16 th Interventional Cardiology Symposium Montreal Quebec June

16 th Interventional Cardiology Symposium Montreal, Quebec / June 14 -16, 2007 Adapted from a presentation by: Shamir R. Mehta, MD, MSc, FRCPC, FACC “Transitioning ACS Patients to the Cath Lab: Optimizing Outcomes with Upstream Fondaparinux and Bivalirudin/UFH” Friday, June 15, 2007

Transitioning ACS Patients to the Cath Lab: Optimizing Outcomes with Upstream Fondaparinux and Bivalirudin/UFH Shamir R. Mehta, MD, MSc, FRCPC, FACC Director, Interventional Cardiology Hamilton Health Sciences Associate Professor of Medicine Mc. Master University Hamilton, Ontario, Canada
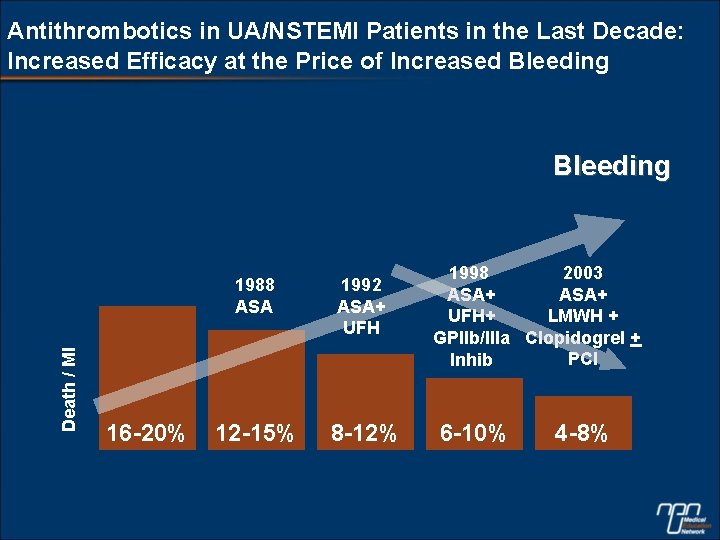
Antithrombotics in UA/NSTEMI Patients in the Last Decade: Increased Efficacy at the Price of Increased Bleeding Death / MI Bleeding 16 -20% 1988 ASA 1992 ASA+ UFH 12 -15% 8 -12% 2003 1998 ASA+ LMWH + UFH+ GPIIb/IIIa Clopidogrel + PCI Inhib 6 -10% 4 -8%
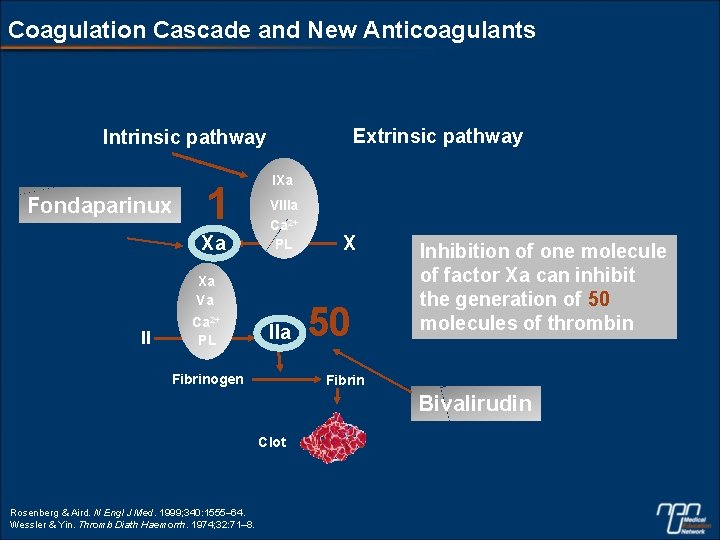
Coagulation Cascade and New Anticoagulants Extrinsic pathway Intrinsic pathway Fondaparinux 1 Xa IXa VIIIa Ca 2+ PL Xa Va II Ca 2+ PL IIa Fibrinogen X 50 Inhibition of one molecule of factor Xa can inhibit the generation of 50 molecules of thrombin Fibrin Bivalirudin Clot Rosenberg & Aird. N Engl J Med. 1999; 340: 1555– 64. Wessler & Yin. Thromb Diath Haemorrh. 1974; 32: 71– 8.

