14 Compressed Air Learning Objectives Describe the characteristics


14 Compressed Air
Learning Objectives • Describe the characteristics of free air. • Identify the problems associated with the use of unconditioned air in a pneumatic system. Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Learning Objectives • Compare the composition of compressed air in a pneumatic system with the characteristics of free air. • Explain the terms used to describe characteristics of compressed air. Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.
Learning Objectives • Compare the scales used to measure the pressure of free and compressed air. • Compare the isothermal, adiabatic, and actual modes of air compression. Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Learning Objectives • Apply the principles of the general gas law to the compression and expansion of the air used in a pneumatic system. Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Basic Source of System Air • Pneumatic system air source is the atmosphere that blankets earth • Approximately 620 miles deep • Supplies oxygen that supports combustion Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

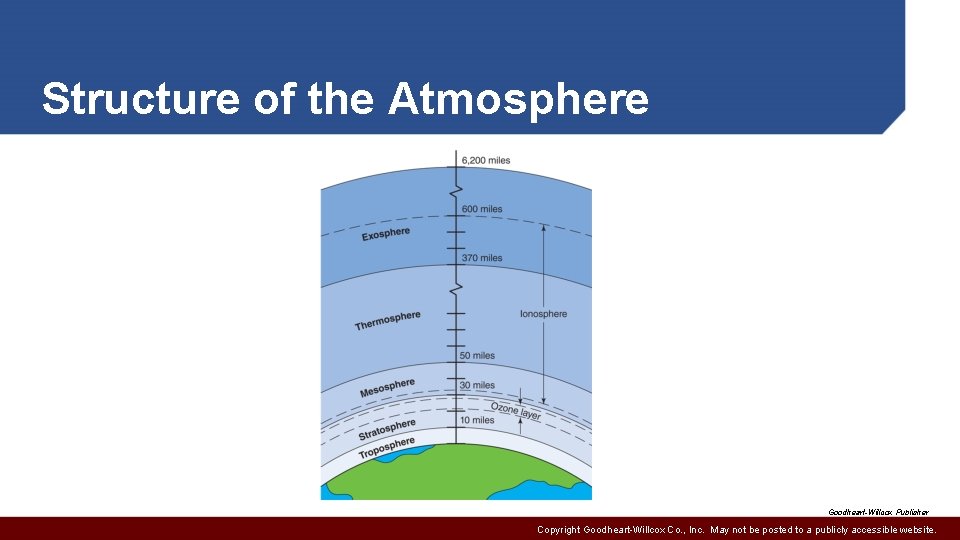
Structure of the Atmosphere • Distinct layers separated by transition zones • Troposphere from earth’s surface to maximum 10 miles above surface • Stratosphere from troposphere to about 30 miles above earth’s surface • Ozone layer in upper limits, filters ultraviolet energy • Mesosphere above stratosphere and about 50 miles above earth’s surface • Ionosphere uppermost layers and extends to edge of space about 600 miles from earth’s surface Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.
Structure of the Atmosphere Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.
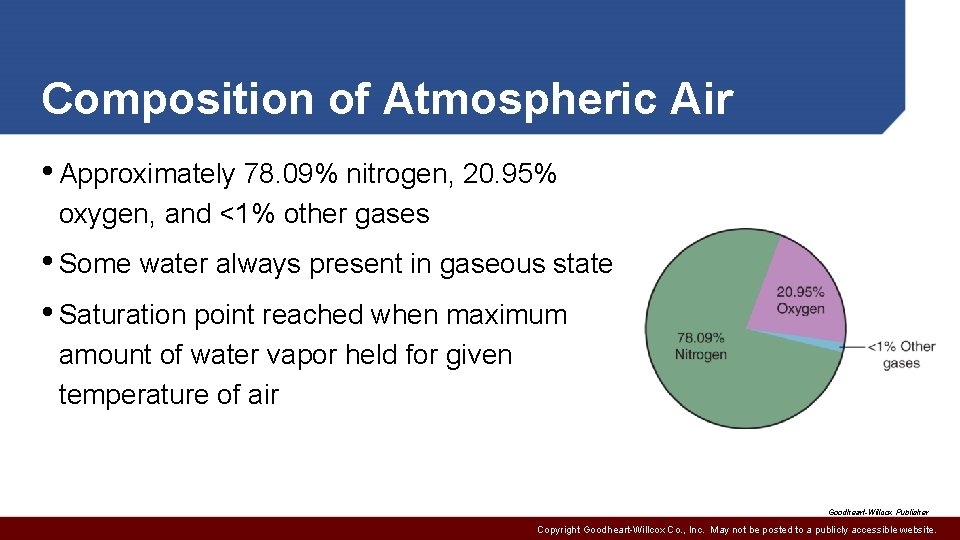
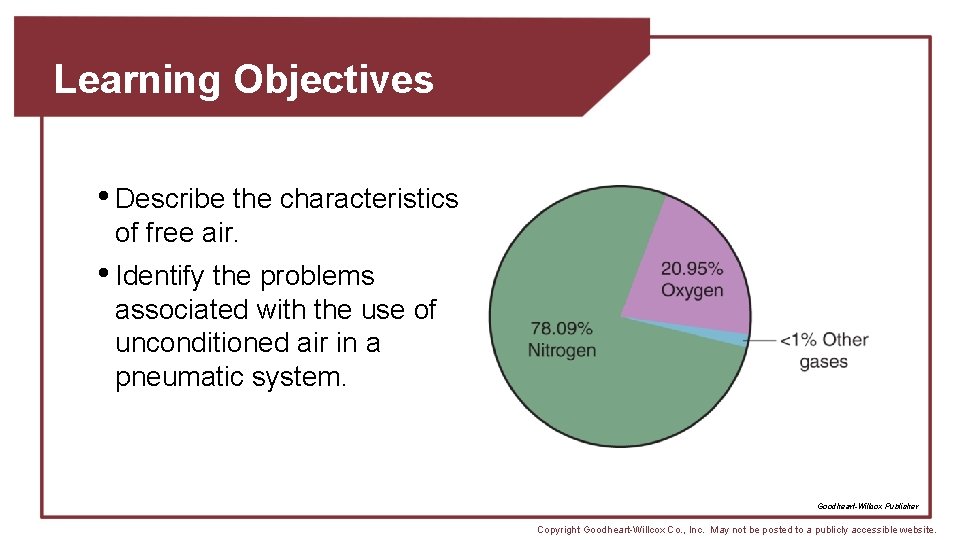
Composition of Atmospheric Air • Approximately 78. 09% nitrogen, 20. 95% oxygen, and <1% other gases • Some water always present in gaseous state • Saturation point reached when maximum amount of water vapor held for given temperature of air Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Composition of Atmospheric Air • Dust and dirt may become suspended and cause problems • Must consider weight and pressure • 14. 7 psia or 0 psig • One cubic foot at 68°F weighs about 0. 0752 lbs Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Pneumatic System Compressed Air • Atmospheric air typically referred to as free air • Must be conditioned before use in pneumatic system • Conditioning involves removing entrapped dirt, water vapor, heat, and incorporating oil for lubrication • Location affects amount of dirt in free air • Clean environment vs. dusty construction site Manufactured Housing Institute Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Pneumatic System Compressed Air • Relative humidity amount of water vapor carried by given volume of air • Dew point the temperature water vapor in saturated air begins to be released in liquid form Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Pneumatic System Compressed Air • Dry air contains water vapor • Should have relative humidity low enough to prevent formation of liquid water at ambient temperature of workstation • Compressed air lubricant carrier for components Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Compression and Expansion of Air • Air constantly changes during system operation • Volume decreases and pressure increases when air is compressed • Compressing air produces heat that increases air temperature Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

