16 1 Carboxylic Acids A carboxylic acid contains
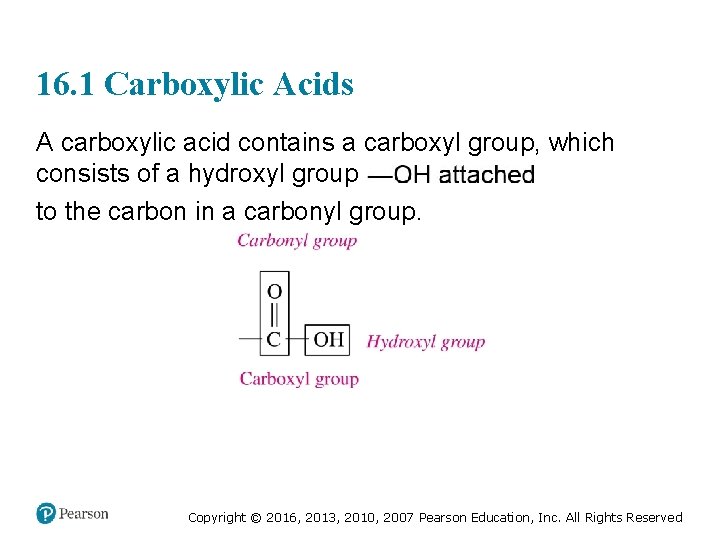
16. 1 Carboxylic Acids A carboxylic acid contains a carboxyl group, which consists of a hydroxyl group to the carbon in a carbonyl group. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chapter 16 - Objectives • Define and recognize carboxylic acids and describe their charactersitics and properties • Define and recognize esters and describe their charactersitics and properties Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
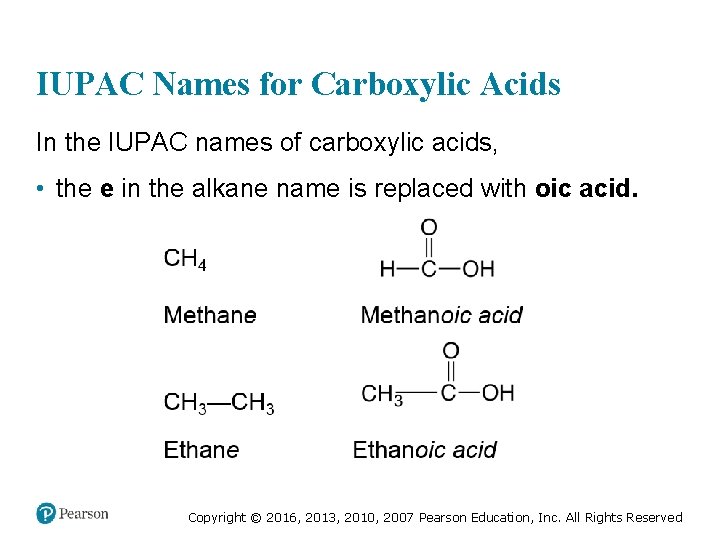
IUPAC Names for Carboxylic Acids In the IUPAC names of carboxylic acids, • the e in the alkane name is replaced with oic acid. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Common Carboxylic Acids A red ant sting contains methanoic acid (formic acid) that irritates the skin. The sour taste of vinegar is due to ethanoic acid (acetic acid). Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
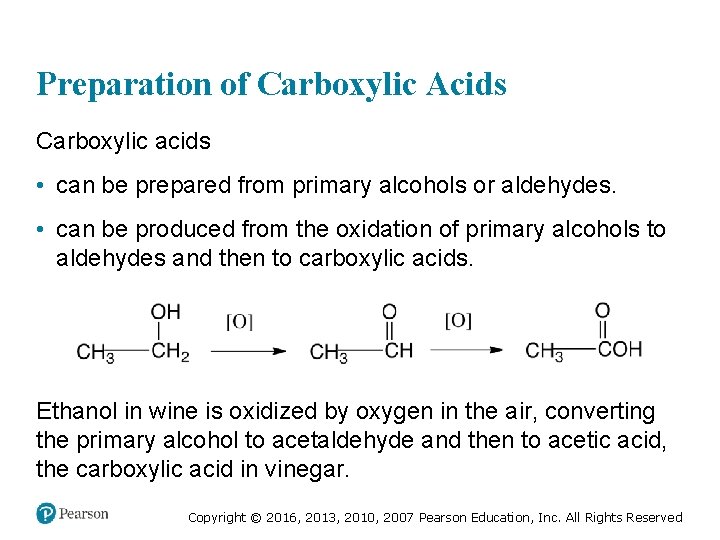
Preparation of Carboxylic Acids Carboxylic acids • can be prepared from primary alcohols or aldehydes. • can be produced from the oxidation of primary alcohols to aldehydes and then to carboxylic acids. Ethanol in wine is oxidized by oxygen in the air, converting the primary alcohol to acetaldehyde and then to acetic acid, the carboxylic acid in vinegar. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Health: Alpha Hydroxy Acids Alpha hydroxy acids (AHAs) • are found in fruits, milk, and sugar cane. • are naturally occurring carboxylic acids with a hydroxyl group on the carbon atom that is adjacent to the carboxyl group. Dermatologists have been using products with high concentrations (20– 70%) of AHAs to remove acne scars and in skin peels to reduce irregular pigmentation and age spots. