1411 Chapter 7 Periodic Properties of the Elements





















- Slides: 30

1411 Chapter 7 Periodic Properties of the Elements 1

Development of Periodic Table • Elements in the same group generally have similar chemical properties. – Because they have the same number of electrons in their valence orbitals – the orbitals that hold electrons involved in bonding • But they do not necessarily share physical properties. O 2(g) O: [He]2 s 22 p 4 S 8(s): S: [Ne]3 s 23 p 4 2

Development of Periodic Table Dmitri Mendeleev and Lothar Meyer • (1869): Independently came to the same conclusion about how elements should be grouped • Similar physical & chemical properties recur periodically with increasing atomic weights • Arranged elements in proper sequence 3

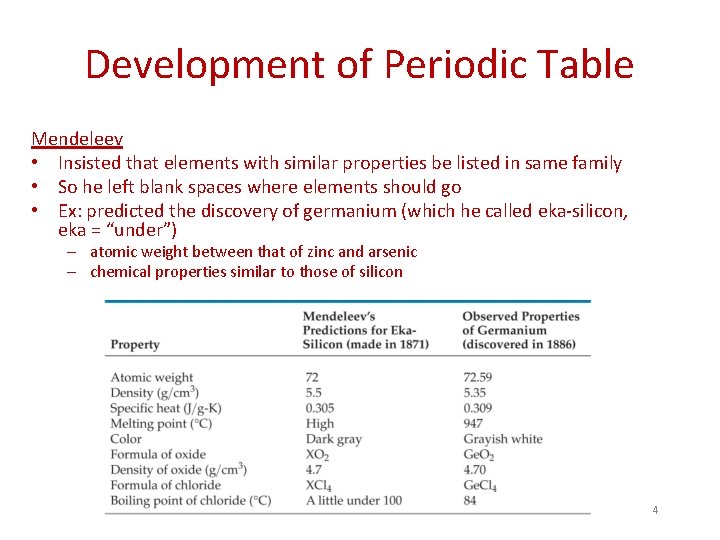
Development of Periodic Table Mendeleev • Insisted that elements with similar properties be listed in same family • So he left blank spaces where elements should go • Ex: predicted the discovery of germanium (which he called eka-silicon, eka = “under”) – atomic weight between that of zinc and arsenic – chemical properties similar to those of silicon 4

Periodic Trends • In this chapter, we will rationalize observed trends in – Sizes of atoms and ions. – Ionization energy. – Electron affinity. 5

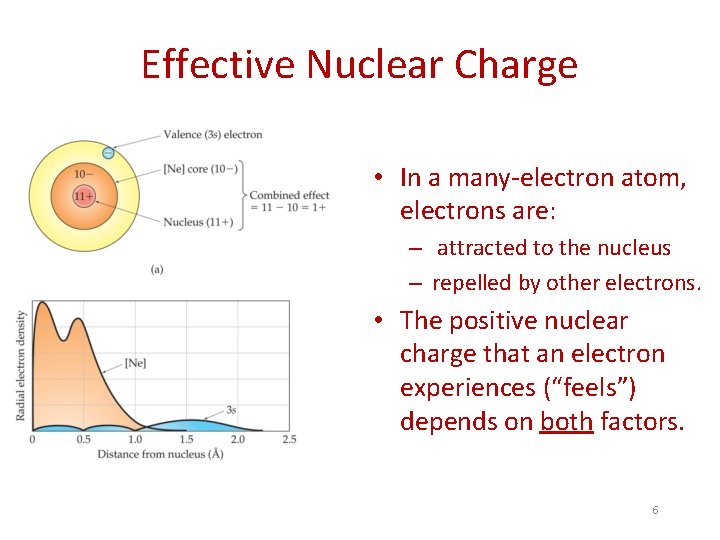
Effective Nuclear Charge • In a many-electron atom, electrons are: – attracted to the nucleus – repelled by other electrons. • The positive nuclear charge that an electron experiences (“feels”) depends on both factors. 6

