16 1 Which statement best defines reduction 1

16. 1 Which statement best defines reduction? 1. 2. 3. 4. 5. Tro IC. 3 The loss of protons The gain of oxygen atoms The loss of hydrogen atoms The gain of electrons Two of the above

16. 1 Which statement best defines reduction? 1. 2. 3. 4. 5. Tro IC. 3 The loss of protons The gain of oxygen atoms The loss of hydrogen atoms The gain of electrons Two of the above

16. 2 Which of the following statement best describes oxidation and reduction reactions? 1. An oxidation reaction must occur when a reduction reaction occurs. 2. The reactions occur only between two metals. 3. Half electrons are transferred in half reactions. 4. The reactions always involve oxygen. 5. All of the above Tro IC. 3

16. 2 Which of the following statement best describes oxidation and reduction reactions? 1. An oxidation reaction must occur when a reduction reaction occurs. 2. The reactions occur only between two metals. 3. Half electrons are transferred in half reactions. 4. The reactions always involve oxygen. 5. All of the above Tro IC. 3

16. 3 Tro IC. 3 A reducing agent: 1. Causes another substance to gain electrons 2. Causes another substance to gain oxygen atoms 3. Causes another substance to gain hydrogen atoms 4. Causes another substance to lose electrons 5. Two of the above

16. 3 Tro IC. 3 A reducing agent: 1. Causes another substance to gain electrons 2. Causes another substance to gain oxygen atoms 3. Causes another substance to gain hydrogen atoms 4. Causes another substance to lose electrons 5. Two of the above

16. 4 In the following reaction: Fe(s) + Cu 2+(aq) → Fe 2+(aq) + Cu(s) iron is: 1. 2. 3. 4. 5. Tro IC. 3 Oxidized Reduced Synthesized Neutralized None of the above

16. 4 In the following reaction: Fe(s) + Cu 2+(aq) → Fe 2+(aq) + Cu(s) iron is: 1. 2. 3. 4. 5. Tro IC. 3 Oxidized Reduced Synthesized Neutralized None of the above

16. 5 In the following reaction: Cu(s) + 2 Ag. NO 3(aq) → Cu(NO 3)2(aq) + 2 Ag(s) which species is reduced? 1. 2. 3. 4. 5. Tro IC. 3 Cu Ag 1+ NO 31– Cu 2+ Ag

16. 5 In the following reaction: Cu(s) + 2 Ag. NO 3(aq) → Cu(NO 3)2(aq) + 2 Ag(s) which species is reduced? 1. 2. 3. 4. 5. Tro IC. 3 Cu Ag 1+ NO 31– Cu 2+ Ag

16. 6 In the following reaction: 3 Mg(s) + N 2(g) → Mg 3 N 2(s) nitrogen is the: 1. 2. 3. 4. 5. Tro IC. 3 Oxidizing agent Redox agent Reducing agent Neutralizing agent None of the above

16. 6 In the following reaction: 3 Mg(s) + N 2(g) → Mg 3 N 2(s) nitrogen is the: 1. 2. 3. 4. 5. Tro IC. 3 Oxidizing agent Redox agent Reducing agent Neutralizing agent None of the above

16. 7 In the following reaction: Fe 2 O 3(s) + 3 CO(g) → 2 Fe(s) + 3 CO 2(g) the reducing agent is: 1. 2. 3. 4. 5. Tro IC. 3 Fe Fe 2+ CO Fe 2 O 3 There is none.

16. 7 In the following reaction: Fe 2 O 3(s) + 3 CO(g) → 2 Fe(s) + 3 CO 2(g) the reducing agent is: 1. 2. 3. 4. 5. Tro IC. 3 Fe Fe 2+ CO Fe 2 O 3 There is none.

