15 Benzene and Aromaticity Aromatic Compounds n Benzene
15. Benzene and Aromaticity

Aromatic Compounds n Benzene is the parent of the family of aromatic compounds. Its six carbons lie in a plane in a hexagon, each carbon has one hydrogen attached. Benzene is a resonance hybrid of two Kekulé structures. Note: It’s okay to use circle representation if explicitly showing π electrons is not necessary, since circle doesn’t indicate number of π electrons in ring 2

Aromatic Compounds n In orbital terms, each carbon is sp 2 -hybridized. These orbitals form σ bonds to hydrogen and the two neighboring carbons are all in the ring plane. n A p orbital at each carbon is perpendicular to this plane, and the six electrons, one from each carbon, form an electron cloud of π bonds, which lie above and below the plane. n The bond angles in benzene are 120. All C C bond distances are equal (1. 39 Å). The compound is more stable than either of the contributing Kekulé structures and has a resonance or stabilization energy of about 36 kcal/mol. 3

Aromatic Compounds n The origin of the term aromatic comes from the description of fragrant substances such as benzaldehyde present in cherries, peaches, and almonds. Benzaldehyde n Today aromatic refers to benzene and its structural relatives. Many compounds synthetic and isolated from nature aromatic in part, including estrone, morphine and Valium. 4

15. 1 Sources of Aromatic Hydrocarbons n Coal and petroleum are two main sources for simple aromatic compounds. n Coal has an array of benzene like rings joined together and some compounds that coal tar yields are benzene, toluene, naphthalene, and xylene. n Petroleum yields mainly alkanes and only a few aromatic compounds. 5
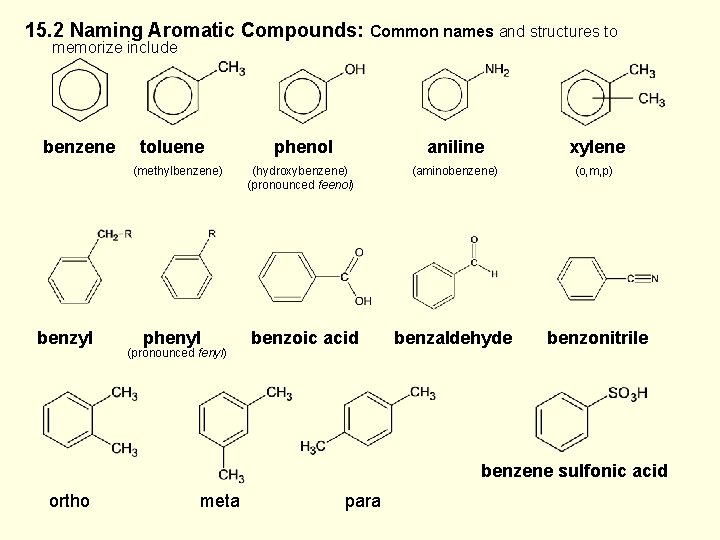
15. 2 Naming Aromatic Compounds: Common names and structures to memorize include benzene toluene (methylbenzene) benzyl phenyl (pronounced fenyl) phenol aniline xylene (hydroxybenzene) (pronounced feenol) (aminobenzene) (o, m, p) benzoic acid benzaldehyde benzonitrile benzene sulfonic acid ortho meta para
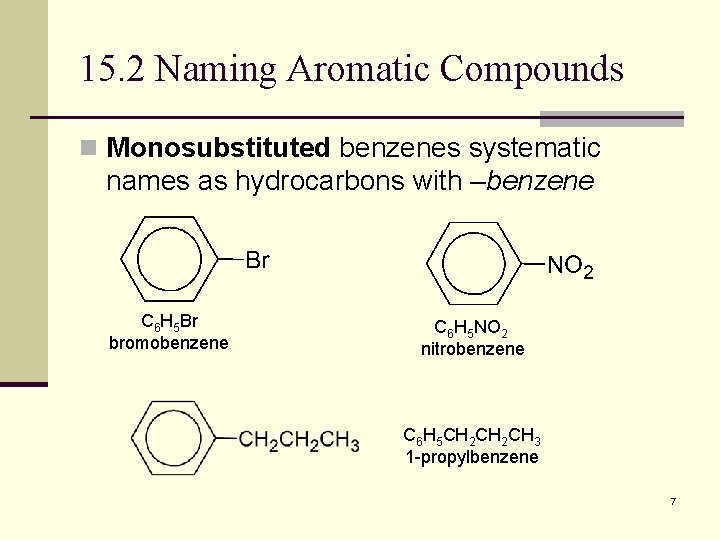
15. 2 Naming Aromatic Compounds n Monosubstituted benzenes systematic names as hydrocarbons with –benzene C 6 H 5 Br bromobenzene C 6 H 5 NO 2 nitrobenzene C 6 H 5 CH 2 CH 3 1 -propylbenzene 7
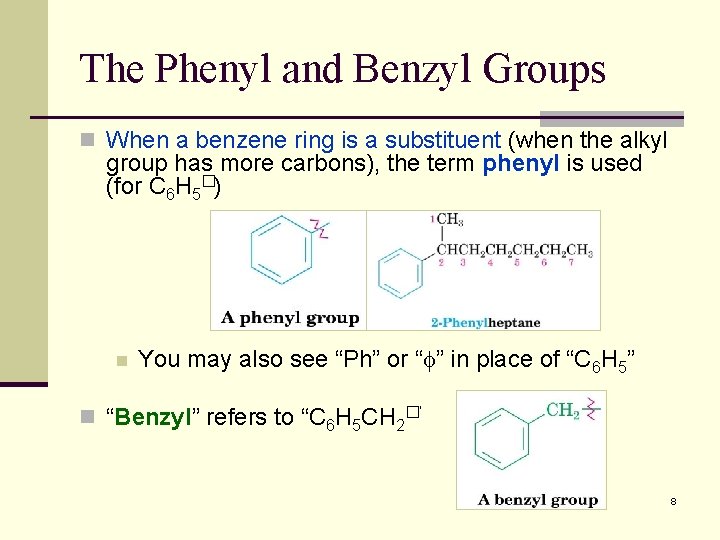
The Phenyl and Benzyl Groups n When a benzene ring is a substituent (when the alkyl group has more carbons), the term phenyl is used (for C 6 H 5�) n You may also see “Ph” or “f” in place of “C 6 H 5” n “Benzyl” refers to “C 6 H 5 CH 2�” 8
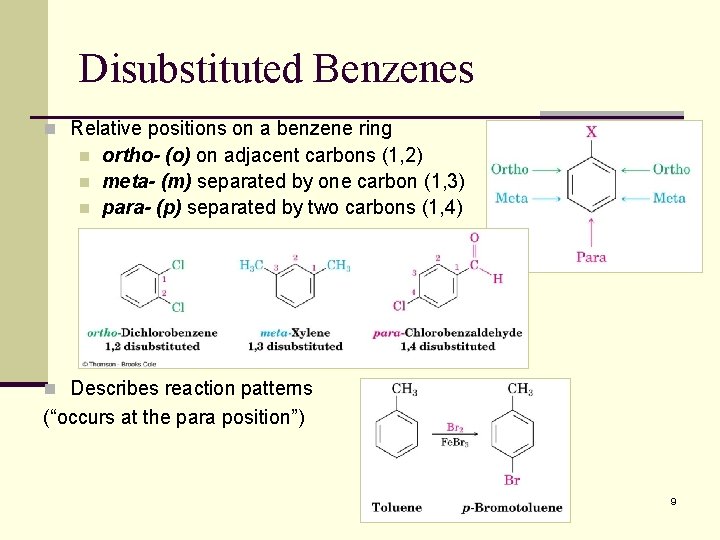
Disubstituted Benzenes n Relative positions on a benzene ring n n n ortho- (o) on adjacent carbons (1, 2) meta- (m) separated by one carbon (1, 3) para- (p) separated by two carbons (1, 4) n Describes reaction patterns (“occurs at the para position”) 9
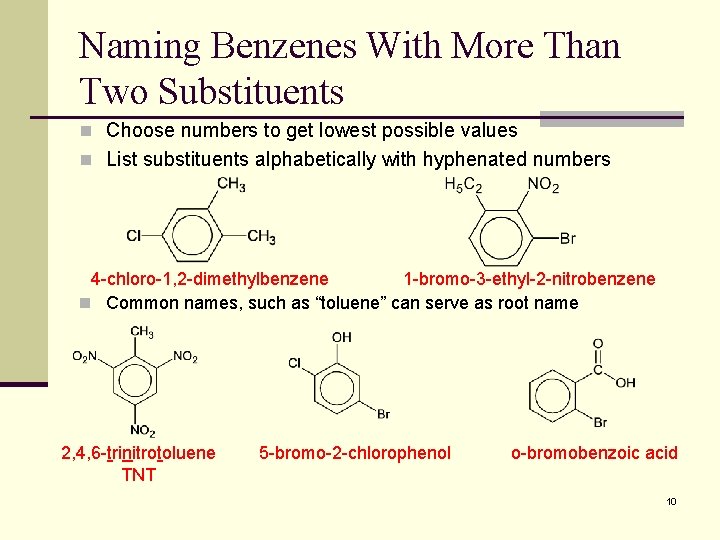
Naming Benzenes With More Than Two Substituents n Choose numbers to get lowest possible values n List substituents alphabetically with hyphenated numbers 4 -chloro-1, 2 -dimethylbenzene 1 -bromo-3 -ethyl-2 -nitrobenzene n Common names, such as “toluene” can serve as root name 2, 4, 6 -trinitrotoluene TNT 5 -bromo-2 -chlorophenol o-bromobenzoic acid 10
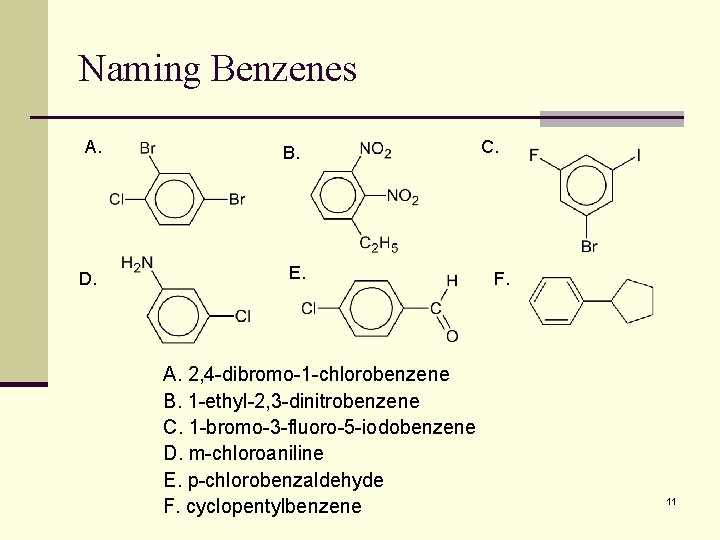
Naming Benzenes A. D. B. E. A. 2, 4 -dibromo-1 -chlorobenzene B. 1 -ethyl-2, 3 -dinitrobenzene C. 1 -bromo-3 -fluoro-5 -iodobenzene D. m-chloroaniline E. p-chlorobenzaldehyde F. cyclopentylbenzene C. F. 11

