14 Chemical Kinetics Copyright c The Mc GrawHill






![Dependence of Reaction Rate on Reactant Concentration rate = k[F 2]x[Cl. O 2]y F Dependence of Reaction Rate on Reactant Concentration rate = k[F 2]x[Cl. O 2]y F](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-19.jpg)
![Dependence of Reaction Rate on Reactant Concentration rate = k[F 2] [Cl. O 2]y Dependence of Reaction Rate on Reactant Concentration rate = k[F 2] [Cl. O 2]y](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-20.jpg)
![Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b. Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b.](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-21.jpg)
![Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b. Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b.](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-22.jpg)
![Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b. Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b.](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-23.jpg)






![Dependence of Reactant Concentration on Time Solution: Plot ln[C 2 H 5 I] vs Dependence of Reactant Concentration on Time Solution: Plot ln[C 2 H 5 I] vs](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-39.jpg)












- Slides: 66

14 Chemical Kinetics Copyright (c) The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

14 Chemical Kinetics 14. 1 Reaction Rates Average Reaction Rates Instantaneous Rate Stoichiometry and Reaction Rate 14. 2 Dependence of Reaction Rate on Reactant Concentration The Rate Law Experimental Determination of the Rate Law 14. 3 Dependence of Reactant Concentration of Time First-Order Reactions Second-Order Reactions 14. 4 Dependence of Reaction Rate on Temperature Collision Theory The Arrhenius Equation 14. 5 Reaction Mechanisms Elementary Reactions Rate-Determining Step Experimental Support for Reaction Mechanisms

14 Chemical Kinetics 14. 6 Catalysis Heterogeneous Catalysis Homogeneous Catalysis Enzymes: Biological Catalysts

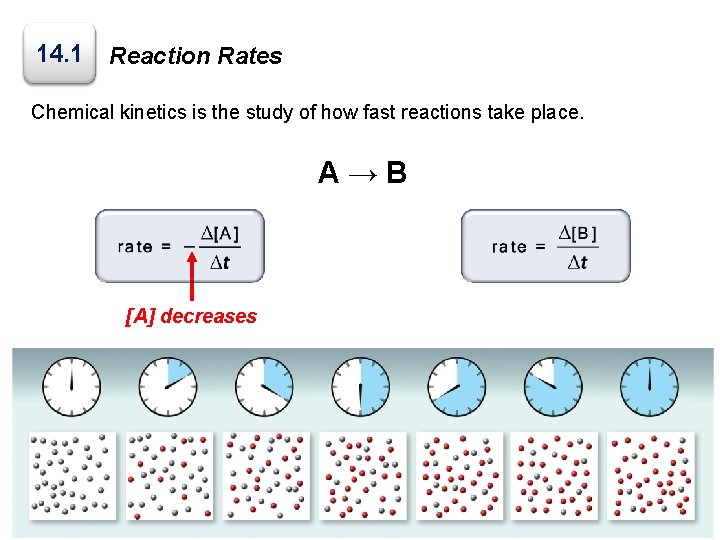
14. 1 Reaction Rates Chemical kinetics is the study of how fast reactions take place. A→B [A] decreases