16. 8 Which of the following is an example of a redox reaction? 1. 2. 3. 4. 5. Tro IC. 3 KOH(aq) + HCl(aq) → H 2 O(l) + KCl(aq) C 3 H 8(g) + 5 O 2(g) → 4 H 2 O(g) + 3 CO 2(g) Ag. NO 3(aq) + Na. Cl(aq) → Ag. Cl(s) + Na. NO 3(aq) All of the above Two of the above

16. 8 Which of the following is an example of a redox reaction? 1. 2. 3. 4. 5. Tro IC. 3 KOH(aq) + HCl(aq) → H 2 O(l) + KCl(aq) C 3 H 8(g) + 5 O 2(g) → 4 H 2 O(g) + 3 CO 2(g) Ag. NO 3(aq) + Na. Cl(aq) → Ag. Cl(s) + Na. NO 3(aq) All of the above Two of the above

16. 9 The oxidation state of S in S 8 is: 1. 2. 3. 4. 5. Tro IC. 3 -2 -1 0 +2 -8

16. 9 The oxidation state of S in S 8 is: 1. 2. 3. 4. 5. Tro IC. 3 -2 -1 0 +2 -8

16. 10 The oxidation state of copper in Cu. Br 2 is: 1. -2 2. -1 3. 0 4. +1 5. +2 Tro IC. 3
16. 10 The oxidation state of copper in Cu. Br 2 is: 1. -2 2. -1 3. 0 4. +1 5. +2 Tro IC. 3

16. 11 The sum of the oxidation states for all of the atoms in HCl. O 4 is: 1. 3 2. 6 3. 0 4. -7 5. -8 Tro IC. 3

16. 11 The sum of the oxidation states for all of the atoms in HCl. O 4 is: 1. 3 2. 6 3. 0 4. -7 5. -8 Tro IC. 3

16. 12 What is the oxidation state of Sb in H 2 Sb. O 31– 1. 2. 3. 4. 5. Tro IC. 3 -1 +5 +3 +4 -3

16. 12 What is the oxidation state of Sb in H 2 Sb. O 31– 1. 2. 3. 4. 5. Tro IC. 3 -1 +5 +3 +4 -3

16. 13 What are the oxidation states of oxygen in H 2 O 2 and OH 1–, respectively? 1. 2. 3. 4. 5. Tro IC. 3 0 and 0 -1 and -1 -4 and -2 0 and -1 -1 and -2

16. 13 What are the oxidation states of oxygen in H 2 O 2 and OH 1–, respectively? 1. 2. 3. 4. 5. Tro IC. 3 0 and 0 -1 and -1 -4 and -2 0 and -1 -1 and -2

16. 20 Tro IC. 3 The following is an activity series with the most reactive metal at the top: Mg(s) → Mg 2+(aq) + 2 e– Zn(s) → Zn 2+(aq) + 2 e– H 2(g) → 2 H 1+(aq) + 2 e– Au(s) → Au 3+(aq) + 3 e– Which reaction in this list is NOT spontaneous? 1. 2. 3. 4. 5. Au(s) + Mg 2+(aq) → Mg(s) + Zn 2+(aq) → Zn(s) + Au 3+(aq) → Mg(s) + 2 H 1+(aq) → All of the above

16. 20 Tro IC. 3 The following is an activity series with the most reactive metal at the top: Mg(s) → Mg 2+(aq) + 2 e– Zn(s) → Zn 2+(aq) + 2 e– H 2(g) → 2 H 1+(aq) + 2 e– Au(s) → Au 3+(aq) + 3 e– Which reaction in this list is NOT spontaneous? 1. 2. 3. 4. 5. Au(s) + Mg 2+(aq) → Mg(s) + Zn 2+(aq) → Zn(s) + Au 3+(aq) → Mg(s) + 2 H 1+(aq) → All of the above
16. 21 Tro IC. 3 Based on the following activity series, with the most active metal at the top: Mg(s) → Mg 2+(aq) + 2 e– Zn(s) → Zn 2+(aq) + 2 e– H 2(g) → 2 H 1+(aq) + 2 e– Au(s) → Au 3+(aq) + 3 e– which metal in the list below will dissolve in hydroiodic acid, a strong acid? 1. 2. 3. 4. 5. Mg Zn Au All of the above Two of the above