15. 3 Structure and Stability of Benzene n Benzene is very stable it undergoes a slow substitution reaction, where the ring itself does not react. The addition product is not formed Evidence of Stability n Heat of hydration is 150 k. J/mol less than expected, so it is 150 k. J/mol more stable than expected. n All carbon-carbon bonds (double and single) are the same length. n Resonance 12
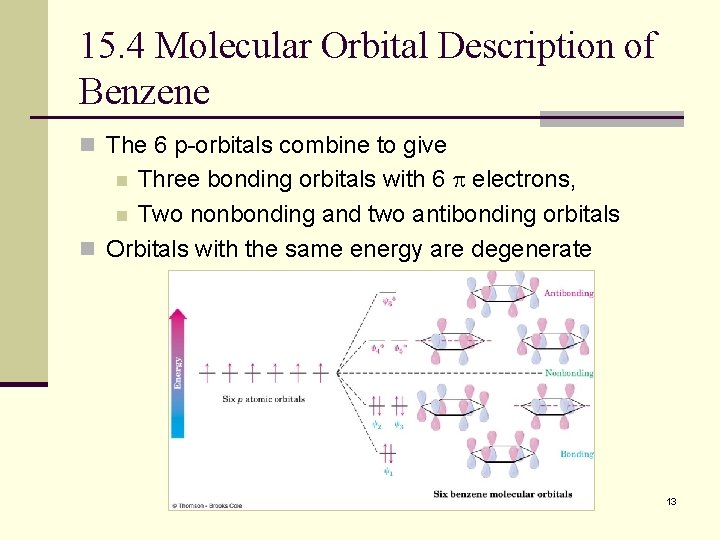
15. 4 Molecular Orbital Description of Benzene n The 6 p-orbitals combine to give Three bonding orbitals with 6 electrons, n Two nonbonding and two antibonding orbitals n Orbitals with the same energy are degenerate n 13

Aromaticity and the 4 n + 2 Rule n Huckel’s rule, based on calculations – a planar cyclic molecule with alternating double and single bonds has aromatic stability if it has 4 n+ 2 electrons (n is 0, 1, 2, 3, 4) Benzene Three double bonds Six π electrons n For benzene = 6 4 n+2 = 6, n = 1 and is aromatic 14

Compounds With 4 n Electrons Are Not Aromatic (May be Anti-aromatic) n Planar, cyclic molecules with 4 n electrons are much less stable than expected (anti-aromatic) n They will distort out of plane and behave like ordinary alkenes n 4 - and 8 -electron compounds are not delocalized (single and double bonds) n Cyclobutadiene is so unstable that it dimerizes by a self-Diels-Alder reaction at low temperature n Cyclooctatetraene has four double bonds, and will distort out of plane and behave like an ordinary alkene, it reacts with Br 2, KMn. O 4, and HCl as if it were four alkenes 15
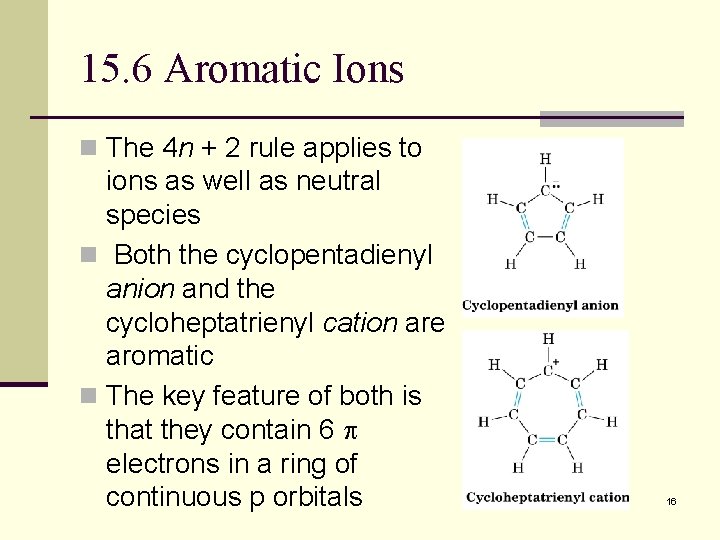
15. 6 Aromatic Ions n The 4 n + 2 rule applies to ions as well as neutral species n Both the cyclopentadienyl anion and the cycloheptatrienyl cation are aromatic n The key feature of both is that they contain 6 electrons in a ring of continuous p orbitals 16

15. 7 Aromatic Heterocycles: Pyridine and Pyrrole n Heterocyclic compounds contain elements other than carbon in a ring, such as N, S, O, P n Aromatic compounds can have elements other than carbon in the ring n. Pyridine: The nitrogen lone pair electrons are not part of the aromatic system (perpendicular orbital) n Pyrrole: Nitrogen atom is sp 2 - hybridized, and lone pair of electrons occupies a p orbital (6 electrons) 17

15. 8 Why 4 n +2? n When electrons fill the various molecular orbitals, it takes two electrons (one pair) to fill the lowest-lying orbital and four electrons (two pairs) to fill each of n succeeding energy level n This is a total of 4 n + 2 18