OASIS-5: A Randomized, Double-Blind, Double-Dummy Trial 20, 078 patients with UA/NSTEMI Aspirin, Clopidogrel, anti-GPIIb/IIIa, planned Cath/PCI as per local practice Randomization Fondaparinux Enoxaparin 2. 5 mg s. c. od up to 8 days 1 mg/kg s. c. bid for 2 -8 days 1 mg/kg s. c. od if Cl. Cr<30 m. L/min Michelangelo OASIS-5 Steering Committee. Am Heart J. 2005; 150: 1107. e 1 -. e 10. OASIS 5 Investigators. I. 1464 -76.
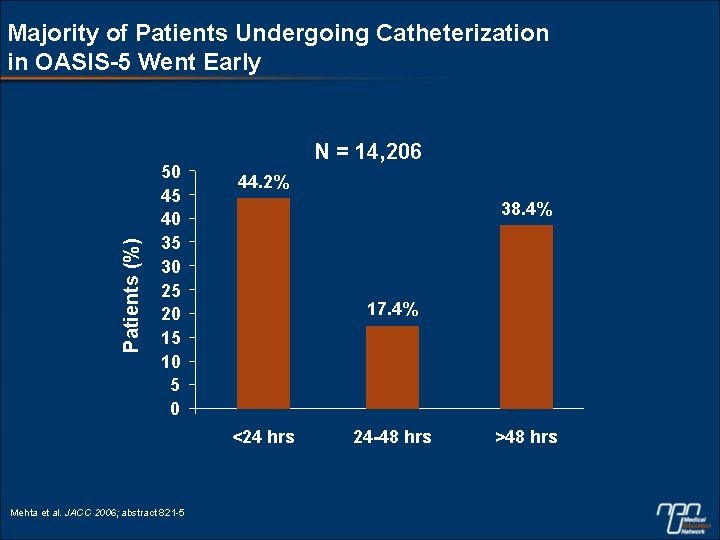
Patients (%) Majority of Patients Undergoing Catheterization in OASIS-5 Went Early 50 45 40 35 30 25 20 15 10 5 0 N = 14, 206 44. 2% 38. 4% 17. 4% <24 hrs Mehta et al. JACC 2006; abstract 821 -5 24 -48 hrs >48 hrs
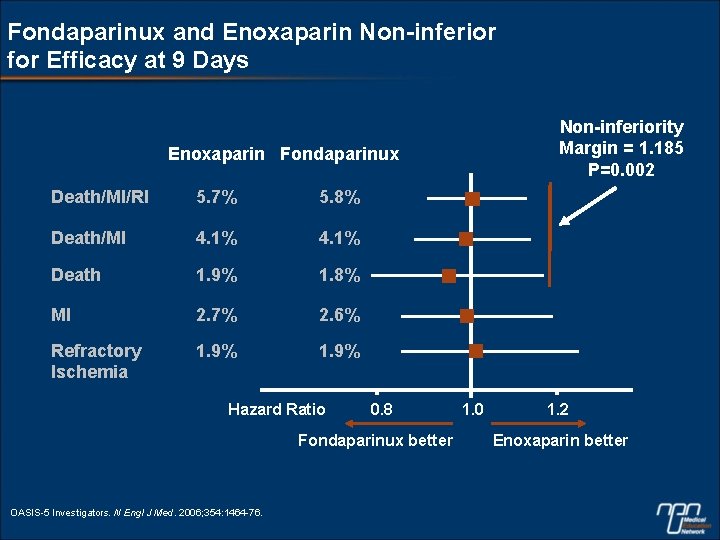
Fondaparinux and Enoxaparin Non-inferior for Efficacy at 9 Days Non-inferiority Margin = 1. 185 P=0. 002 Enoxaparin Fondaparinux Death/MI/RI 5. 7% 5. 8% Death/MI 4. 1% Death 1. 9% 1. 8% MI 2. 7% 2. 6% Refractory Ischemia 1. 9% Hazard Ratio 0. 8 Fondaparinux better OASIS-5 Investigators. N Engl J Med. 2006; 354: 1464 -76. 1. 0 1. 2 Enoxaparin better
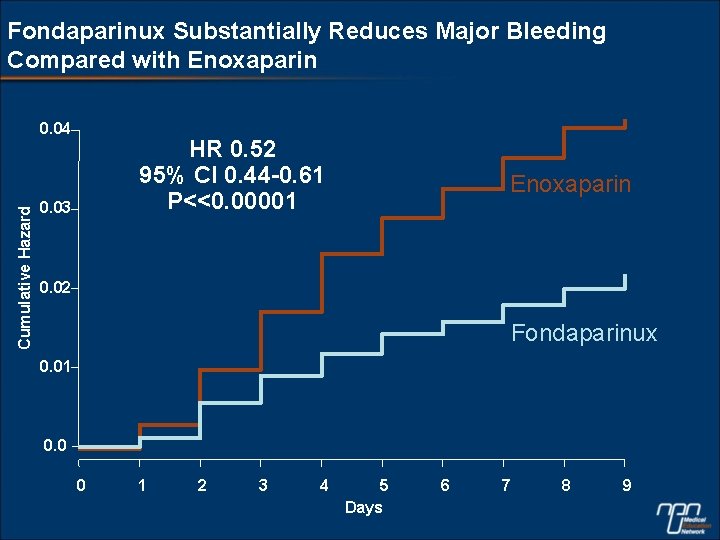
