Elimination of needle stick injuries/infections Ø Compatible with “off-the-shelf” metered nasal spray devices and oral dosage forms Ø High oral bioavailability for certain peptides/non-peptides Ø Greater patient convenience and compliance Ø Low cost of GMP Intravail® excipients © 2013 Aegis Therapeutics LLC

39 Pro. Tek® Summary Ø Increased stability Ø Aggregation reduction Ø Reduced intrinsic immunogenicity Ø No peroxide damage or peroxide induced immunogenicity Ø Higher manufacturing yields Ø Less stringent “cold-chain” requirements Ø Extended patent franchise for existing biotherapeutics through formulation “Compositions of Matter” © 2013 Aegis Therapeutics LLC Provided Courtesy of Aegis Therapeutics, LLC

16870 West Bernardo Drive, Suite 390 San Diego, CA 92127 Phone: 858 -618 -1400 Facsimile: 858 -618 -1441 www. aegisthera. com Contact Information: Edward T. Maggio, Ph. D. , Chief Executive Officer emaggio@aegisthera. com Ralph R. Barry, Chief Business Officer & CFO rbarry@aegisthera. com © 2013 Aegis Therapeutics LLC
- Slides: 40