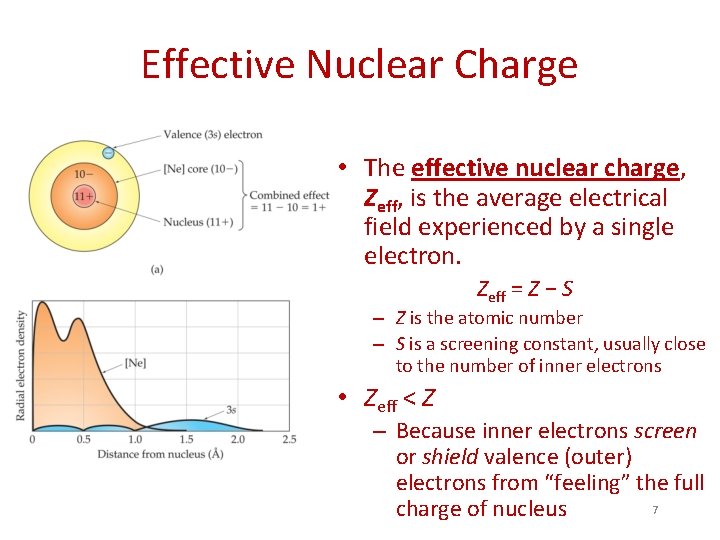
Effective Nuclear Charge • The effective nuclear charge, Zeff, is the average electrical field experienced by a single electron. Zeff = Z − S – Z is the atomic number – S is a screening constant, usually close to the number of inner electrons • Zeff < Z – Because inner electrons screen or shield valence (outer) electrons from “feeling” the full 7 charge of nucleus

Effective Nuclear Charge • Trends for Zeff from periodic table: – Zeff for valence electrons increases moving from left to right across a period – Zeff for valence electrons increases slightly going down a group (but smaller change than moving across a period) 8

Effective Nuclear Charge: Examples 1) Estimate the value of Zeff experienced by a valence electron in the following atoms by assuming core electrons are 100% effective at shielding the valence electrons from the nuclear charge and the valence electrons provide no shielding for each other. a) Al b) O Zeff = Z – S = 13 – 10 = 3+ Zeff = Z – S = 8 – 2 = 6+ 2) In which element, boron or neon, does the outermost electron experience a greater effective nuclear charge? Ne 3) Which will experience the greater effective nuclear charge, the electrons in the n = 2 shell in boron or the n = 2 shell in aluminum? Al: Al has a larger nuclear charge (Z = 13) than boron (Z = 5). The shielding of the 1 s core electrons in the 2 atoms is approx. equal, so the n = 2 electrons in Al experience a greater Zeff and are thus situated closer to the nucleus. 9

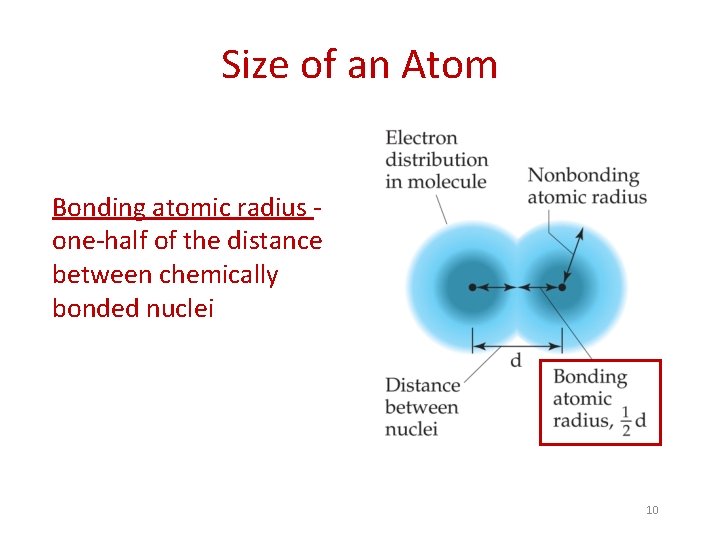
Size of an Atom Bonding atomic radius one-half of the distance between chemically bonded nuclei 10