Fondaparinux Substantially Reduces Major Bleeding Compared with Enoxaparin Cumulative Hazard 0. 04 HR 0. 52 95% CI 0. 44 -0. 61 P<<0. 00001 0. 03 Enoxaparin 0. 02 Fondaparinux 0. 01 0. 0 0 1 2 3 4 5 Days 6 7 8 9
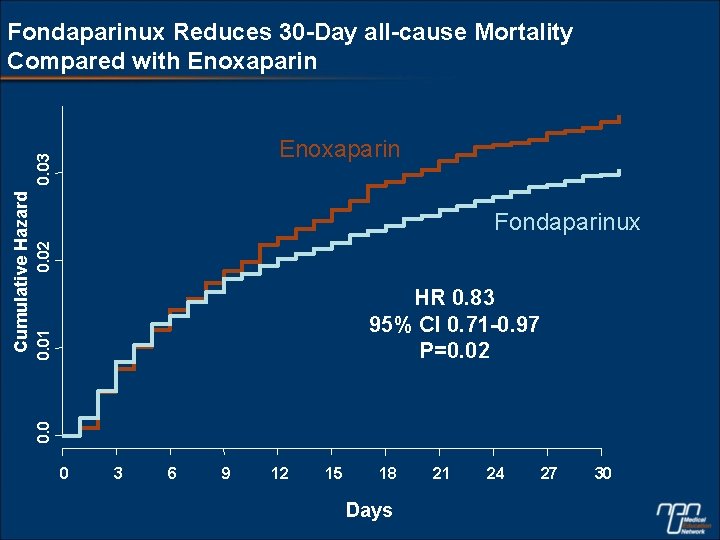
Fondaparinux Reduces 30 -Day all-cause Mortality Compared with Enoxaparin 0. 02 Fondaparinux 0. 01 HR 0. 83 95% CI 0. 71 -0. 97 P=0. 02 0. 0 Cumulative Hazard 0. 03 Enoxaparin 0 3 6 9 12 15 18 Days 21 24 27 30
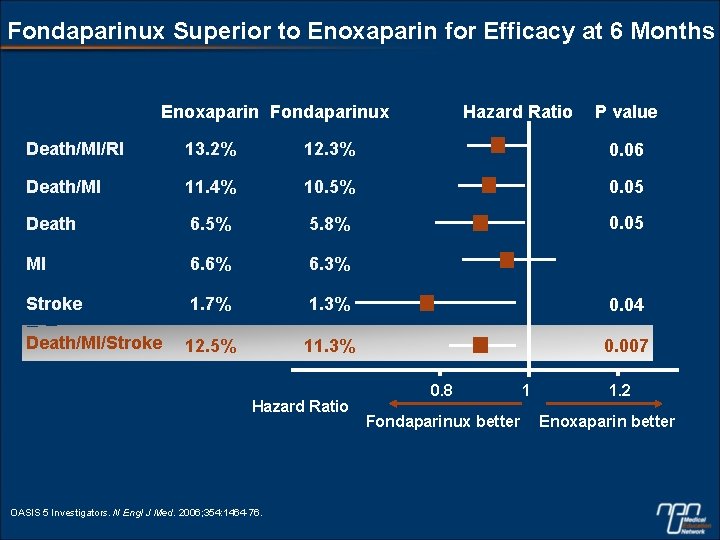
Fondaparinux Superior to Enoxaparin for Efficacy at 6 Months Enoxaparin Fondaparinux Hazard Ratio P value Death/MI/RI 13. 2% 12. 3% 0. 06 Death/MI 11. 4% 10. 5% 0. 05 Death 6. 5% 5. 8% 0. 05 MI 6. 6% 6. 3% Stroke 1. 7% 1. 3% 0. 04 Death/MI/Stroke 12. 5% 11. 3% 0. 007 Hazard Ratio OASIS 5 Investigators. N Engl J Med. 2006; 354: 1464 -76. 0. 8 Fondaparinux better 1 1. 2 Enoxaparin better
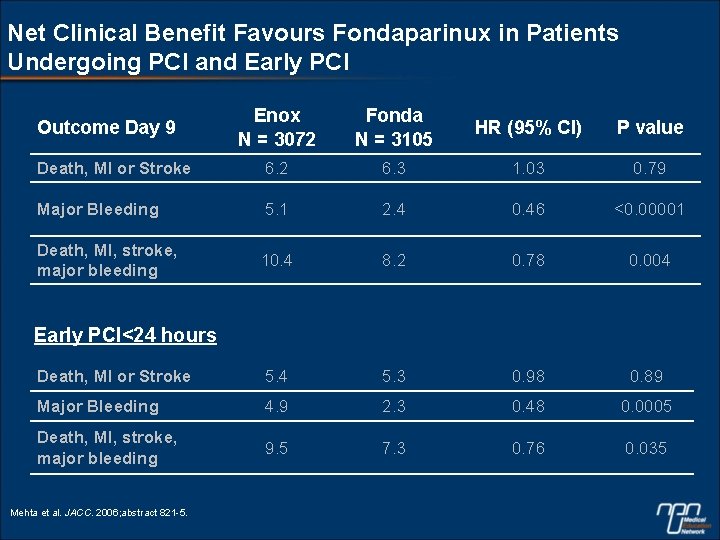
Net Clinical Benefit Favours Fondaparinux in Patients Undergoing PCI and Early PCI Enox N = 3072 Fonda N = 3105 HR (95% CI) P value Death, MI or Stroke 6. 2 6. 3 1. 03 0. 79 Major Bleeding 5. 1 2. 4 0. 46 <0. 00001 Death, MI, stroke, major bleeding 10. 4 8. 2 0. 78 0. 004 Death, MI or Stroke 5. 4 5. 3 0. 98 0. 89 Major Bleeding 4. 9 2. 3 0. 48 0. 0005 Death, MI, stroke, major bleeding 9. 5 7. 3 0. 76 0. 035 Outcome Day 9 Early PCI<24 hours Mehta et al. JACC. 2006; abstract 821 -5.