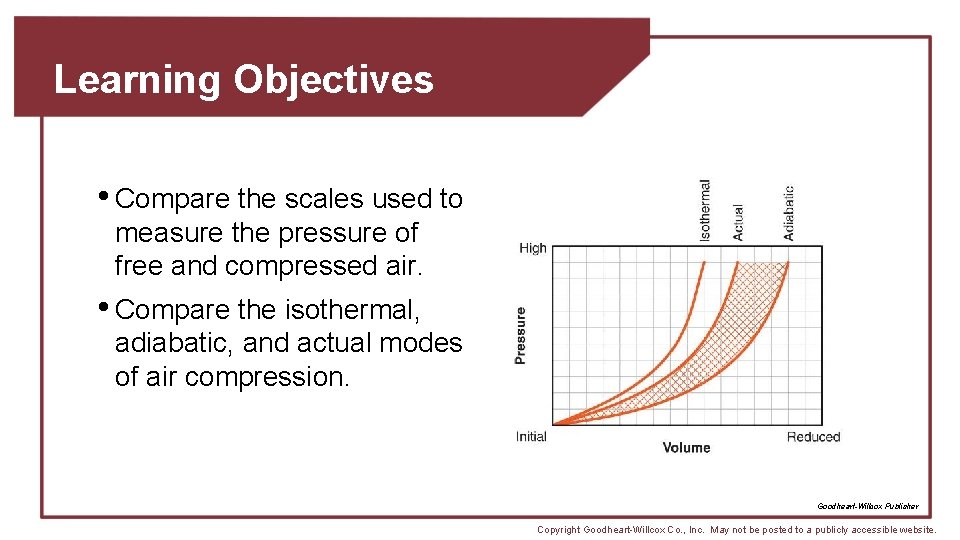
Compression and Expansion of Air • Isothermal compression • Assumes all heat is removed, resulting in constant temperature • Adiabatic compression • Assumes heat is retained, resulting in increased temperature and pressure • Actual compression somewhere between isothermal and adiabatic Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Reaction of Air to Temperature, Pressure, and Volume • Change in temperature, pressure, or volume result in change in at least one other factor • Relationships expressed based on absolute scales • Changes adhere to general gas law Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Pressure Variations • Any change in air pressure results in temperature or volume change Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Volume Variations • Changes in volume of air result in pressure or temperature change Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.

Temperature Variations • Increase or decrease in air temperature results in pressure or volume change Goodheart-Willcox Publisher Copyright Goodheart-Willcox Co. , Inc. May not be posted to a publicly accessible website.
- Slides: 20