15. 9 Polycyclic Aromatic Compounds: Naphthalene n Aromatic compounds can have rings that share a set of carbon atoms (fused rings) n Compounds from fused benzene or aromatic heterocycle rings are themselves aromatic, if the follow Huckel’s rule 19
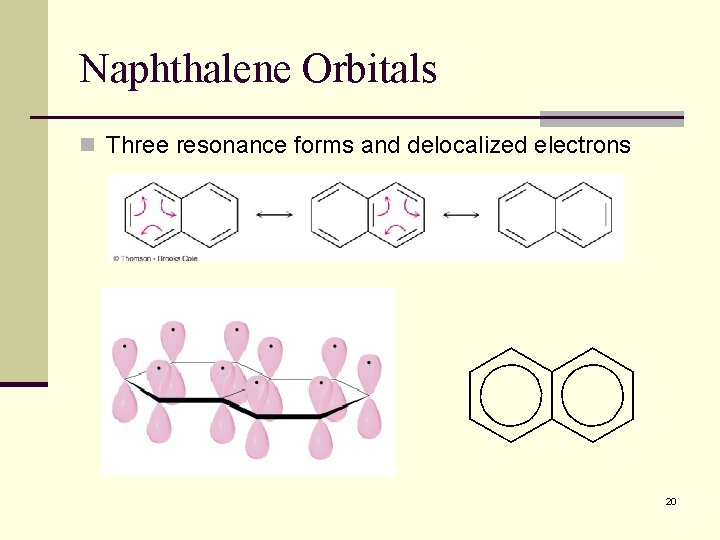
Naphthalene Orbitals n Three resonance forms and delocalized electrons 20
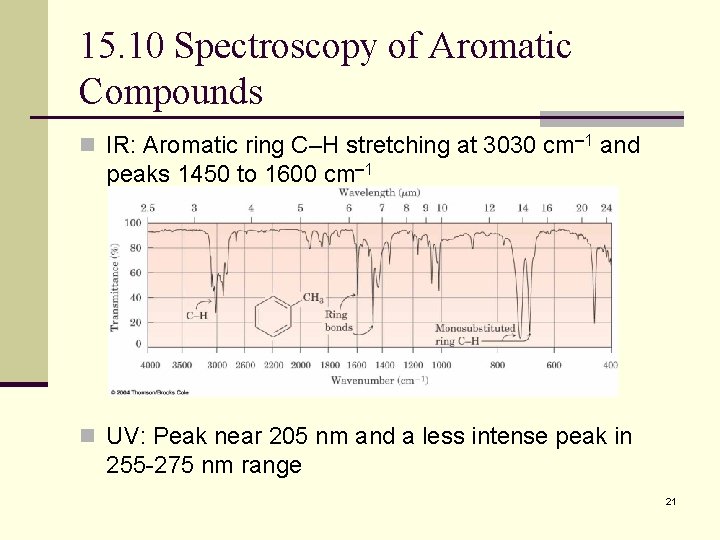
15. 10 Spectroscopy of Aromatic Compounds n IR: Aromatic ring C–H stretching at 3030 cm 1 and peaks 1450 to 1600 cm 1 n UV: Peak near 205 nm and a less intense peak in 255 -275 nm range 21
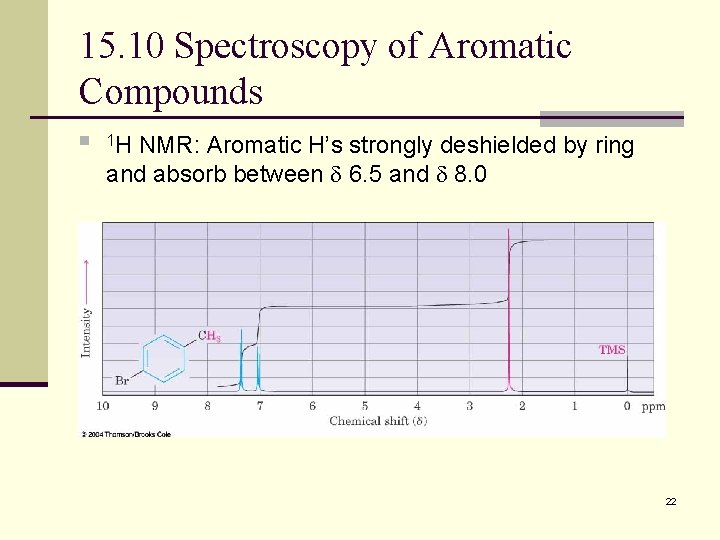
15. 10 Spectroscopy of Aromatic Compounds n 1 H NMR: Aromatic H’s strongly deshielded by ring and absorb between 6. 5 and 8. 0 22
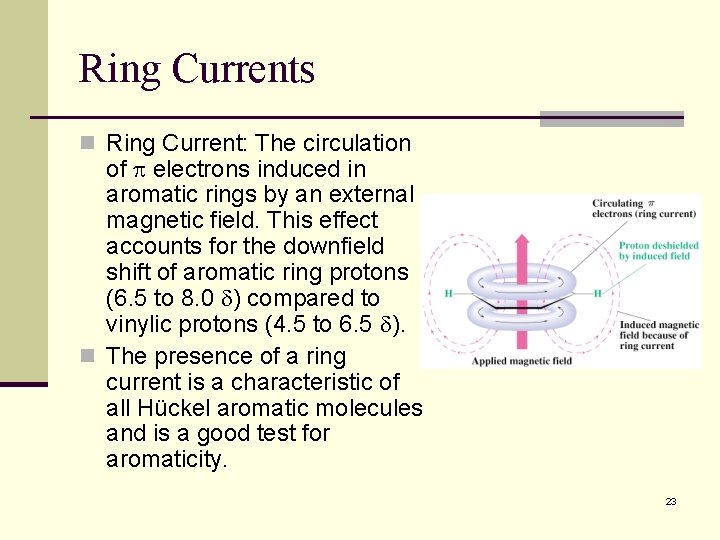
Ring Currents n Ring Current: The circulation of electrons induced in aromatic rings by an external magnetic field. This effect accounts for the downfield shift of aromatic ring protons (6. 5 to 8. 0 ) compared to vinylic protons (4. 5 to 6. 5 ). n The presence of a ring current is a characteristic of all Hückel aromatic molecules and is a good test for aromaticity. 23
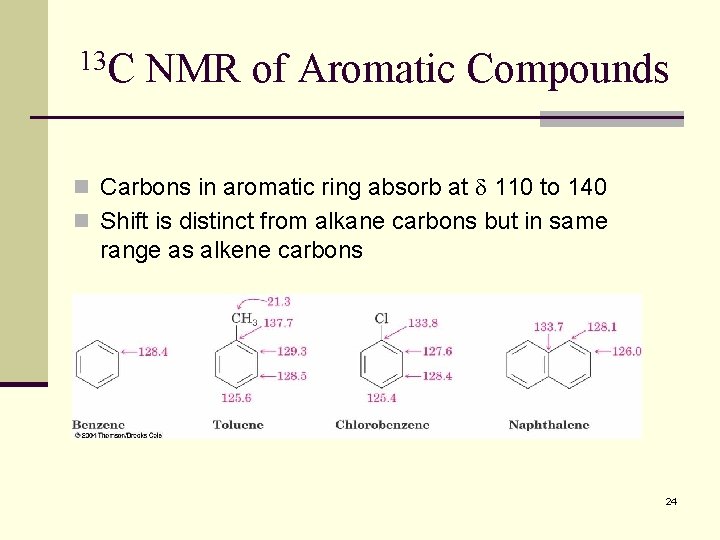
13 C NMR of Aromatic Compounds n Carbons in aromatic ring absorb at 110 to 140 n Shift is distinct from alkane carbons but in same range as alkene carbons 24
16. Chemistry of Benzene: Electrophilic Aromatic Substitution