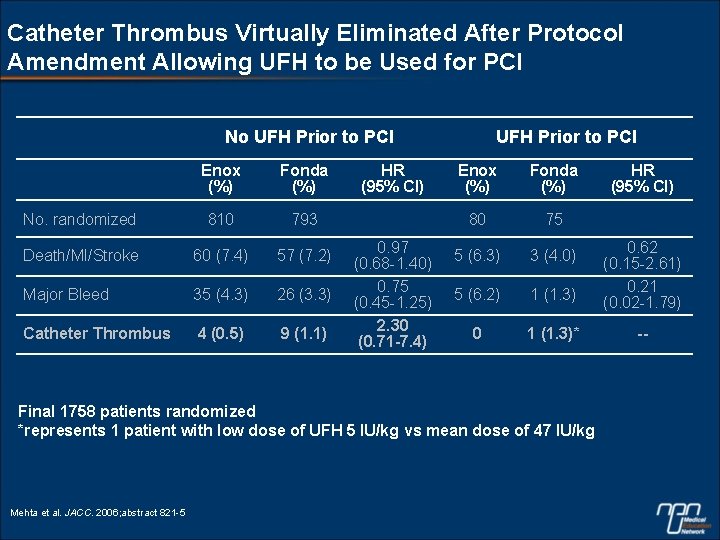
Catheter Thrombus Virtually Eliminated After Protocol Amendment Allowing UFH to be Used for PCI No UFH Prior to PCI Enox (%) Fonda (%) No. randomized 810 793 Death/MI/Stroke 60 (7. 4) 57 (7. 2) Major Bleed 35 (4. 3) 26 (3. 3) Catheter Thrombus 4 (0. 5) 9 (1. 1) HR (95% CI) 0. 97 (0. 68 -1. 40) 0. 75 (0. 45 -1. 25) 2. 30 (0. 71 -7. 4) UFH Prior to PCI Enox (%) Fonda (%) 80 75 5 (6. 3) 3 (4. 0) 5 (6. 2) 1 (1. 3) 0 1 (1. 3)* Final 1758 patients randomized *represents 1 patient with low dose of UFH 5 IU/kg vs mean dose of 47 IU/kg Mehta et al. JACC. 2006; abstract 821 -5 HR (95% CI) 0. 62 (0. 15 -2. 61) 0. 21 (0. 02 -1. 79) --
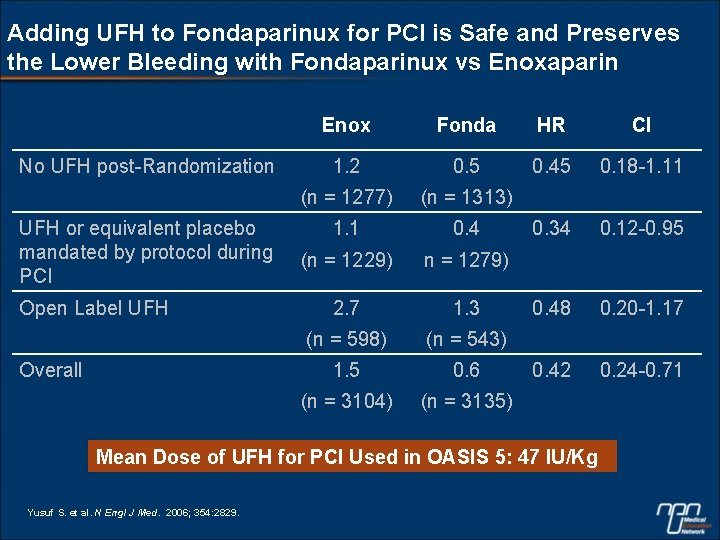
Adding UFH to Fondaparinux for PCI is Safe and Preserves the Lower Bleeding with Fondaparinux vs Enoxaparin No UFH post-Randomization UFH or equivalent placebo mandated by protocol during PCI Open Label UFH Overall Enox Fonda HR CI 1. 2 0. 5 0. 45 0. 18 -1. 11 (n = 1277) (n = 1313) 1. 1 0. 4 0. 34 0. 12 -0. 95 (n = 1229) n = 1279) 2. 7 1. 3 0. 48 0. 20 -1. 17 (n = 598) (n = 543) 1. 5 0. 6 0. 42 0. 24 -0. 71 (n = 3104) (n = 3135) Mean Dose of UFH for PCI Used in OASIS 5: 47 IU/Kg Yusuf S. et al. N Engl J Med. 2006; 354: 2829.