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Health: Alpha Hydroxy Acids Several different alpha hydroxy acids may be found in skin care products singly or in combination. Alpha Hydroxy Acid (Source) Condensed Structural Formula Glycolic acid (sugar cane) carboxyl group bonded to C H 2 bonded to an O H group. Lactic acid (sour milk) carboxyl group bonded to C H bonded to a C H 3 group and an O H group. Tartaric acid (grapes) 2 carboxyl groups, one on each end, bonded to 2 carbons, each bonded to an O H group Malic acid (Apples) 2 carboxyl groups, one on each end, bonded to 2 carbons, one bonded to an O H group Citric acid (citrus fruits) central carbon bonded to an O H group, a carboxyl group, and 2 C H 2 groups each bonded to a carboxyl group. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

16. 2 Properties of Carboxylic Acids Carboxylate salts are often used as preservatives and flavor enhancers in soups and seasonings. Sodium propionate, a preservative, is added to cheeses, bread, and other bakery items to inhibit the spoilage of the food by microorganisms. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
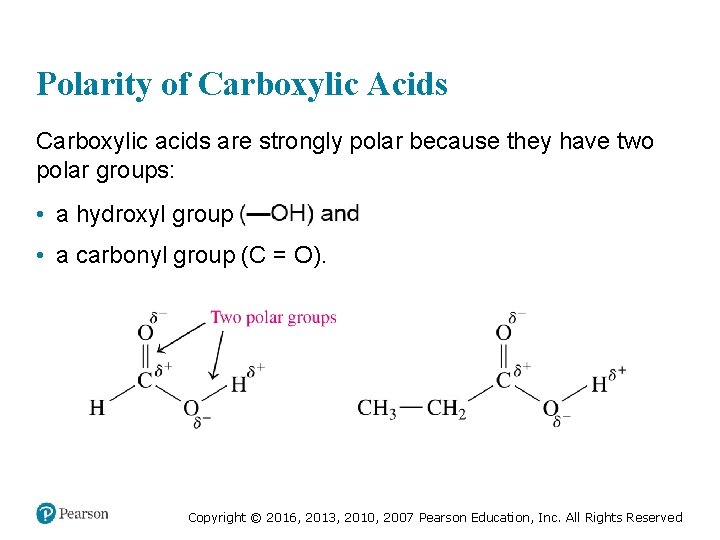
Polarity of Carboxylic Acids Carboxylic acids are strongly polar because they have two polar groups: • a hydroxyl group • a carbonyl group (C = O). Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
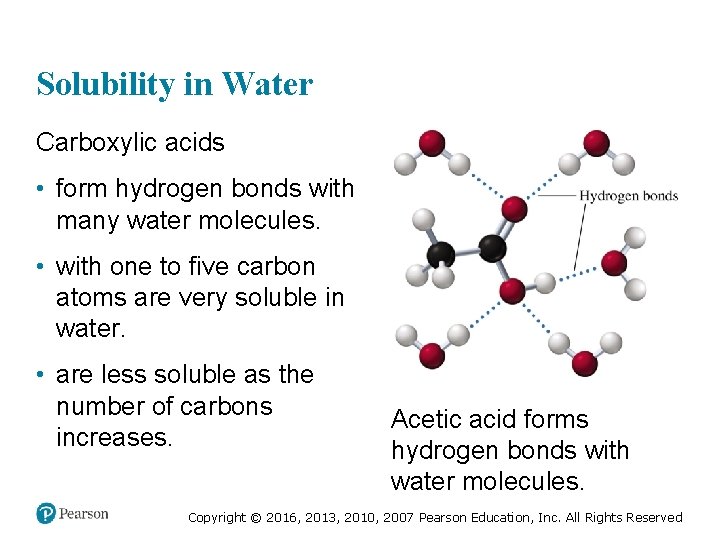