16. 21 Tro IC. 3 Based on the following activity series, with the most active metal at the top: Mg(s) → Mg 2+(aq) + 2 e– Zn(s) → Zn 2+(aq) + 2 e– H 2(g) → 2 H 1+(aq) + 2 e– Au(s) → Au 3+(aq) + 3 e– which metal in the list below will dissolve in hydroiodic acid, a strong acid? 1. Mg 2. Zn 3. Au 4. All of the above 5. Two of the above

16. 22 Tro IC. 3 Based on the following activity series, with the most active metal at the top: Mg(s) → Mg 2+(aq) + 2 e– Zn(s) → Zn 2+(aq) + 2 e– H 2(g) → 2 H+(aq) + 2 e– Au(s) → Au 3+(aq) + 3 e– which metal listed is the most resistant to corrosion? 1. 2. 3. 4. 5. Mg Zn Au Mg 2+ Au 3+
16. 22 Tro IC. 3 Based on the following activity series, with the most active metal at the top: Mg(s) → Mg 2+(aq) + 2 e– Zn(s) → Zn 2+(aq) + 2 e– H 2(g) → 2 H+(aq) + 2 e– Au(s) → Au 3+(aq) + 3 e– which metal listed is the most resistant to corrosion? 1. Mg 2. Zn 3. Au 4. Mg 2+ 5. Au 3+

16. 23 The usual electric current in a wire is the flow of: 1. 2. 3. 4. 5. Tro IC. 3 Protons Electrons Neutrons Quarks Alpha particles