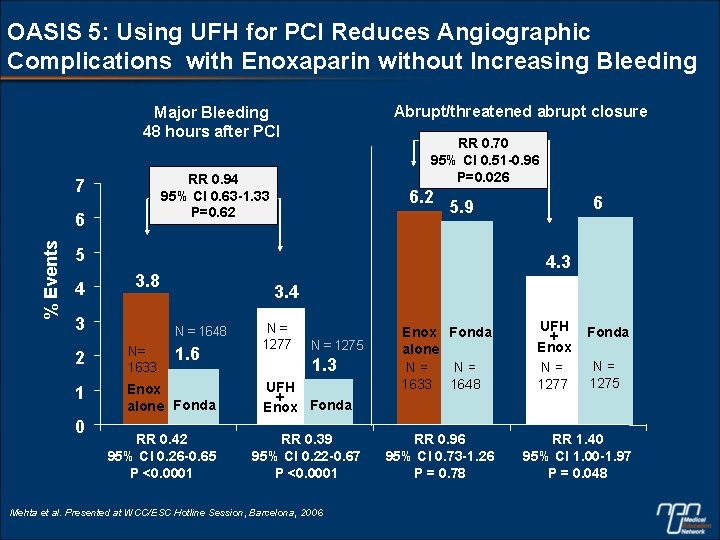
OASIS 5: Using UFH for PCI Reduces Angiographic Complications with Enoxaparin without Increasing Bleeding Abrupt/threatened abrupt closure Major Bleeding 48 hours after PCI RR 0. 94 95% CI 0. 63 -1. 33 P=0. 62 7 6 % Events RR 0. 70 95% CI 0. 51 -0. 96 P=0. 026 6. 2 5 4 4. 3 3. 8 3 3. 4 N = 1648 1. 6 2 N= 1633 1 Enox alone Fonda 0 6 5. 9 RR 0. 42 95% CI 0. 26 -0. 65 P <0. 0001 NN == 1277 N = 1275 1. 3 UFH + Enox Fonda RR 0. 39 95% CI 0. 22 -0. 67 P <0. 0001 Mehta et al. Presented at WCC/ESC Hotline Session, Barcelona, 2006 N= 1648 Enox Fonda N= alone 1633 N= N= 1633 1648 UFH Fonda + Enox == N= NN 1275 1277 1275 RR 0. 96 95% CI 0. 73 -1. 26 P = 0. 78 RR 1. 40 95% CI 1. 00 -1. 97 P = 0. 048
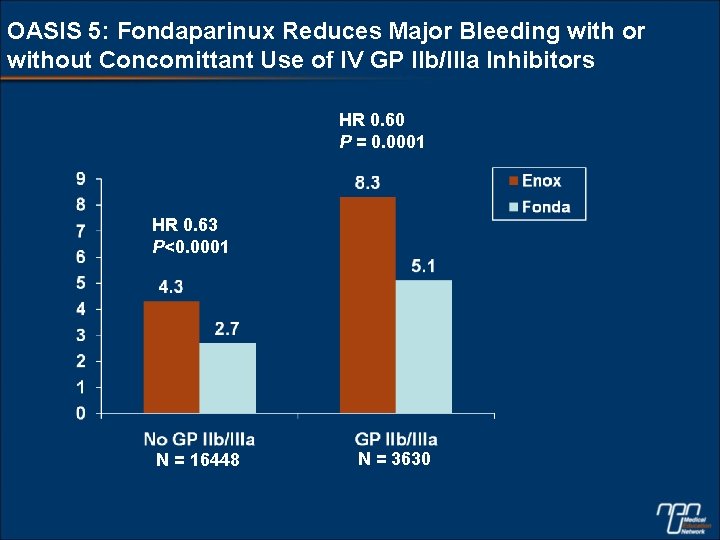
OASIS 5: Fondaparinux Reduces Major Bleeding with or without Concomittant Use of IV GP IIb/IIIa Inhibitors HR 0. 60 P = 0. 0001 HR 0. 63 P<0. 0001 N = 16448 N = 3630
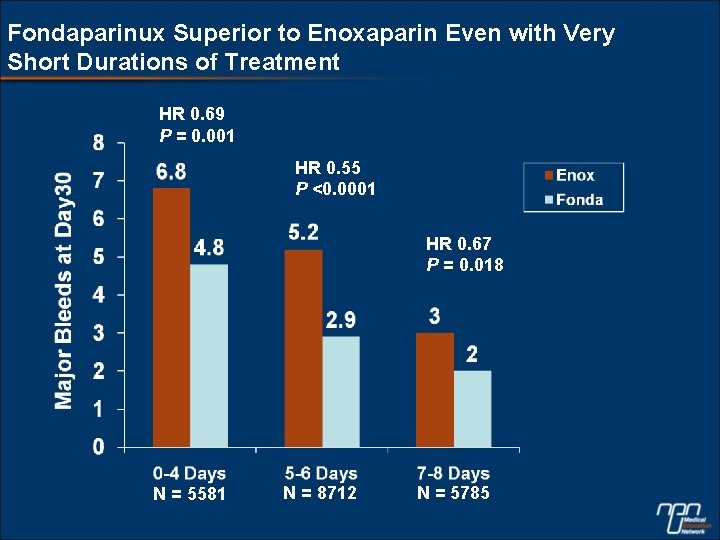
Fondaparinux Superior to Enoxaparin Even with Very Short Durations of Treatment HR 0. 69 P = 0. 001 HR 0. 55 P <0. 0001 HR 0. 67 P = 0. 018 N = 5581 N = 8712 N = 5785