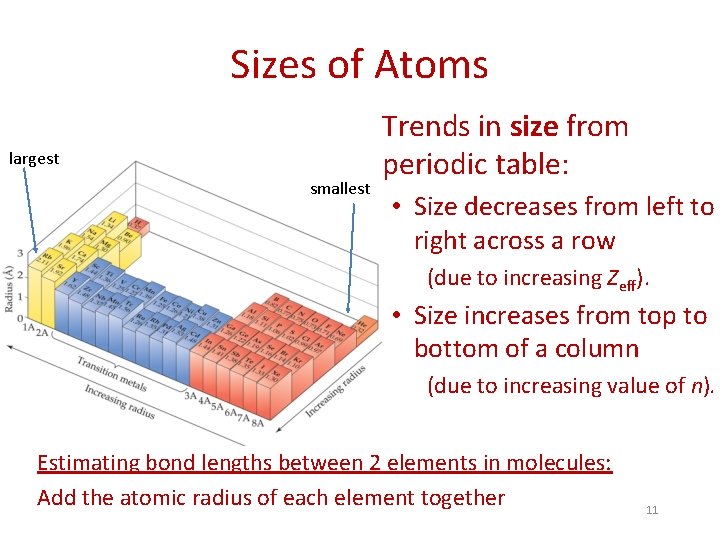
Sizes of Atoms largest smallest Trends in size from periodic table: • Size decreases from left to right across a row (due to increasing Zeff). • Size increases from top to bottom of a column (due to increasing value of n). Estimating bond lengths between 2 elements in molecules: Add the atomic radius of each element together 11

Sizes of Atoms Examples: 1) Predict the bond length of the C–H bond in CH 3 OH. Atomic radii: C: 0. 77 Å H: 0. 37 Å Bond length = 0. 77 Å + 0. 37 Å = 1. 14 Å (FYI: compares to experimental bond length of 1. 108 Å) 2) Arrange the following elements in order of increasing atomic radius: 12 O < N < P < Al O, P, Al, N

Sizes of Ions • Cations are smaller than their parent atoms. – Electrons are removed so repulsions between electrons are reduced. • Anions are larger than their parent atoms. – Electrons are added and repulsions between electrons are increased. • Ions increase in size as you go down a column. – This is due to increasing value of n. 13

Sizes of Ions • Cations are smaller than their parent atoms. – Electrons are removed so repulsions between electrons are reduced. • Anions are larger than their parent atoms. – Electrons are added so repulsions between electrons are increased. • Ions increase in size as you go down a column. – This is due to increasing value of n. 14

Sizes of Ions • Cations are smaller than their parent atoms. – Electrons are removed so repulsions between electrons are reduced. • Anions are larger than their parent atoms. – Electrons are added so repulsions between electrons are increased. • Ions increase in size as you go down a column. – This is due to increasing value of n. 15

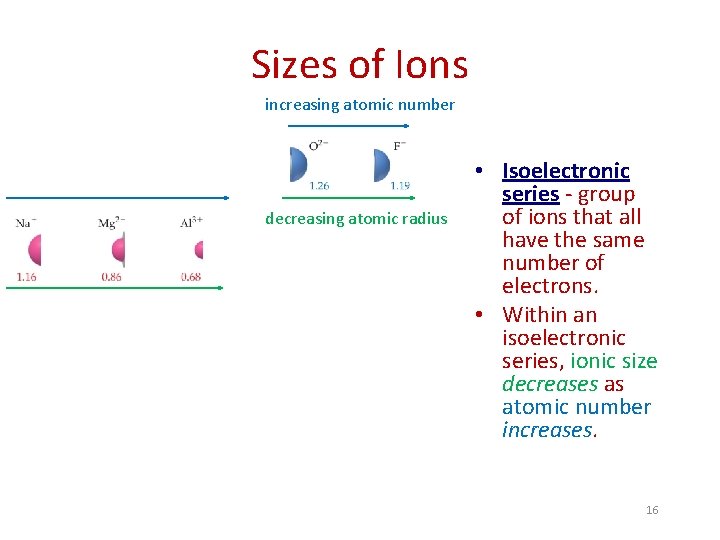
Sizes of Ions increasing atomic number decreasing atomic radius • Isoelectronic series - group of ions that all have the same number of electrons. • Within an isoelectronic series, ionic size decreases as atomic number increases. 16

Sizes of Ions: Examples 1. Arrange the ions and/or atoms in order of increasing atomic radius: a) Ca 2+, Ca Ca 2+ < Ca b) Te 2 -, O 2 -, Se 2 -, O O < O 2 - < Se 2 - < Te 2 - c) Rb+, Sr 2+, Br- Sr 2+ < Rb+ < Br 17