16. 23 The usual electric current in a wire is the flow of: 1. 2. 3. 4. 5. Tro IC. 3 Protons Electrons Neutrons Quarks Alpha particles
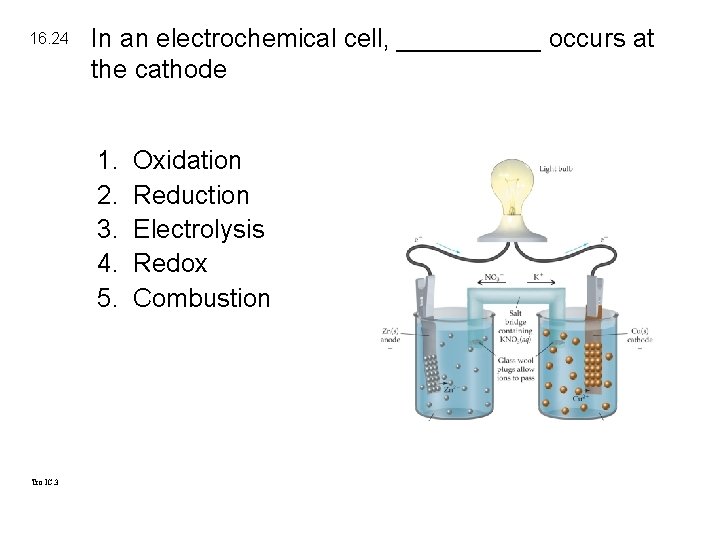
16. 24 In an electrochemical cell, _____ occurs at the cathode 1. 2. 3. 4. 5. Tro IC. 3 Oxidation Reduction Electrolysis Redox Combustion
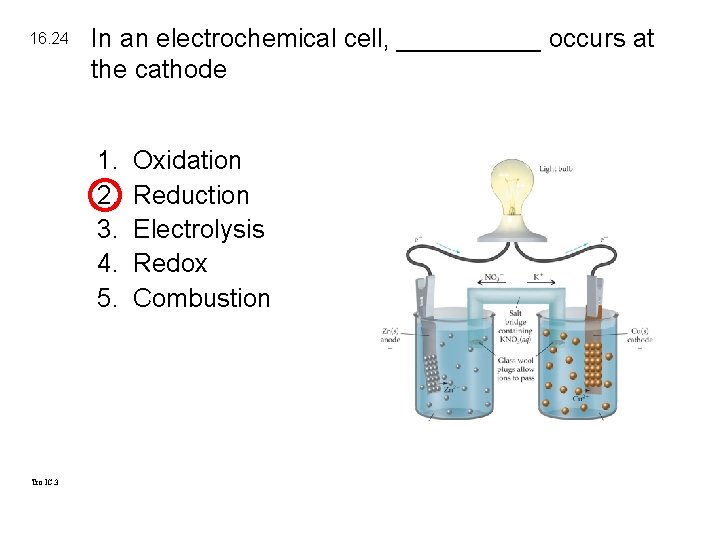
16. 24 In an electrochemical cell, _____ occurs at the cathode 1. 2. 3. 4. 5. Tro IC. 3 Oxidation Reduction Electrolysis Redox Combustion
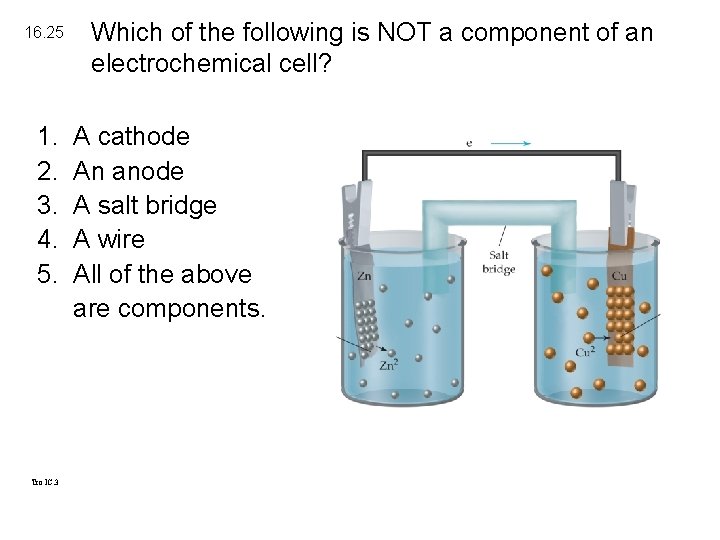
16. 25 1. 2. 3. 4. 5. Tro IC. 3 Which of the following is NOT a component of an electrochemical cell? A cathode An anode A salt bridge A wire All of the above are components.
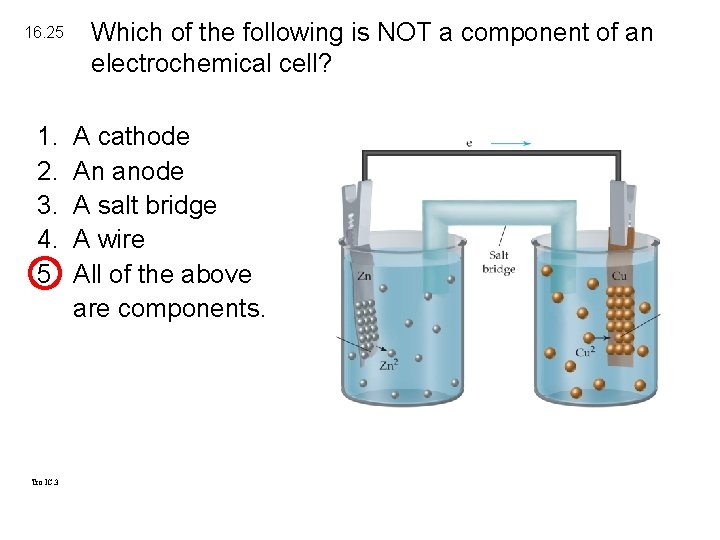
16. 25 1. 2. 3. 4. 5. Tro IC. 3 Which of the following is NOT a component of an electrochemical cell? A cathode An anode A salt bridge A wire All of the above are components.
- Slides: 38