How to Transition Patients Initiated on Fonda to the Cath Lab • Treat with ASA, Clopidogrel, Statin and Fondaparinux +/- IV nitro in the ER • Proceed to Cath Lab as usual* • If PCI needed, give bivalirudin or UFH (dose 50 IU/kg) +/- IIb/IIIa • Post procedure, follow usual practice for sheath removal. Immediate removal if closure device or radial and 6 hours after last fonda sc dose if no closure device used *May perform cath>6 hours after last sc dose if this was center’s usual practice with using LMWH
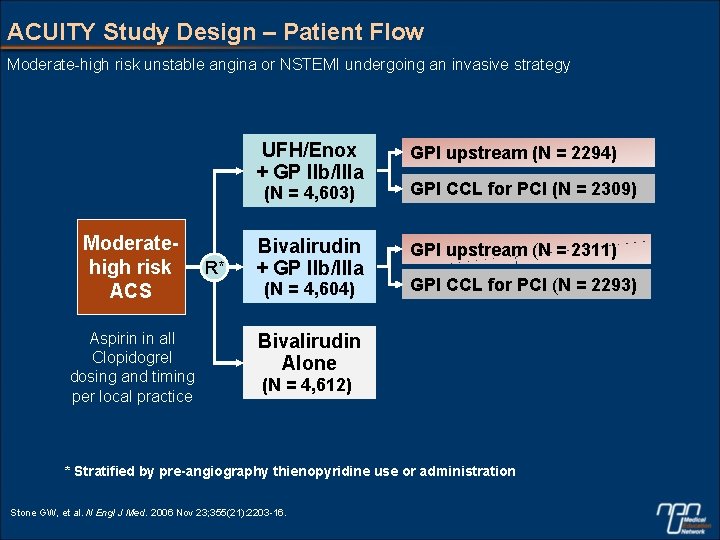
ACUITY Study Design – Patient Flow Moderate-high risk unstable angina or NSTEMI undergoing an invasive strategy UFH/Enox + GP IIb/IIIa GPI upstream (N = 2294) Bivalirudin + GP IIb/IIIa GPI upstream (N = 2311) (N = 4, 603) Moderatehigh risk ACS Aspirin in all Clopidogrel dosing and timing per local practice R* (N = 4, 604) GPI CCL for PCI (N = 2309) GPI CCL for PCI (N = 2293) Bivalirudin Alone (N = 4, 612) * Stratified by pre-angiography thienopyridine use or administration Stone GW, et al. N Engl J Med. 2006 Nov 23; 355(21): 2203 -16.
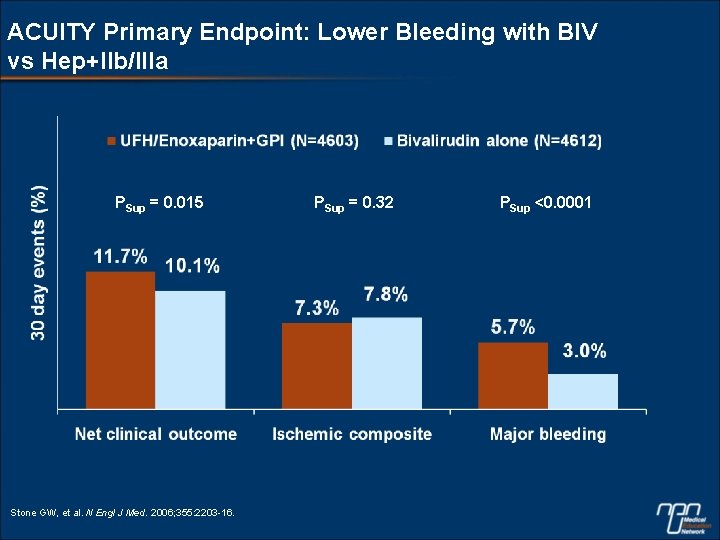