Substitution Reactions of Benzene and Its Derivatives n Reactions of benzene lead to the retention of the aromatic core n Electrophilic aromatic substitution replaces a hydrogen on benzene with another electrophile n Typical reactions include chlorination, bromination, iodination, nitration, sulfonation, alkylation, and acylation ( the last two are Friedel. Crafts reactions) 26
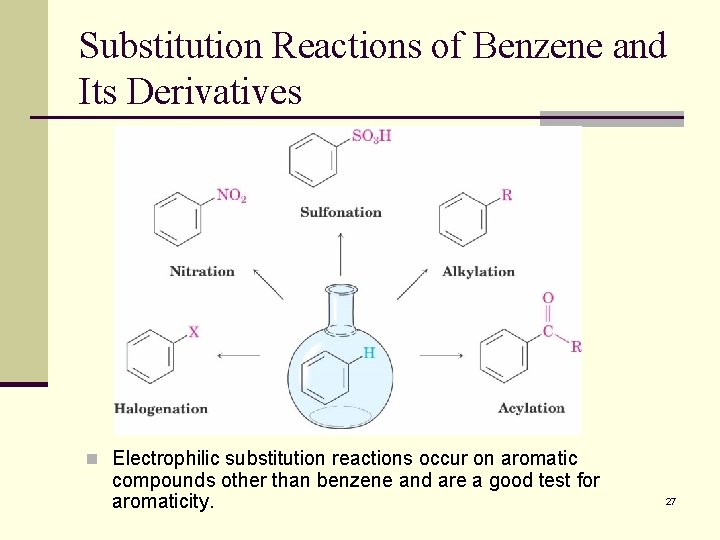
Substitution Reactions of Benzene and Its Derivatives n Electrophilic substitution reactions occur on aromatic compounds other than benzene and are a good test for aromaticity. 27
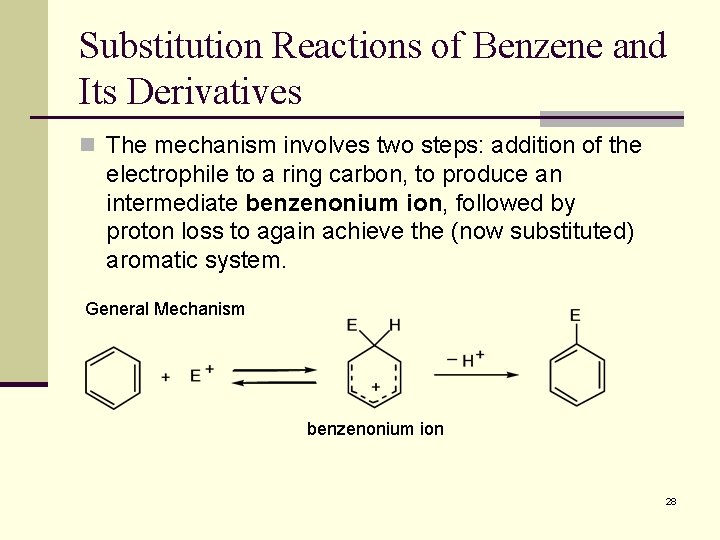
Substitution Reactions of Benzene and Its Derivatives n The mechanism involves two steps: addition of the electrophile to a ring carbon, to produce an intermediate benzenonium ion, followed by proton loss to again achieve the (now substituted) aromatic system. General Mechanism benzenonium ion 28
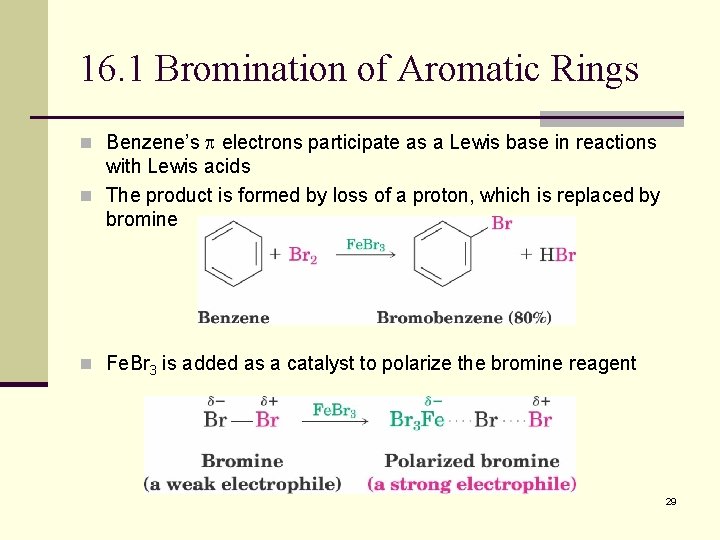
16. 1 Bromination of Aromatic Rings n Benzene’s electrons participate as a Lewis base in reactions with Lewis acids n The product is formed by loss of a proton, which is replaced by bromine n Fe. Br 3 is added as a catalyst to polarize the bromine reagent 29

Mechanism: Formation of Product from Intermediate n The addition of bromine occurs in two steps n In the first step the electrons act as a nucleophile toward Br 2 (in a complex with Fe. Br 3) n This forms a cationic addition intermediate from benzene and a bromine cation n The cationic addition intermediate transfers a proton to Fe. Br 4(from Br- and Fe. Br 3) n This restores aromaticity (in contrast with addition in alkenes) slow fast Aromatic rings are less reactive towards electrophiles than alkenes. Alkenes undergo addition reactions while aromatic compounds prefer to undergo substitution reactions. 30

16. 2 Other Aromatic Substitutions n The reaction with bromine involves a mechanism that is similar to many other reactions of benzene with electrophiles n The cationic intermediate was first proposed by G. W. Wheland of the University of Chicago and is often called the Wheland intermediate George Willard Wheland 1907 -1974 31
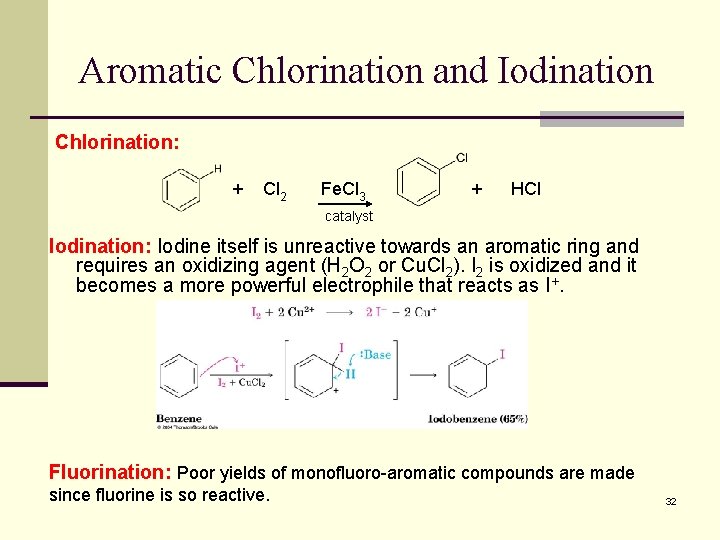
Aromatic Chlorination and Iodination Chlorination: + Cl 2 Fe. Cl 3 + HCl catalyst Iodination: Iodine itself is unreactive towards an aromatic ring and requires an oxidizing agent (H 2 O 2 or Cu. Cl 2). I 2 is oxidized and it becomes a more powerful electrophile that reacts as I+. Fluorination: Poor yields of monofluoro-aromatic compounds are made since fluorine is so reactive. 32
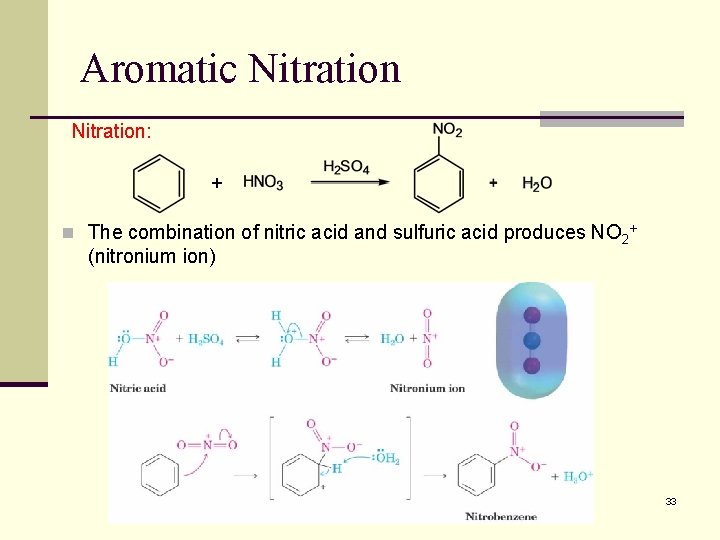
Aromatic Nitration: + n The combination of nitric acid and sulfuric acid produces NO 2+ (nitronium ion) 33
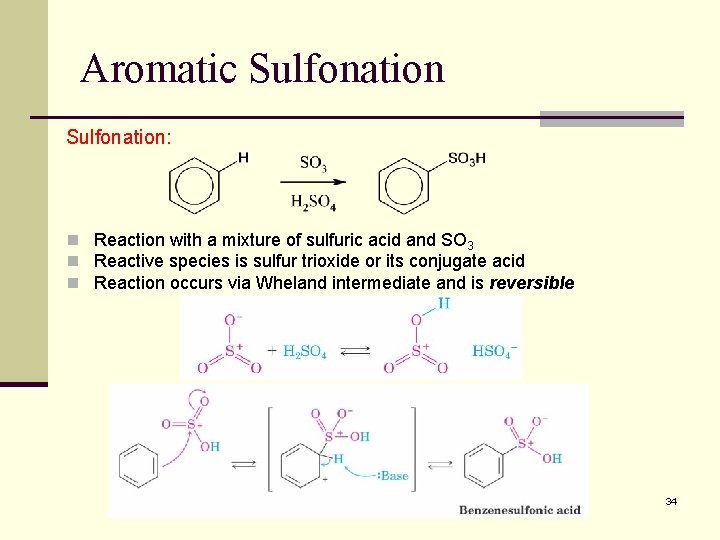
Aromatic Sulfonation: n Reaction with a mixture of sulfuric acid and SO 3 n Reactive species is sulfur trioxide or its conjugate acid n Reaction occurs via Wheland intermediate and is reversible 34