Ionization Energy • The ionization energy is the amount of energy required to remove an electron from the ground state of a gaseous atom or ion. – first ionization energy I 1 - energy required to remove first electron • Ex: Na(g) Na+(g) + e- ΔE = 495 k. J/mol – second ionization energy I 2 - energy required to remove second electron • Ex: Na+(g) Na 2+(g) + e- ΔE = 4562 k. J/mol – etc 18

Ionization Energy • Greater ionization energy means harder to remove the electron. • It requires more energy to remove each successive electron: I 1 < I 2 < I 3 … • When all valence electrons have been removed, the ionization energy increases by a very large amount (because removing core electrons): I 1 < I 2 … < Ivalence <<< Ivalence+1 • That’s why only valence electrons are involved in bonding! 19

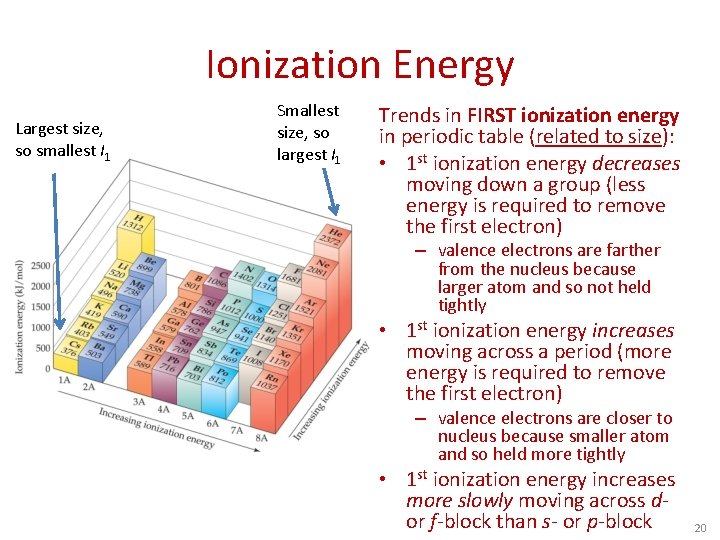
Ionization Energy Largest size, so smallest I 1 Smallest size, so largest I 1 Trends in FIRST ionization energy in periodic table (related to size): • 1 st ionization energy decreases moving down a group (less energy is required to remove the first electron) – valence electrons are farther from the nucleus because larger atom and so not held tightly • 1 st ionization energy increases moving across a period (more energy is required to remove the first electron) – valence electrons are closer to nucleus because smaller atom and so held more tightly • 1 st ionization energy increases more slowly moving across dor f-block than s- or p-block 20

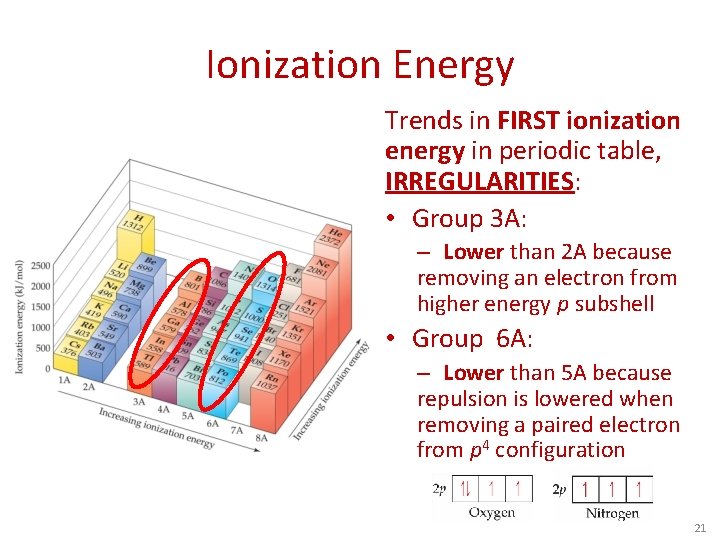
Ionization Energy Trends in FIRST ionization energy in periodic table, IRREGULARITIES: • Group 3 A: – Lower than 2 A because removing an electron from higher energy p subshell • Group 6 A: – Lower than 5 A because repulsion is lowered when removing a paired electron from p 4 configuration 21