ACUITY Primary Endpoint: Lower Bleeding with BIV vs Hep+IIb/IIIa PSup = 0. 015 Stone GW, et al. N Engl J Med. 2006; 355: 2203 -16. PSup = 0. 32 PSup <0. 0001
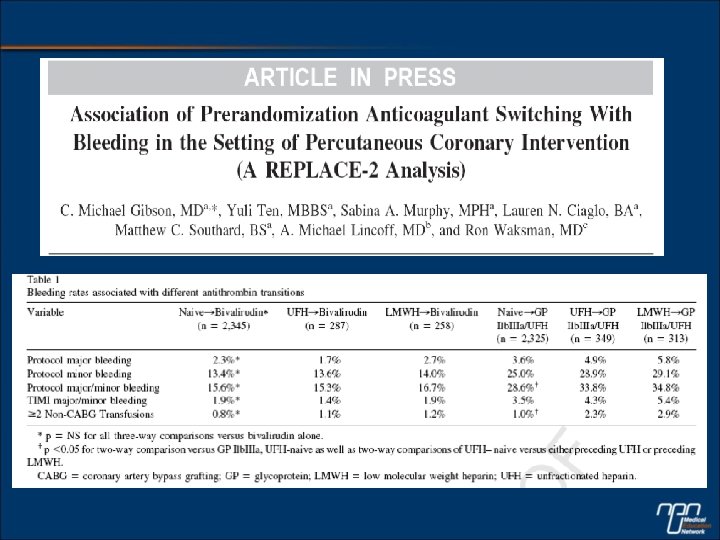
![ACUITY PCI: Influence of Thienopyridine Exposure – PCI pts RR [95%CI] 0. 81 (0. ACUITY PCI: Influence of Thienopyridine Exposure – PCI pts RR [95%CI] 0. 81 (0.](http://slidetodoc.com/presentation_image_h/01ee703402e15477b0d1f27d0cd9d58a/image-21.jpg)
ACUITY PCI: Influence of Thienopyridine Exposure – PCI pts RR [95%CI] 0. 81 (0. 68 -0. 96) RR [95%CI] 0. 96 (0. 77 -1. 20) RR [95%CI] 0. 50 (0. 37 -0. 67) RR [95%CI] 1. 07 (0. 83 -1. 39) Thienopyridine Exposed* *Thienopyridine at any time, any dose, up to time of PCI Interaction P values = 0. 17, 0. 19 and 0. 65 respectively RR [95%CI] 1. 37 (1. 00 -1. 88) RR [95%CI] 0. 61 (0. 39 -0. 97) Not Thienopyridine Exposed
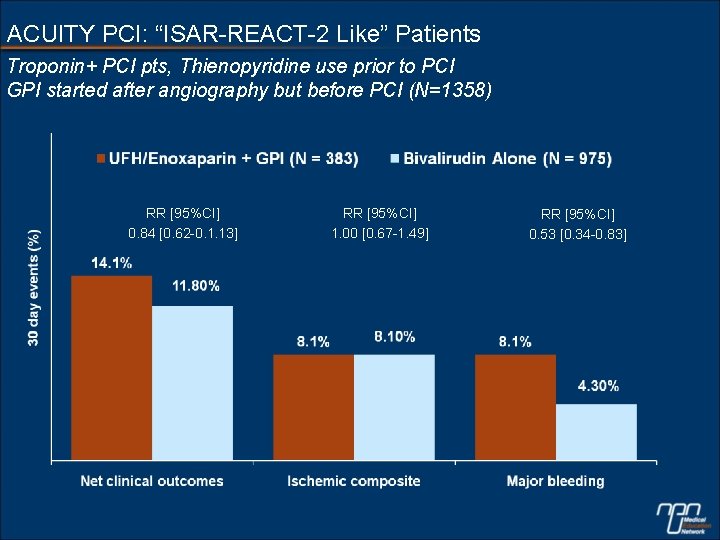
ACUITY PCI: “ISAR-REACT-2 Like” Patients Troponin+ PCI pts, Thienopyridine use prior to PCI GPI started after angiography but before PCI (N=1358) RR [95%CI] 0. 84 [0. 62 -0. 1. 13] RR [95%CI] 1. 00 [0. 67 -1. 49] RR [95%CI] 0. 53 [0. 34 -0. 83]