Solubility in Water Carboxylic acids • form hydrogen bonds with many water molecules. • with one to five carbon atoms are very soluble in water. • are less soluble as the number of carbons increases. Acetic acid forms hydrogen bonds with water molecules. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Acidity of Carboxylic Acids Carboxylic acids • are weak acids. • dissociate in water to produce carboxylate ions and hydronium ions. • can lose a proton because two oxygen atoms in a carboxylate ion stabilize negative charge. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
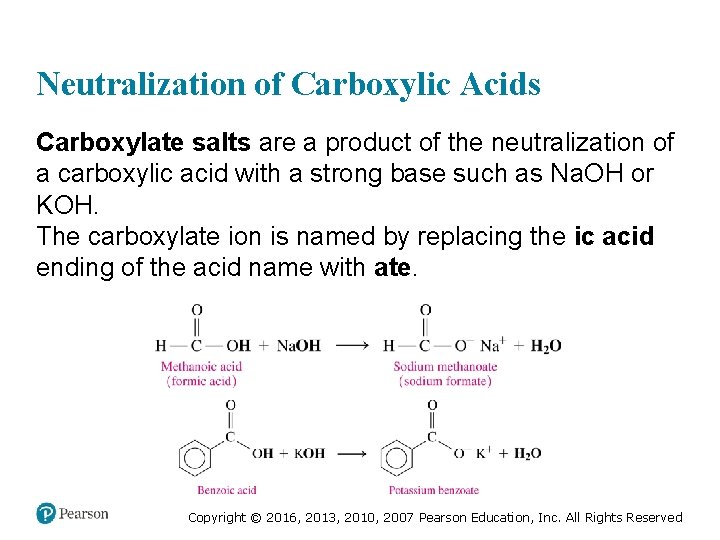
Neutralization of Carboxylic Acids Carboxylate salts are a product of the neutralization of a carboxylic acid with a strong base such as Na. OH or KOH. The carboxylate ion is named by replacing the ic acid ending of the acid name with ate. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
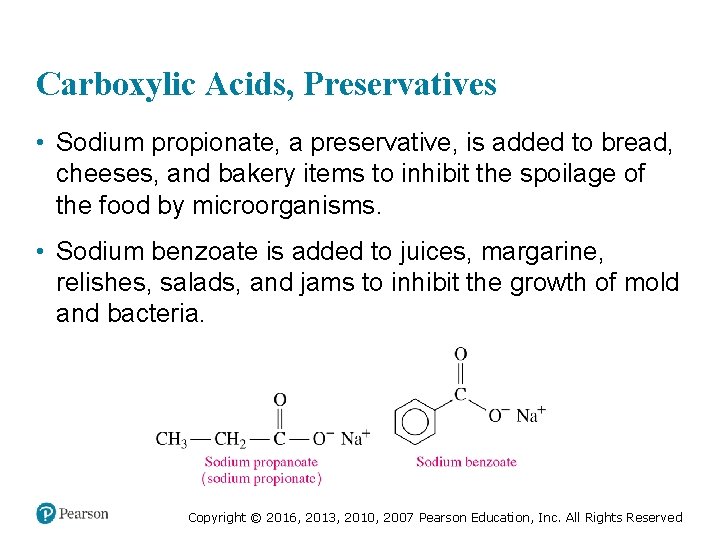
Carboxylic Acids, Preservatives • Sodium propionate, a preservative, is added to bread, cheeses, and bakery items to inhibit the spoilage of the food by microorganisms. • Sodium benzoate is added to juices, margarine, relishes, salads, and jams to inhibit the growth of mold and bacteria. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Carboxylic Acids, Preservatives • Monosodium glutamate (MSG) is added to meats, fish, vegetables, and bakery items to enhance flavor, although it may cause headaches in some people. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Health: Carboxylic Acids in Metabolism At the p. H of the aqueous environment in the cells, • the carboxylic acids are dissociated. • it is actually the carboxylate ions that take part in the reactions of the citric acid cycle. For example, in water, succinic acid is in equilibrium with its carboxylate ion, succinate. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Health: Carboxylic Acids in Metabolism Carboxylates are part of the metabolic processes within our cells. For example, • during glycolysis, a molecule of glucose is broken down into two molecules of pyruvate, the carboxylate salt of pyruvic acid. • during strenuous exercise when oxygen levels are low (anaerobic), pyruvate is reduced to give lactic acid or the lactate ion. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Health: Carboxylic Acids in Metabolism In the citric acid cycle (Krebs cycle), • di- and tricarboxylates are oxidized and decarboxylated (loss of CO 2) to produce energy for cells. • citrate, with six carbons, is oxidized to a five-carbon Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Health: Carboxylic Acids in Metabolism The citric acid cycle continues as loses CO 2 to give a four-carbon succinate. • • a series of reactions then converts succinate to oxaloacetate. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

16. 3 Esters By the 1800 s, chemists had discovered that • salicin from willow tree bark and leaves was responsible for pain relief. • the body converts salicin to salicylic acid, which irritates the stomach lining. Bayer discovered that an ester form of the compound, acetylsalicylic acid, is less irritating but still effective. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
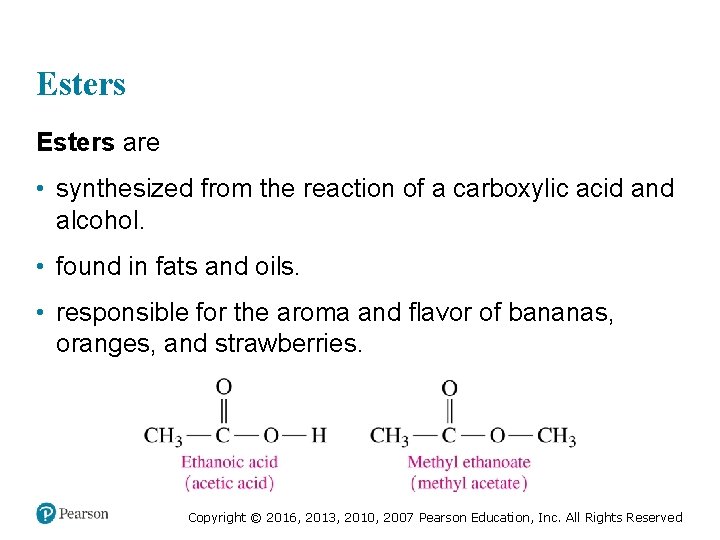
Esters are • synthesized from the reaction of a carboxylic acid and alcohol. • found in fats and oils. • responsible for the aroma and flavor of bananas, oranges, and strawberries. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
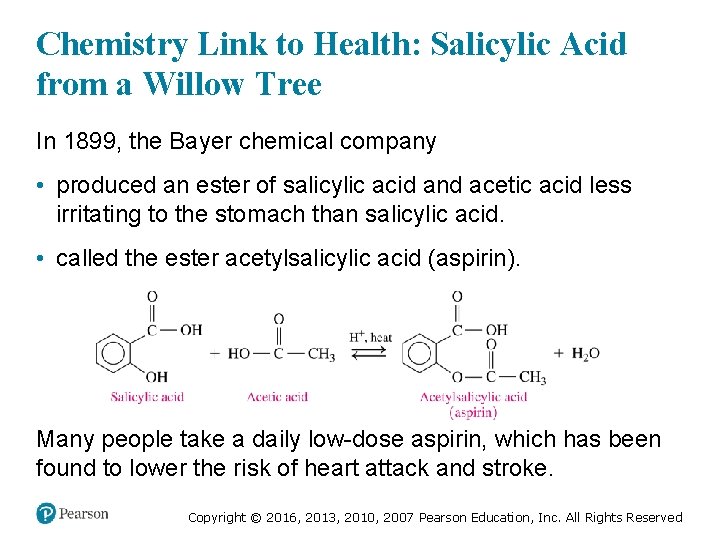