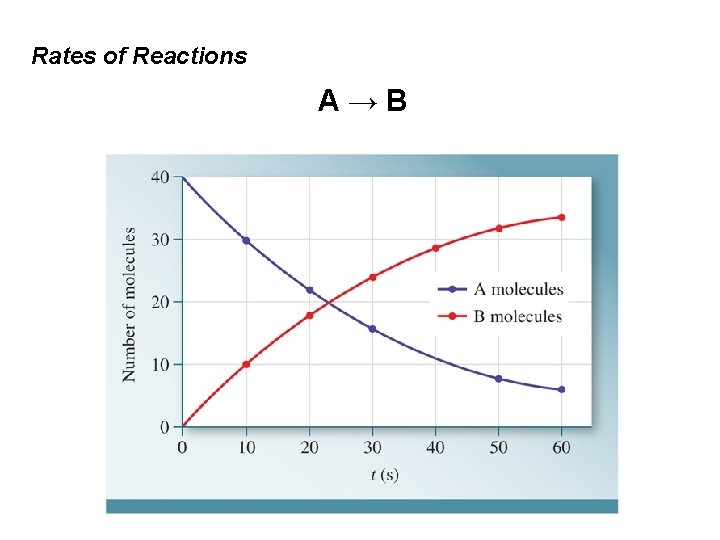
Rates of Reactions A→B

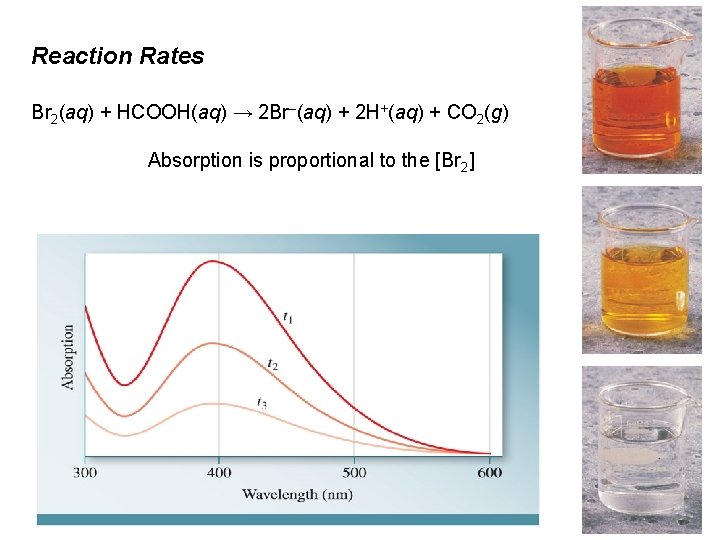
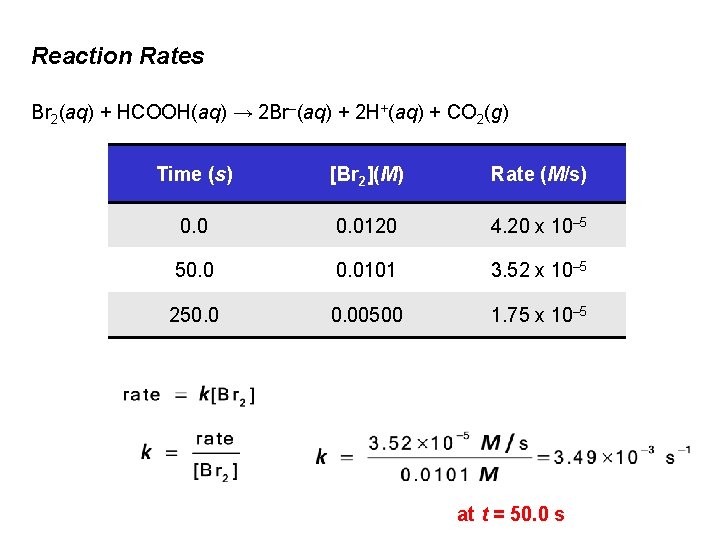
Types of Solutions Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) reddishbrown

Reaction Rates Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) Absorption is proportional to the [Br 2]

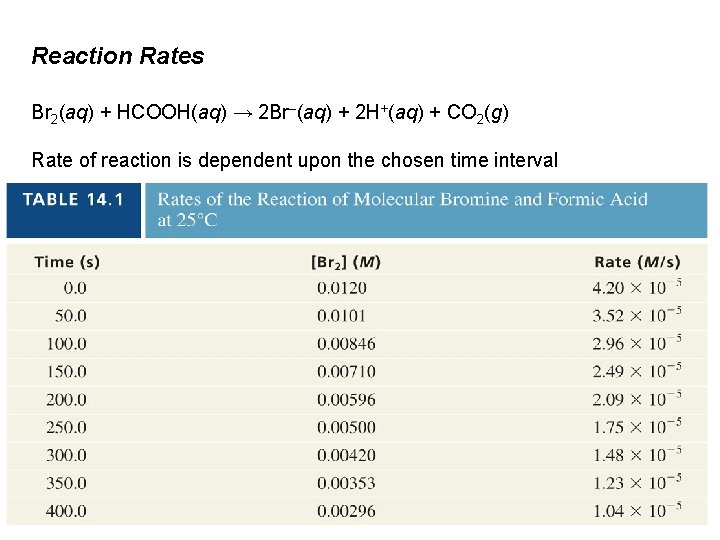
Reaction Rates Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) Rate of reaction is dependent upon the chosen time interval

