14 gases pressure Boyles Law Bernoullis Principle RQ

14 • gases, pressure, Boyle’s Law & Bernoulli’s Principle • RQ: 4, 13, 15, 17, 20. • Exercises: 7, 56. • Problems: 1, 2. 1

Gases • • independent molecular motion expand “fluid” exert “isotropic” pressure 2

Archimedes’ Principle for Air • Air provides a small buoyancy, noticeable when object has very low density • Examples: helium or hot-air balloon. Temperature Density (kg/m 3) 0°C (32°F) 10°C (50°F) 20°C (68°F) 30°C (86°F) 3

Boyle’s Law • for a confined gas, (pressure)x(volume) = constant, i. e. , PV = constant. • Ex. A balloon squeezed to half its original size has. . . • Reasoning: in a smaller container, the molecules will hit the walls _______ exerting ______ on them. 4

Bernoulli’s Principle • When the speed of a fluid* _______ internal pressure in the fluid* ____ *or gas 5

Why does speed decrease pressure? • before the air blows, air molecules exert isotropic pressure, moving in all directions. • air-flow _____________ giving a preferred direction for air molecules at top of tube. The air molecules below are able to move up…. 6
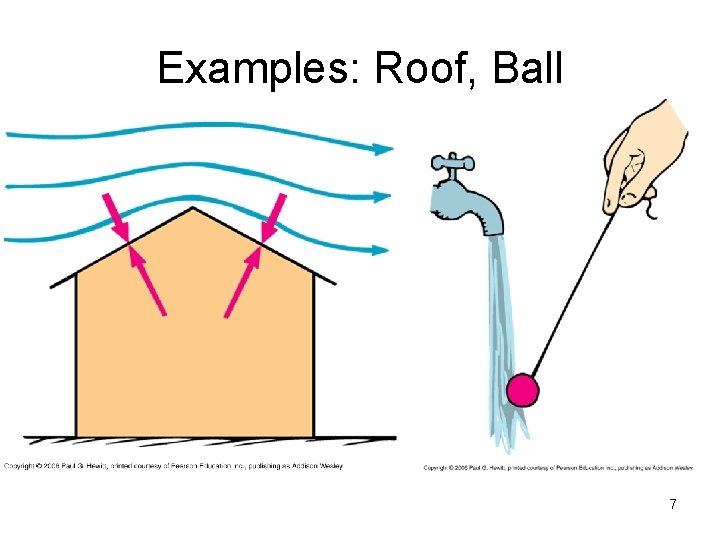
Examples: Roof, Ball 7
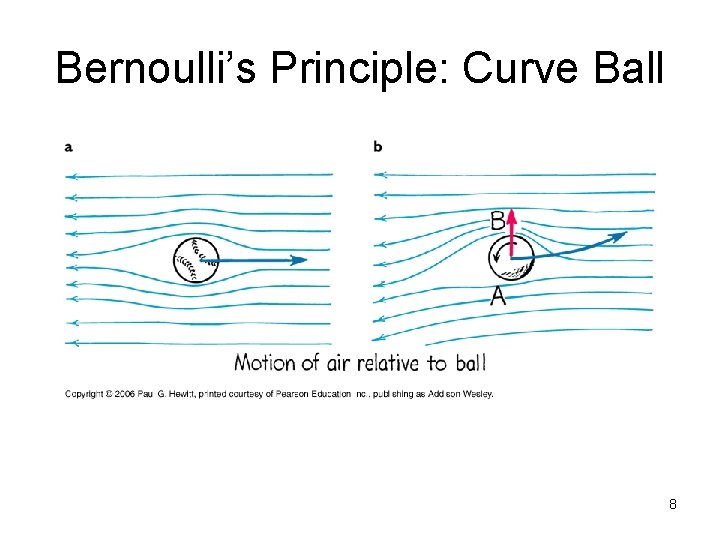
Bernoulli’s Principle: Curve Ball 8
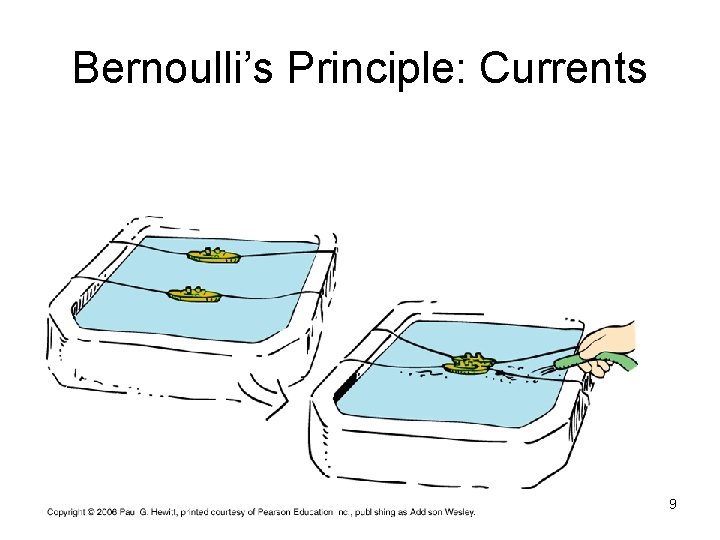
Bernoulli’s Principle: Currents 9

summary • gases exert isotropic pressure, but loose this effect when flowing (Bernoulli). • gases exert a small buoyant force on all objects • confined gases have constant product of pressure and volume 10
- Slides: 10