Alkali Fusion of Aromatic Sulfonic Acids n Sulfonic acids are useful as intermediates n Heating with Na. OH at 300 ºC followed by neutralization with acid replaces the SO 3 H group with an OH n Must be alkyl substituted (17. 10) n Example is the synthesis of p-cresol 35
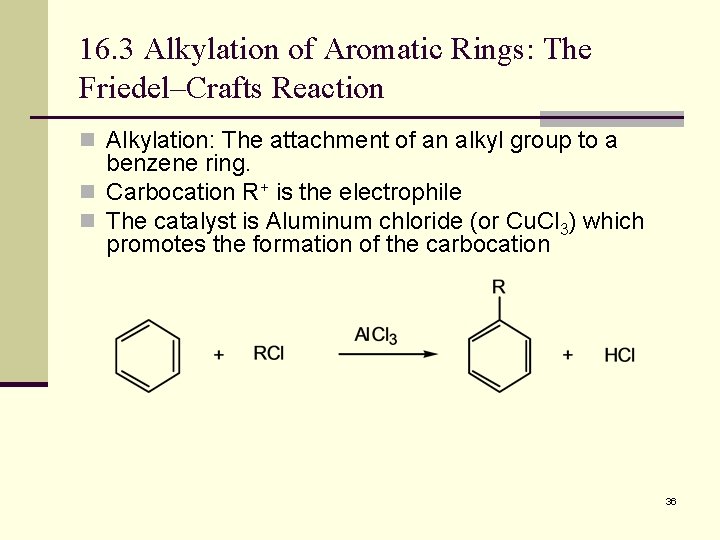
16. 3 Alkylation of Aromatic Rings: The Friedel–Crafts Reaction n Alkylation: The attachment of an alkyl group to a benzene ring. n Carbocation R+ is the electrophile n The catalyst is Aluminum chloride (or Cu. Cl 3) which promotes the formation of the carbocation 36
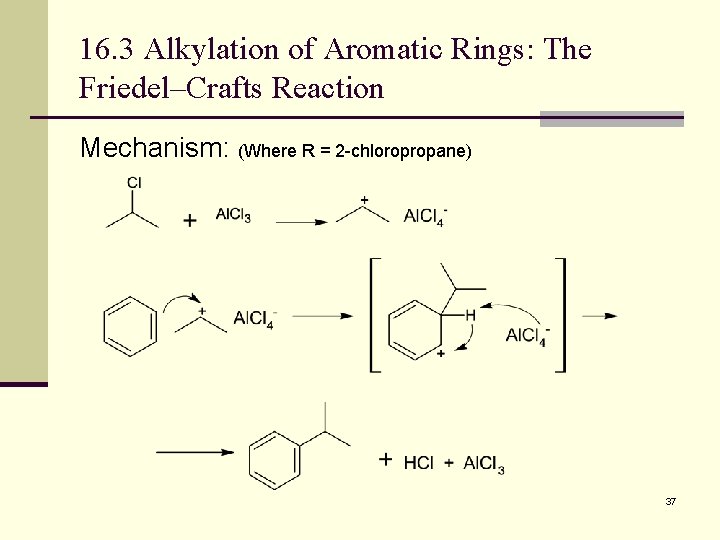
16. 3 Alkylation of Aromatic Rings: The Friedel–Crafts Reaction Mechanism: (Where R = 2 -chloropropane) 37

Limitations of the Friedel-Crafts Alkylation n n Only alkyl halides can be used (F, Cl, I, Br) Aryl halides and vinylic halides do not react (their carbocations are too hard to form) Will not work with rings containing an amino group substituent or a strongly electron-withdrawing group Often difficult to stop after a single substitution. Occasional skeletal rearrangement of alkyl group forming a mixture of products 38
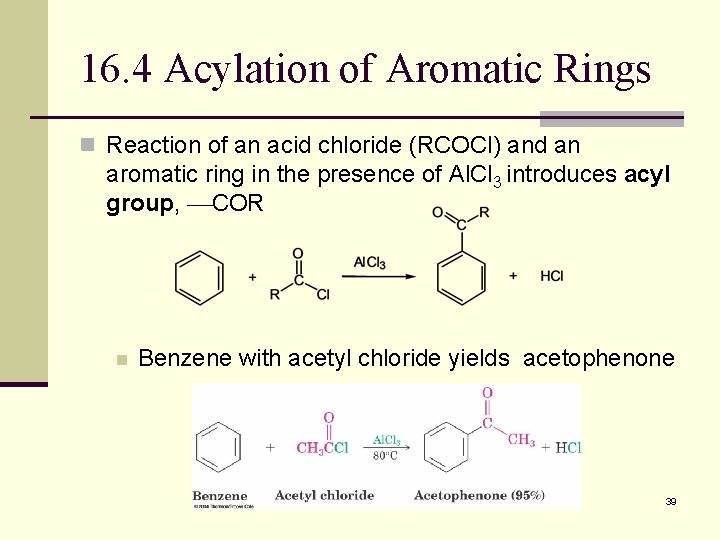
16. 4 Acylation of Aromatic Rings n Reaction of an acid chloride (RCOCl) and an aromatic ring in the presence of Al. Cl 3 introduces acyl group, COR n Benzene with acetyl chloride yields acetophenone 39
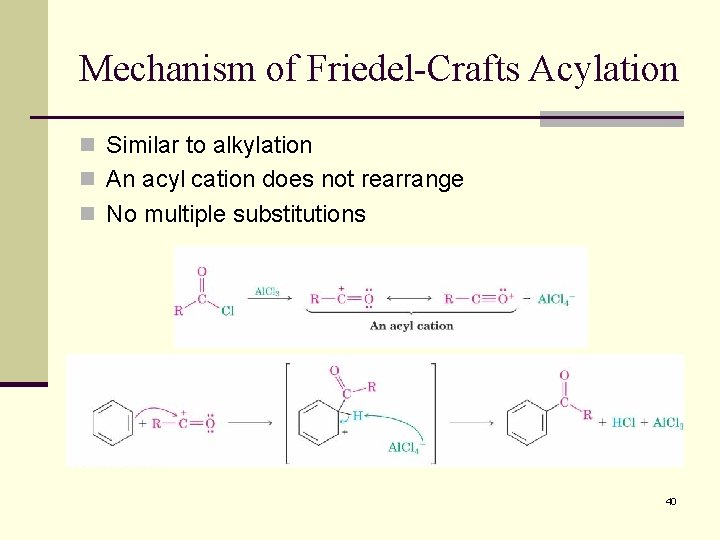
Mechanism of Friedel-Crafts Acylation n Similar to alkylation n An acyl cation does not rearrange n No multiple substitutions 40

16. 5 Substituent Effects in Aromatic Rings A substituent already present on a benzene ring has two effects: 1. Reactivity of the ring. n Some substituents activate the ring and make it more reactive than benzene. (Reactivity increases ) Phenyl, -CH 3, -NHCOCH 3, -OH, -NH 2 (O, P) n Some substituents deactivate the ring and make it less reactive than benzene. (Reactivity increases ) -N+R 3, -NO 2, -CN, -SO 3 H, carbonyl Halogens, Hydrogens (M) (O, P) 41

16. 5 Substituent Effects in Aromatic Rings A substituent already present on a benzene ring has two effects: 2. Orientation of the reaction. n The three possible disubstituted products - ortho, para and meta - are not formed in equal amounts. The substituent already on the ring determines the position of the second substituent (see labels on 1). n Substituents are classified into three groups; n n ortho-para directing activators ortho-para directing deactivators meta directing deactivators Note: there are no meta directing activators Inductive and resonance effects control the reactivity and orientation in electrophilic aromatic substitutions. 42
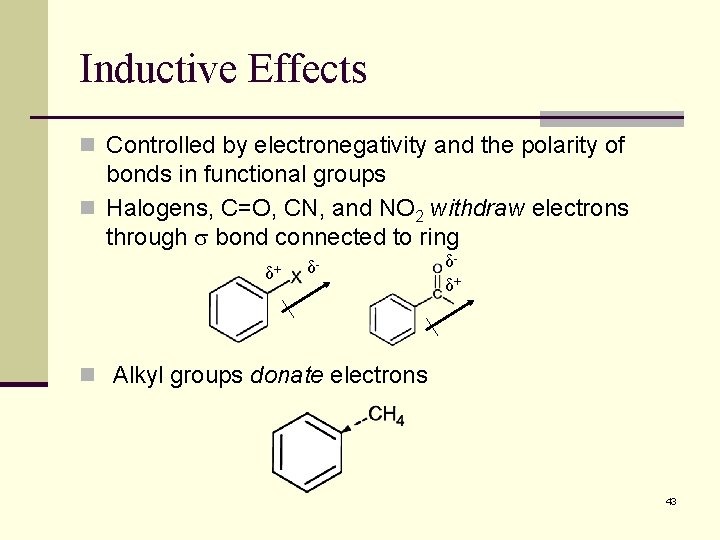
Inductive Effects n Controlled by electronegativity and the polarity of bonds in functional groups n Halogens, C=O, CN, and NO 2 withdraw electrons through s bond connected to ring δ+ δ- δδ+ n Alkyl groups donate electrons 43