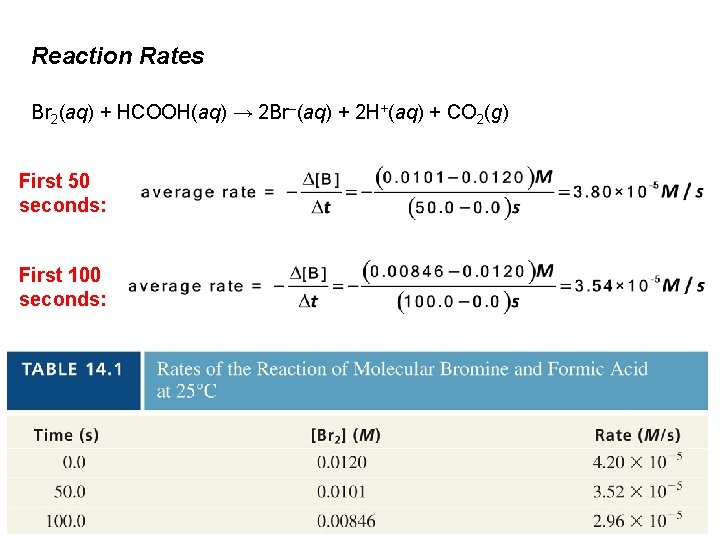
Reaction Rates Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) First 50 seconds: First 100 seconds:

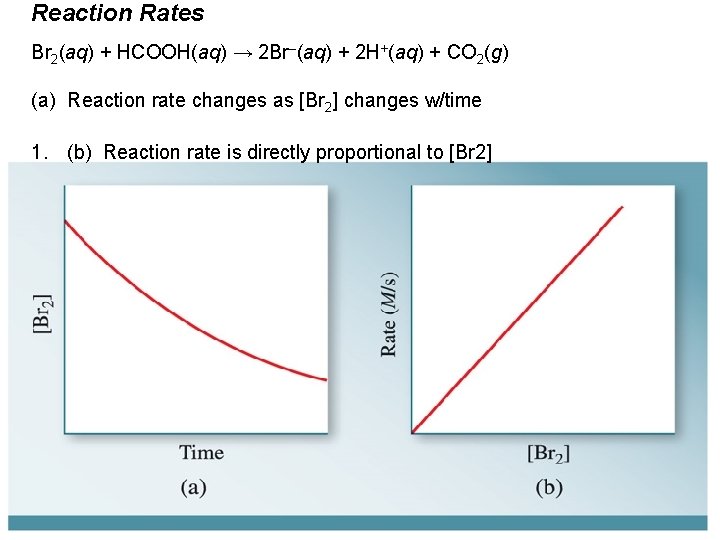
Reaction Rates Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) (a) Reaction rate changes as [Br 2] changes w/time 1. (b) Reaction rate is directly proportional to [Br 2]

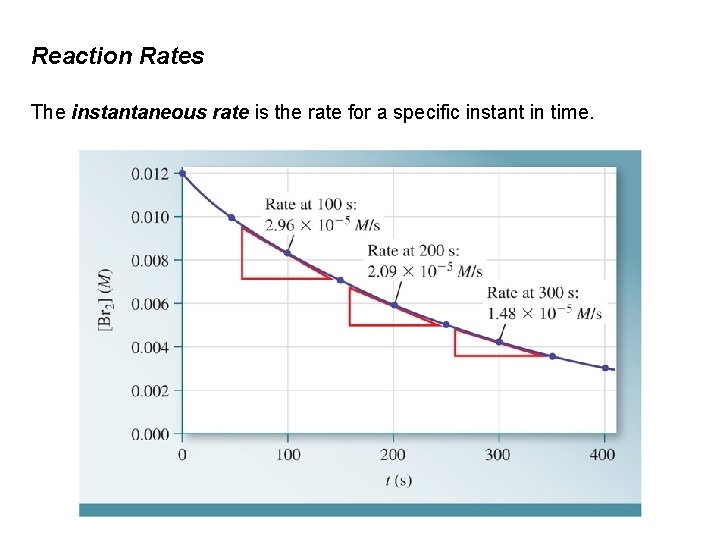
Reaction Rates The instantaneous rate is the rate for a specific instant in time.