• Anticoagulation is recommended for all patients in addition to antiplatelet therapy (I-A) • Anticoagulation should be selected according to the risk of both ischaemic and bleeding events (I-B) • Several anticoagulants are available, namely UFH



Evidence-Based Risk Stratification to Target Therapies in UA/NSTEMI Aspirin, Clopidogrel, Fondaparinux (Class 1 A) β-blocker, Nitrates Higher Risk +Troponin, ST s, TRS 3, Recurrent Ischemia, CHF, Prior Revascularization Invasive Strategy PCI Bivalirudin or UFH (+IV GP IIb/IIIa) UFH dose 50 IU/kg Lower Risk – ECG, – Markers, TRS 0 -2 Recurrent Ischemia or high risk stress test No PCI Medical Rx or CABG (hold fonda and clopidogrel) Conservative Strategy

Summary • Fondaparinux reduces bleeding and mortality in patients with NSTEACS compared with enoxaparin including those undergoing early intervention (<24 hours) • Bivalirudin with a thienopyridine reduces bleeding compared with UFH/enox + GPI • Fondaparinux and bivalirudin are likely to be complementary— fondaparinux for initial upstream therapy of ACS and bivalirudin in place of UFH+GP IIb/IIIa in those undergoing PCI • This strategy has the potential to lead to the lowest rates of bleeding (and enhanced efficacy) we have ever seen in ACS
- Slides: 27