Ionization Energy Example: • Pick the element with the higher first ionization energy: a) Ti or Ba b) Ag or Cu c) Cl or As Ti Cu Cl • Use the periodic table to select the element in the following list for which there is the largest difference between the second and third ionization energies: Na, Mg, Al, Si, P, Se, Cl Mg 22

Electron Configurations of Ions • • Determine electron configuration of neutral atom If removing electrons, remove from: 1) The highest principal quantum number n FIRST 2) Within a shell, remove from highest value of l • If adding electrons, add to: 1) Empty or partially filled orbital with lowest value of n 2) Within a shell, add to empty or partially filled orbital with lowest value of l Examples: Determine the condensed electron configuration of the following ions. a)N 3 - [He]2 s 22 p 6 or just [Ne] d) Mo 4+ [Kr]4 d 2 [Kr]5 s 24 d 105 p 5 b)Ge+ [Ar]4 s 23 d 104 p 1 e) Sb 2 c)Fe 2+ [Ar]3 d 6 f) Nb 5+ [Kr] 23

Electron Affinity Electron affinity is the energy change accompanying the addition of an electron to a gaseous atom: (The ease with which an atom gains an electron) Cl(g) + e− Cl−(g) ΔE = -347 k. J/mol 24

Electron Affinity Smallest size, Largest I 1, Most exothermic EA Largest size, Smallest I 1, Least exothermic EA Trends in electron affinity in periodic table (in general – much less pronounced): • Electron affinity becomes more exothermic (more negative) moving across a period • Electron affinity becomes somewhat more exothermic moving up a group 25

Trends in Electron Affinity Trends in electron affinity in periodic table, IRREGULARITIES: • Group 2 A - The added electron must go in a higher energy p-orbital, not an s -orbital. • Group 5 A - p-orbitals are already half-filled so added electron must go into occupied orbital, creating repulsion 26

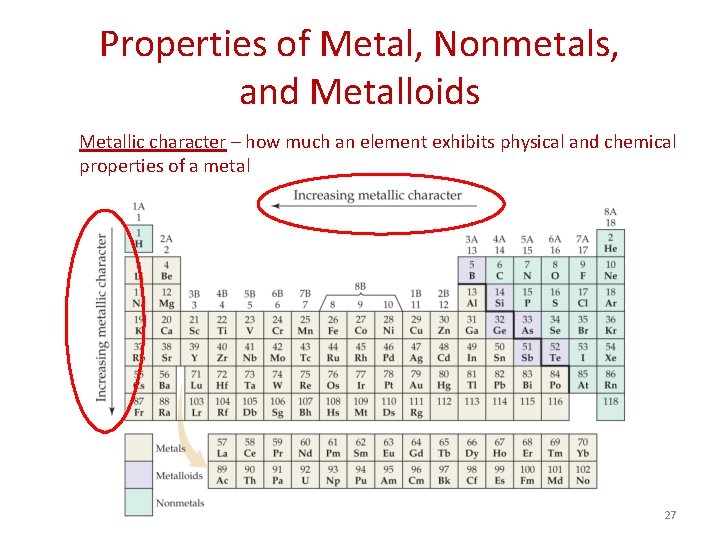
Properties of Metal, Nonmetals, and Metalloids Metallic character – how much an element exhibits physical and chemical properties of a metal 27

Metals • • • Common properties: Shiny luster Ductile – can be drawn into wires Malleable – can be pounded into sheets Good conductors of heat and electricity Solids at room temperature (except Hg) 28

Nonmetals Common properties: • Dull • Brittle • Poor conductors of heat and electricity • Variety of forms under normal conditions: a) Diatomic molecules Br 2(l) I 2(s) N 2(g) Cl 2(g) H 2(g) O 2(g) F 2(g) b) Monatomic gases Noble gases c) Solids (soft or hard) 30

Metalloids • Some characteristics of metals and some of nonmetals • Example: Silicon – Shiny – Brittle – Fairly poor conductor • Intermediate electrical conductivity = semiconductor! (used in integrated circuits and computer chips) 32