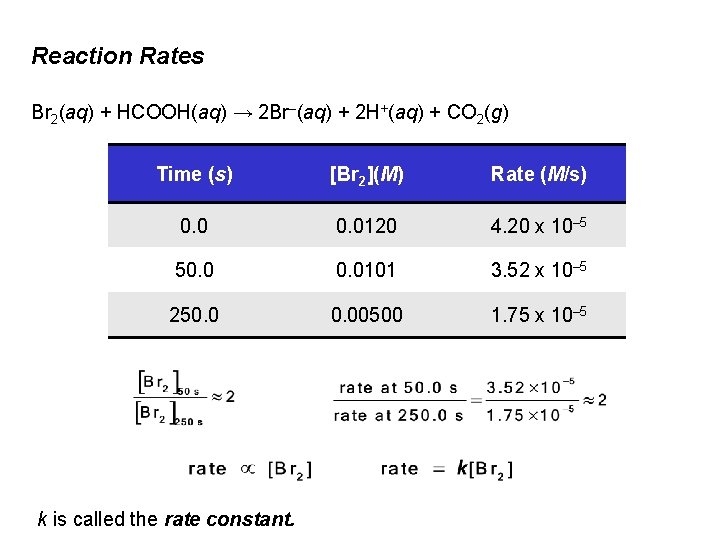
Reaction Rates Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) Time (s) [Br 2](M) Rate (M/s) 0. 0120 4. 20 x 10– 5 50. 0101 3. 52 x 10– 5 250. 00500 1. 75 x 10– 5 k is called the rate constant.

Reaction Rates Br 2(aq) + HCOOH(aq) → 2 Br–(aq) + 2 H+(aq) + CO 2(g) Time (s) [Br 2](M) Rate (M/s) 0. 0120 4. 20 x 10– 5 50. 0101 3. 52 x 10– 5 250. 00500 1. 75 x 10– 5 at t = 50. 0 s

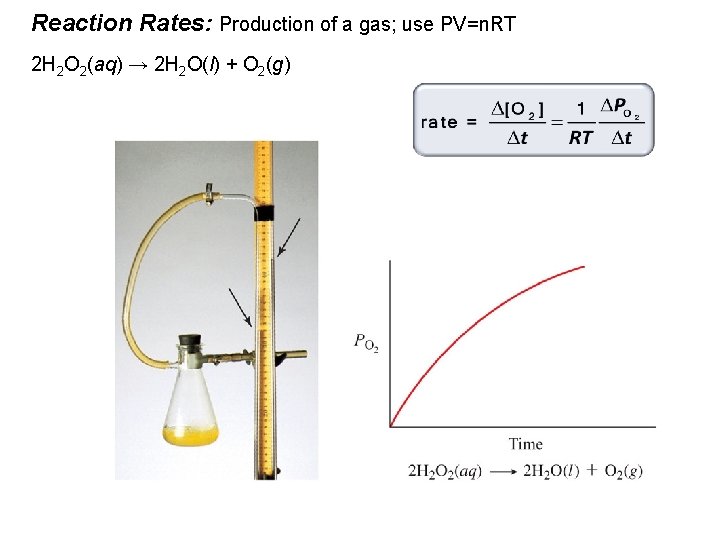
Reaction Rates: Production of a gas; use PV=n. RT 2 H 2 O 2(aq) → 2 H 2 O(l) + O 2(g)