Chemistry Link to Health: Salicylic Acid from a Willow Tree In 1899, the Bayer chemical company • produced an ester of salicylic acid and acetic acid less irritating to the stomach than salicylic acid. • called the ester acetylsalicylic acid (aspirin). Many people take a daily low-dose aspirin, which has been found to lower the risk of heart attack and stroke. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
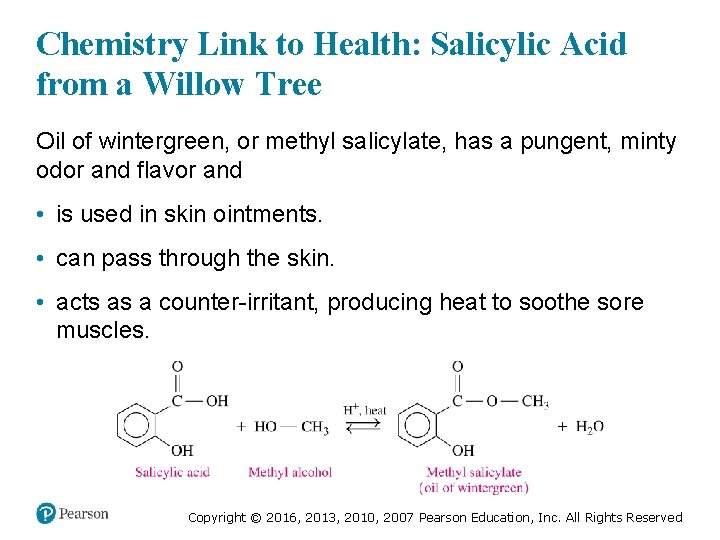
Chemistry Link to Health: Salicylic Acid from a Willow Tree Oil of wintergreen, or methyl salicylate, has a pungent, minty odor and flavor and • is used in skin ointments. • can pass through the skin. • acts as a counter-irritant, producing heat to soothe sore muscles. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Chemistry Link to Environment: Plastics Dacron has many uses; it • forms a permanent-press fabric that resists wrinkles. • is used to make artificial blood vessels and valves that are biologically inert and does not clot blood. • Is made into a film called Mylar and a plastic known as PETE (polyethyleneterephthalate) used for plastic drink bottles. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved

Esters in Fruits • Many of the fragrances of perfumes and flowers and the flavors of fruits are due to esters. • Small esters are volatile, so we can smell them, and they are soluble in water, so we can taste them. • The odor of grapes is due to ethyl heptanoate. Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
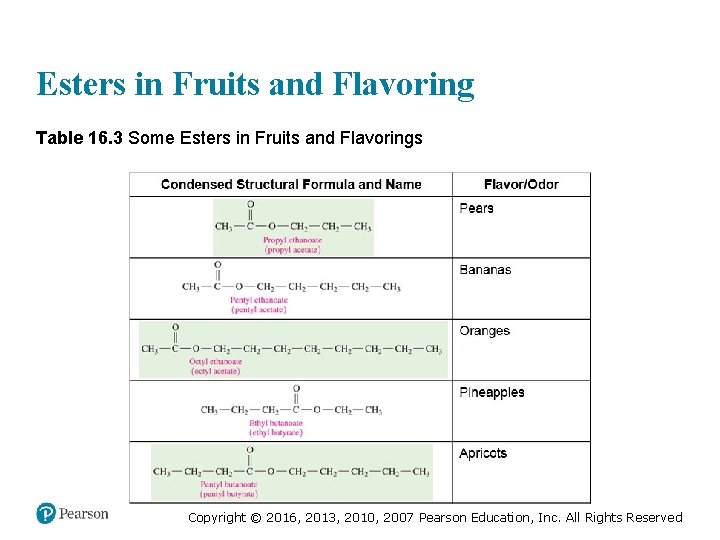
Esters in Fruits and Flavoring Table 16. 3 Some Esters in Fruits and Flavorings Copyright © 2016, 2013, 2010, 2007 Pearson Education, Inc. All Rights Reserved
- Slides: 25