Resonance Effects – Electron Withdrawal & Donation n The withdrawal or donation of electrons through bond due to the overlap of the p orbital on the substituent with a p orbital on the aromatic ring. n Carbonyl, cyano, and nitro substituents withdraw electrons from the aromatic ring through resonance. n Halogen, hydroxyl alkoxy (-OR) and amino substituents donate electrons to the ring. 44

Summary Table: Effect of Substituents in Aromatic Substitution (Table 16. 2) 45
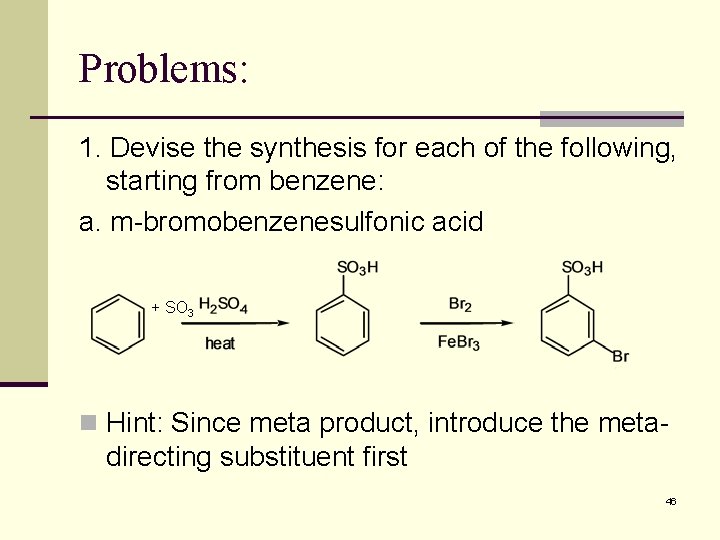
Problems: 1. Devise the synthesis for each of the following, starting from benzene: a. m-bromobenzenesulfonic acid + SO 3 n Hint: Since meta product, introduce the meta- directing substituent first 46
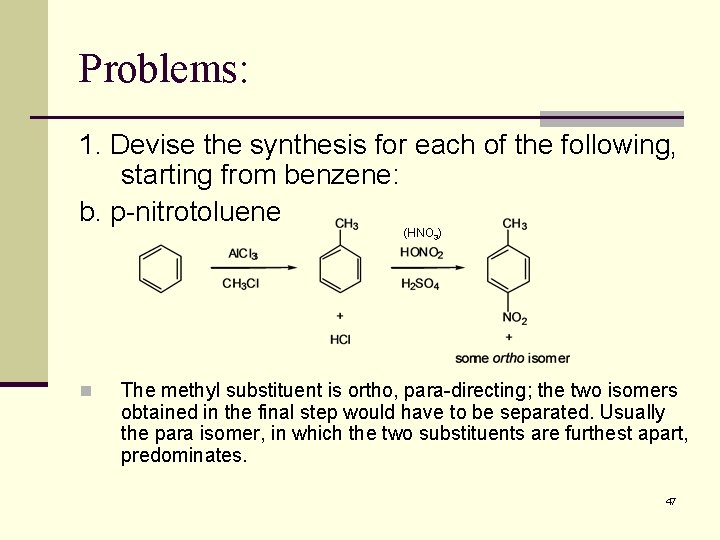
Problems: 1. Devise the synthesis for each of the following, starting from benzene: b. p-nitrotoluene (HNO 3) n The methyl substituent is ortho, para-directing; the two isomers obtained in the final step would have to be separated. Usually the para isomer, in which the two substituents are furthest apart, predominates. 47

Problems: 2. Is it possible to prepare mbromochlorobenzene by two successive electrophilic aromatic substitutions? Explain. No, because both Br and Cl are ortho, paradirecting. 48
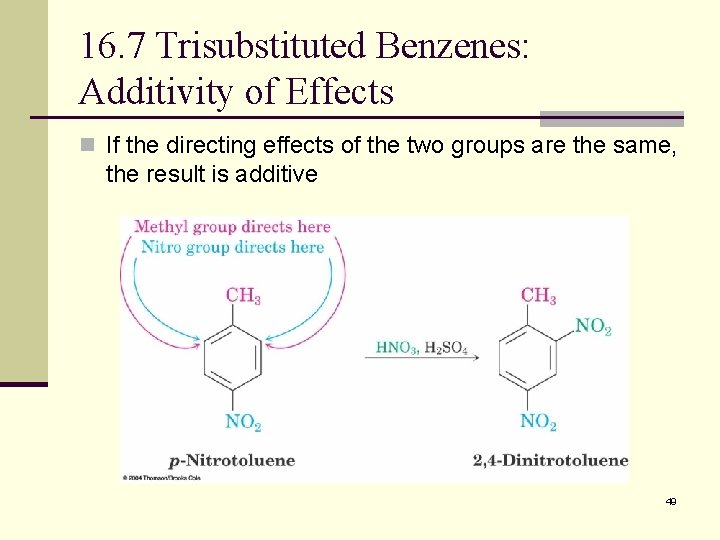
16. 7 Trisubstituted Benzenes: Additivity of Effects n If the directing effects of the two groups are the same, the result is additive 49

Substituents with Opposite Effects n If the directing effects of two groups oppose each other, the more powerful activating group decides the principal outcome n Usually gives mixtures of products -CH 3, -OH both OP directors -OH stronger activating group 50