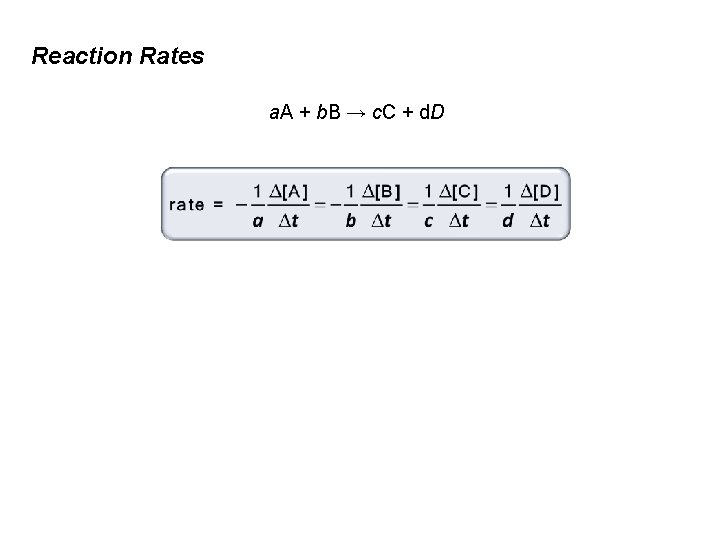
Reaction Rates a. A + b. B → c. C + d. D

Reaction Rates Write the rate expressions for the following reaction: CO 2(g) + 2 H 2 O(g) → CH 4(g) + 2 O 2(g) Solution: Use the equation below to write the rate expressions

14. 2 Dependence of Reaction Rate on Reactant Concentration The rate law is an equation that relates the rate of reaction to the concentrations of reactants. a. A + b. B → c. C + d. D rate = k[A]x[B]y k: x: y: the rate constant order with respect to A order with respect to B x + y: determined experimentally represents the overall reaction order.

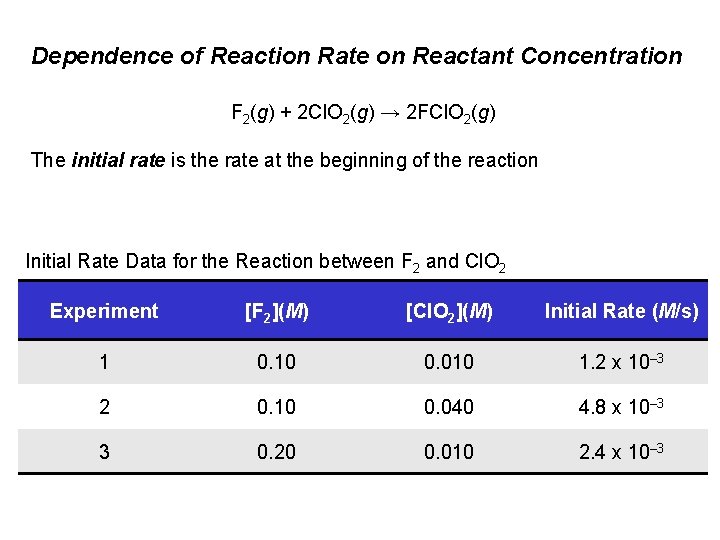
Dependence of Reaction Rate on Reactant Concentration F 2(g) + 2 Cl. O 2(g) → 2 FCl. O 2(g) The initial rate is the rate at the beginning of the reaction Initial Rate Data for the Reaction between F 2 and Cl. O 2 Experiment [F 2](M) [Cl. O 2](M) Initial Rate (M/s) 1 0. 10 0. 010 1. 2 x 10– 3 2 0. 10 0. 040 4. 8 x 10– 3 3 0. 20 0. 010 2. 4 x 10– 3
![Dependence of Reaction Rate on Reactant Concentration rate kF 2xCl O 2y F Dependence of Reaction Rate on Reactant Concentration rate = k[F 2]x[Cl. O 2]y F](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-19.jpg)
Dependence of Reaction Rate on Reactant Concentration rate = k[F 2]x[Cl. O 2]y F 2(g) + 2 Cl. O 2(g) → 2 FCl. O 2(g) Initial Rate Data for the Reaction between F 2 and Cl. O 2 Experiment [F 2](M) [Cl. O 2](M) Initial Rate (M/s) 1 0. 10 0. 010 1. 2 x 10– 3 2 [F 2] 0. 10 doubles 3 0. 20 [Cl. O 2] Rate 0. 040 4. 8 x 10– 3 constant doubles The reaction is first order in F 2; x = 1 0. 010 2. 4 x 10– 3 rate = k[F 2][Cl. O 2]y
![Dependence of Reaction Rate on Reactant Concentration rate kF 2 Cl O 2y Dependence of Reaction Rate on Reactant Concentration rate = k[F 2] [Cl. O 2]y](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-20.jpg)
Dependence of Reaction Rate on Reactant Concentration rate = k[F 2] [Cl. O 2]y F 2(g) + 2 Cl. O 2(g) → 2 FCl. O 2(g) Initial Rate Data for the Reaction between F 2 and Cl. O 2 Experiment 1 [F 2](M) [Cl. O 2](M) Initial Rate (M/s) 2 [F 2] 0. 10 constant 0. 10 [Cl. O 2] 0. 010 x 4 0. 040 – 3 1. 2 x 10 Rate x 4 4. 8 x 10– 3 3 0. 20 0. 010 2. 4 x 10– 3 The reaction is first order in Cl. O 2; y = 1 rate = k[F 2][Cl. O 2]
![Dependence of Reaction Rate on Reactant Concentration rate kAxBy a A b Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b.](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-21.jpg)
Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b. B → c. C + d. D Initial Rate Data for the Reaction between A and B Experiment [A](M) [B](M) Initial Rate (M/s) 1 0. 10 0. 015 2. 1 x 10– 4 2 0. 20 0. 015 4. 2 x 10– 4 3 0. 10 0. 030 8. 4 x 10– 4
![Dependence of Reaction Rate on Reactant Concentration rate kAxBy a A b Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b.](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-22.jpg)
Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b. B → c. C + d. D Initial Rate Data for the Reaction between A and B Experiment 1 [F 2](M) [Cl. O 2](M) Initial Rate (M/s) 2 [F 2] 0. 10 x 2 0. 20 [Cl. O 2] 0. 015 constant 0. 015 – 4 2. 1 x 10 Rate x 2 4. 2 x 10– 4 3 0. 10 0. 030 8. 4 x 10– 4 The reaction is first order in A; x = 1 rate = k[A][B]y
![Dependence of Reaction Rate on Reactant Concentration rate kAxBy a A b Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b.](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-23.jpg)
Dependence of Reaction Rate on Reactant Concentration rate = k[A]x[B]y a. A + b. B → c. C + d. D Initial Rate Data for the Reaction between A and B Experiment [A](M) [B](M) Initial Rate (M/s) 1 0. 10 0. 015 2. 1 x 10– 4 2 [A] 0. 20 constant [B] 0. 015 x 2 Rate 4. 2 x 10– 4 x 4 3 0. 10 0. 030 8. 4 x 10– 4 The reaction is second order in B; y = 2 rate = k[A][B]2

Dependence of Reaction Rate on Reactant Concentration Three important things to remember about the rate law: 1) The exponents in a rate law must be determined from a table of experimental data. 2) Comparing changes in individual reactant concentrations with changes in rate shows how the rate depends on each reactant concentration. 3) Reaction order is always defined in terms of reactant concentrations, never product concentrations.