Meta-Disubstituted Compounds Are Unreactive Between the Substituents n Meta: The reaction site is too hindered n Ortho: To make aromatic rings with three adjacent substituents, it is best to start with an ortho-disubstituted compound 51
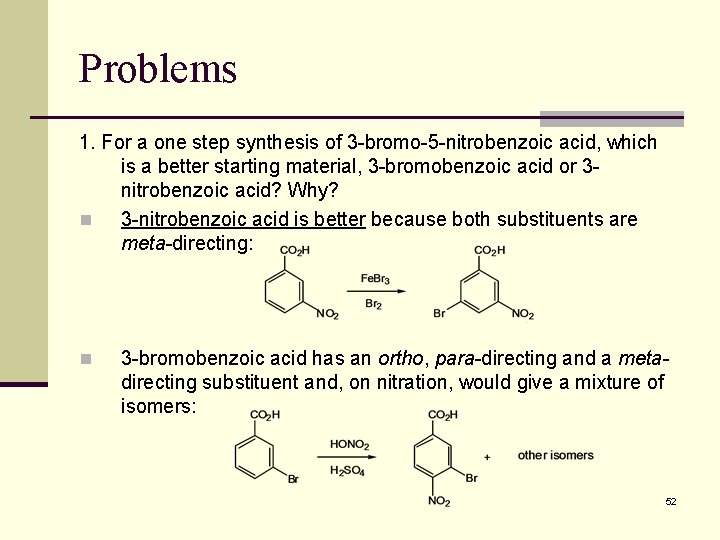
Problems 1. For a one step synthesis of 3 -bromo-5 -nitrobenzoic acid, which is a better starting material, 3 -bromobenzoic acid or 3 nitrobenzoic acid? Why? n 3 -nitrobenzoic acid is better because both substituents are meta-directing: n 3 -bromobenzoic acid has an ortho, para-directing and a metadirecting substituent and, on nitration, would give a mixture of isomers: 52
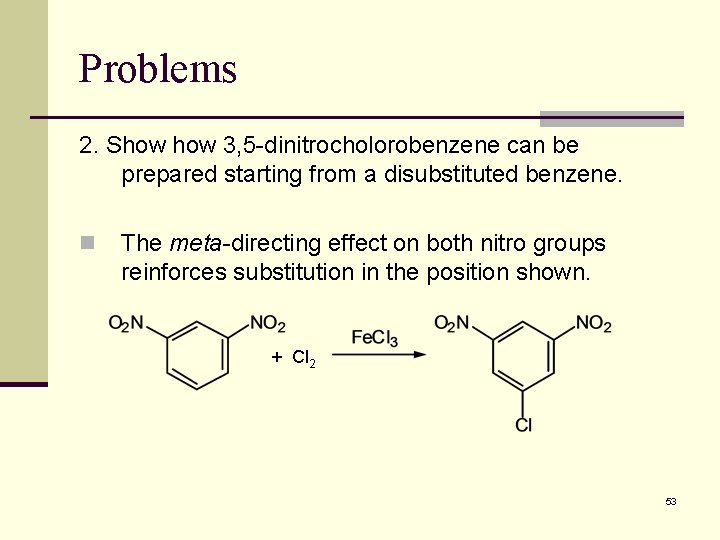
Problems 2. Show 3, 5 -dinitrocholorobenzene can be prepared starting from a disubstituted benzene. n The meta-directing effect on both nitro groups reinforces substitution in the position shown. + Cl 2 53
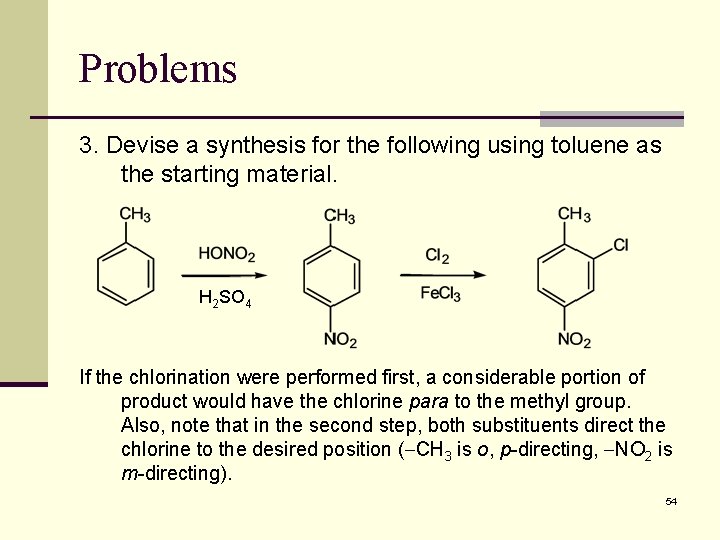
Problems 3. Devise a synthesis for the following using toluene as the starting material. H 2 SO 4 If the chlorination were performed first, a considerable portion of product would have the chlorine para to the methyl group. Also, note that in the second step, both substituents direct the chlorine to the desired position ( CH 3 is o, p-directing, NO 2 is m-directing). 54
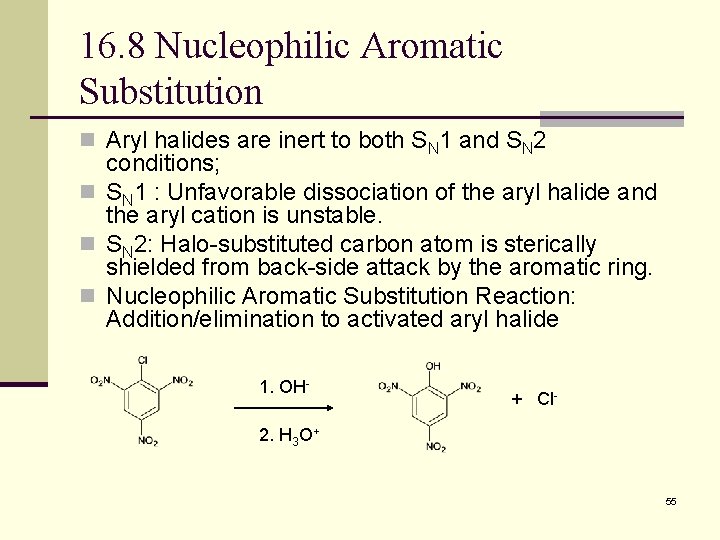
16. 8 Nucleophilic Aromatic Substitution n Aryl halides are inert to both SN 1 and SN 2 conditions; n SN 1 : Unfavorable dissociation of the aryl halide and the aryl cation is unstable. n SN 2: Halo-substituted carbon atom is sterically shielded from back-side attack by the aromatic ring. n Nucleophilic Aromatic Substitution Reaction: Addition/elimination to activated aryl halide 1. OH- + Cl- 2. H 3 O+ 55
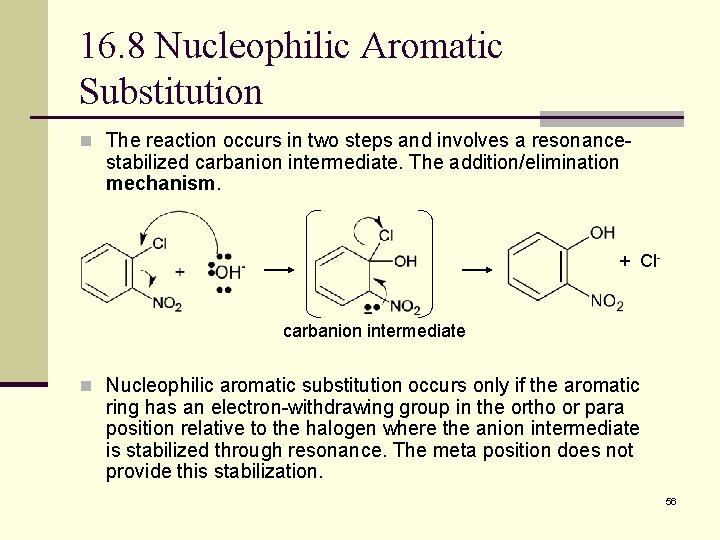
16. 8 Nucleophilic Aromatic Substitution n The reaction occurs in two steps and involves a resonance- stabilized carbanion intermediate. The addition/elimination mechanism. + Cl- carbanion intermediate n Nucleophilic aromatic substitution occurs only if the aromatic ring has an electron-withdrawing group in the ortho or para position relative to the halogen where the anion intermediate is stabilized through resonance. The meta position does not provide this stabilization. 56

16. 8 Nucleophilic Aromatic Substitution Differences between electrophilic and nucleophilic aromatic substitutions: n Electrophilic substitutions are favored by electron donating substituents, which stabilize the carbocation intermediate. n Nucleophilic substitutions are favored by electron withdrawing substituents, which stabilize the carbanion intermediate. n The electron withdrawing groups that deactivate rings for electrophilic substitution (NO 2, carbonyl, cyano, etc. ), activate them for nucleophilic substitution. These groups are meta directors in electrophilic substitution, but ortho, para directors in nucleophilic substitution. 57
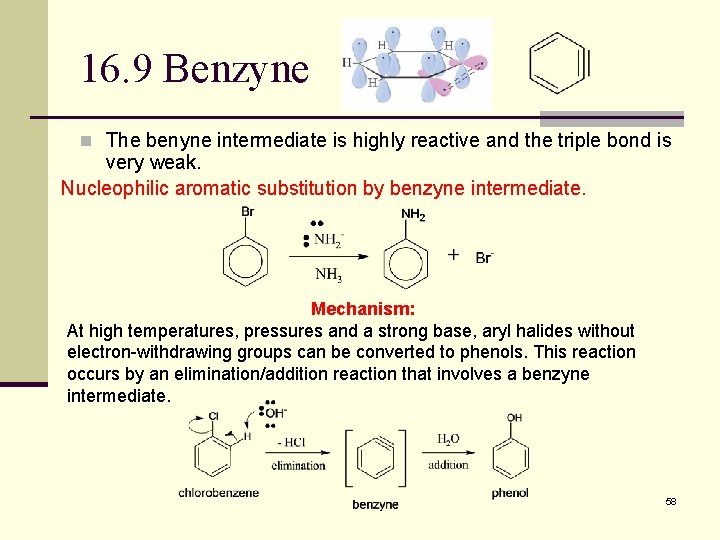
16. 9 Benzyne n The benyne intermediate is highly reactive and the triple bond is very weak. Nucleophilic aromatic substitution by benzyne intermediate. Mechanism: At high temperatures, pressures and a strong base, aryl halides without electron-withdrawing groups can be converted to phenols. This reaction occurs by an elimination/addition reaction that involves a benzyne intermediate. 58

16. 10 Oxidation of Aromatic Compounds n The benzene ring cannot be oxidized, but the alkyl side chains can n Many of the reactions happen at the benzylic position. Reactions occur with 1º, 2º but not 3º, alkyl side chains. A mixture of products may be formed. 59
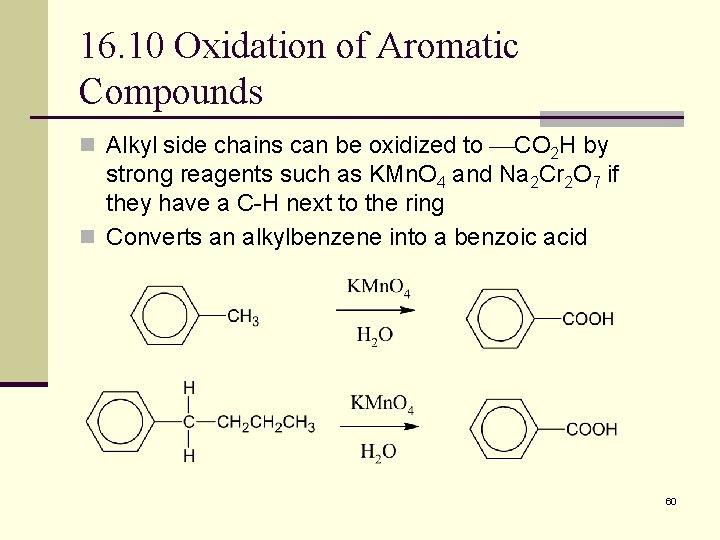
16. 10 Oxidation of Aromatic Compounds n Alkyl side chains can be oxidized to CO 2 H by strong reagents such as KMn. O 4 and Na 2 Cr 2 O 7 if they have a C-H next to the ring n Converts an alkylbenzene into a benzoic acid 60
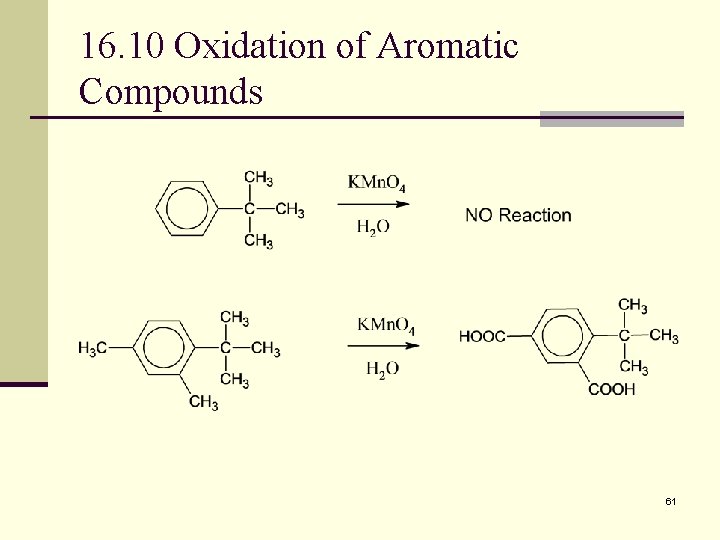
16. 10 Oxidation of Aromatic Compounds 61
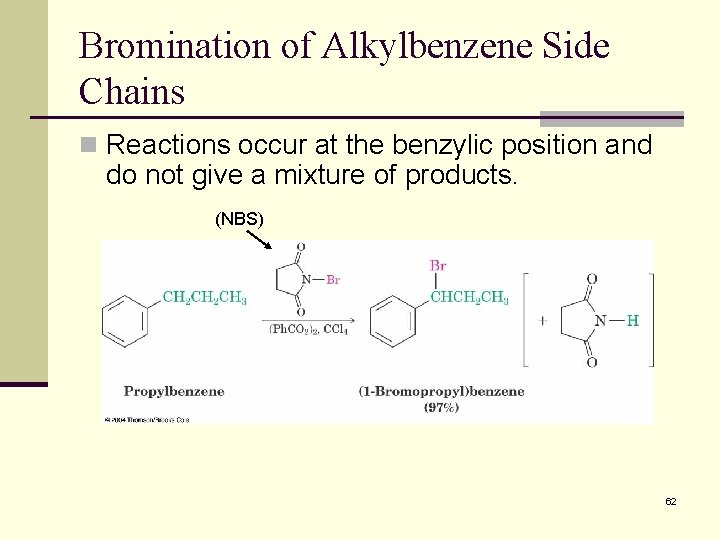
Bromination of Alkylbenzene Side Chains n Reactions occur at the benzylic position and do not give a mixture of products. (NBS) 62
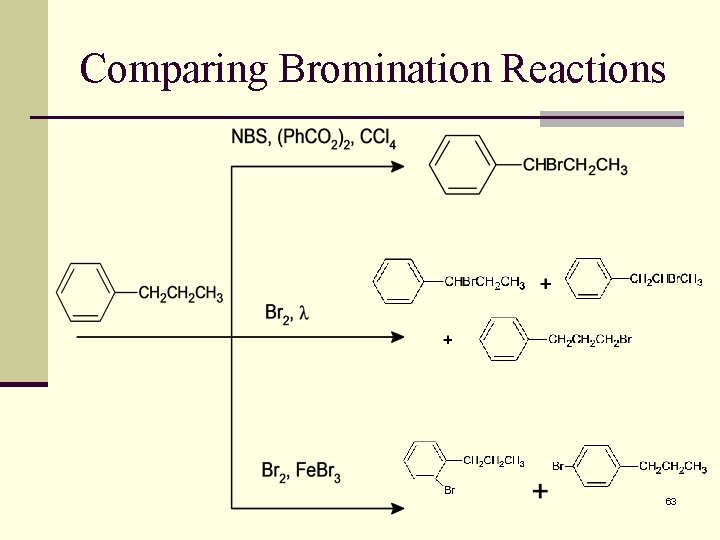
Comparing Bromination Reactions 63

16. 11 Reduction of Aromatic Compounds n Aromatic rings are inert to catalytic hydrogenation under conditions that reduce alkene double bonds n Can selectively reduce an alkene double bond in the presence of an aromatic ring 64
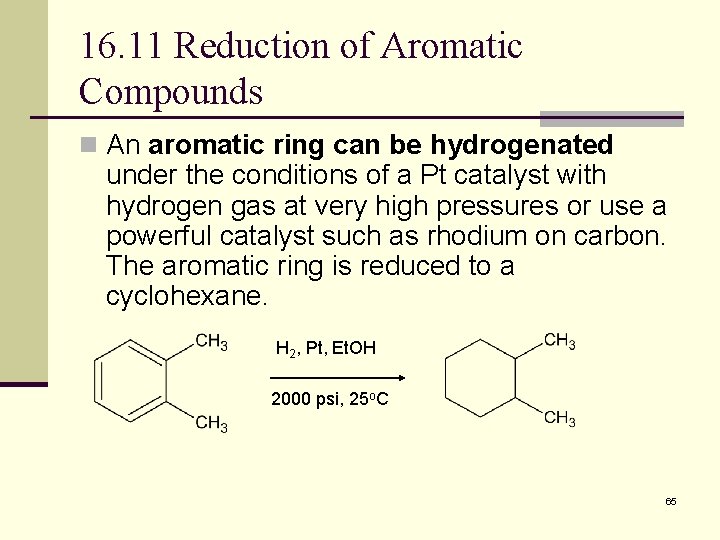
16. 11 Reduction of Aromatic Compounds n An aromatic ring can be hydrogenated under the conditions of a Pt catalyst with hydrogen gas at very high pressures or use a powerful catalyst such as rhodium on carbon. The aromatic ring is reduced to a cyclohexane. H 2, Pt, Et. OH 2000 psi, 25 o. C 65
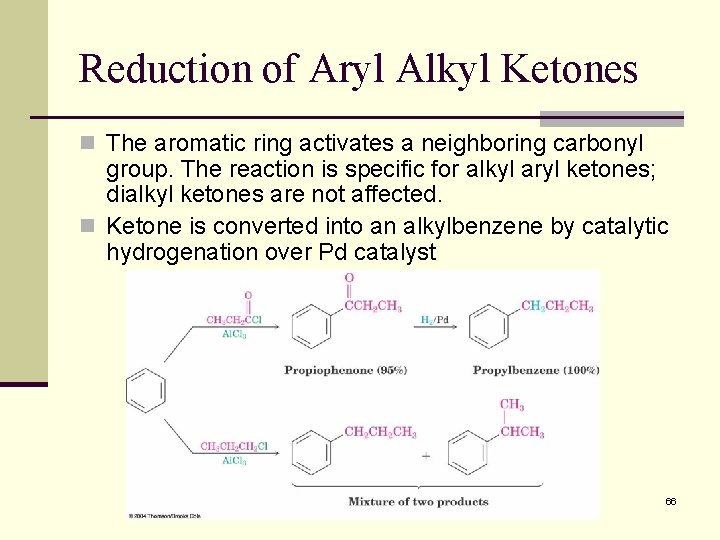
Reduction of Aryl Alkyl Ketones n The aromatic ring activates a neighboring carbonyl group. The reaction is specific for alkyl aryl ketones; dialkyl ketones are not affected. n Ketone is converted into an alkylbenzene by catalytic hydrogenation over Pd catalyst 66

Brief Review of Organic Reactions & Mechanisms For Chapters 6 -16 n Look over this review, It is a good summary for what you will need for this semester. n I suggest writing a reaction summary for you to review. 67
- Slides: 67