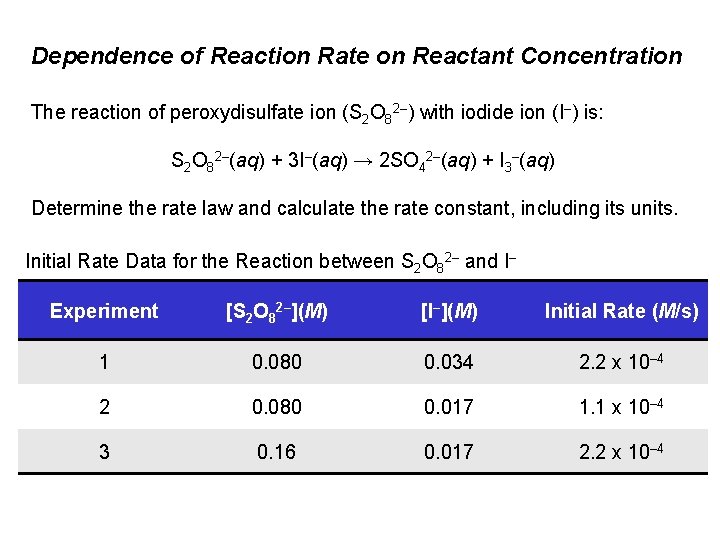
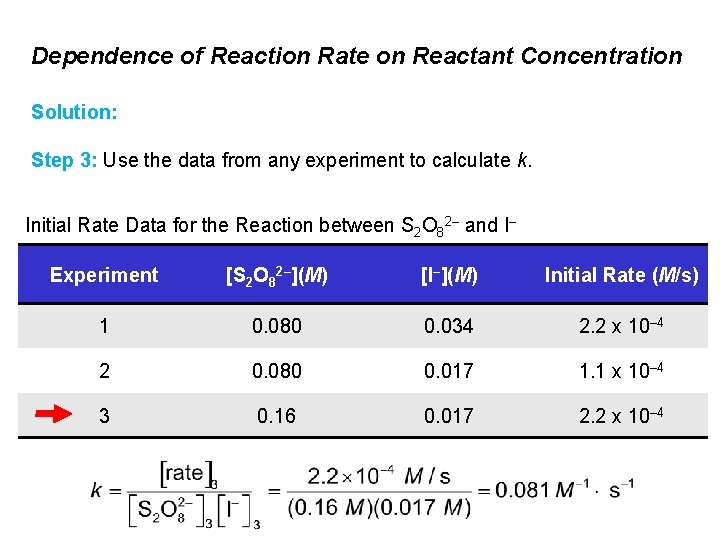
Dependence of Reaction Rate on Reactant Concentration The reaction of peroxydisulfate ion (S 2 O 82–) with iodide ion (I–) is: S 2 O 82–(aq) + 3 I–(aq) → 2 SO 42–(aq) + I 3–(aq) Determine the rate law and calculate the rate constant, including its units. Initial Rate Data for the Reaction between S 2 O 82– and I– Experiment [S 2 O 82–](M) [I–](M) Initial Rate (M/s) 1 0. 080 0. 034 2. 2 x 10– 4 2 0. 080 0. 017 1. 1 x 10– 4 3 0. 16 0. 017 2. 2 x 10– 4

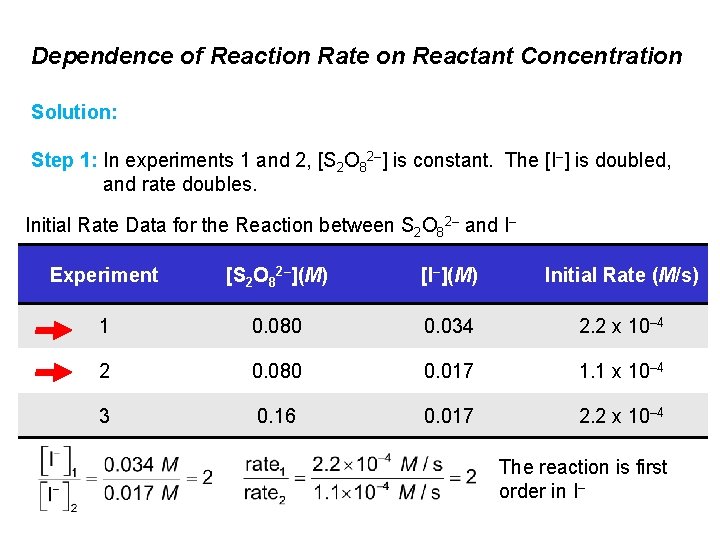
Dependence of Reaction Rate on Reactant Concentration Solution: Step 1: In experiments 1 and 2, [S 2 O 82–] is constant. The [I–] is doubled, and rate doubles. Initial Rate Data for the Reaction between S 2 O 82– and I– Experiment [S 2 O 82–](M) [I–](M) Initial Rate (M/s) 1 0. 080 0. 034 2. 2 x 10– 4 2 0. 080 0. 017 1. 1 x 10– 4 3 0. 16 0. 017 2. 2 x 10– 4 The reaction is first order in I–

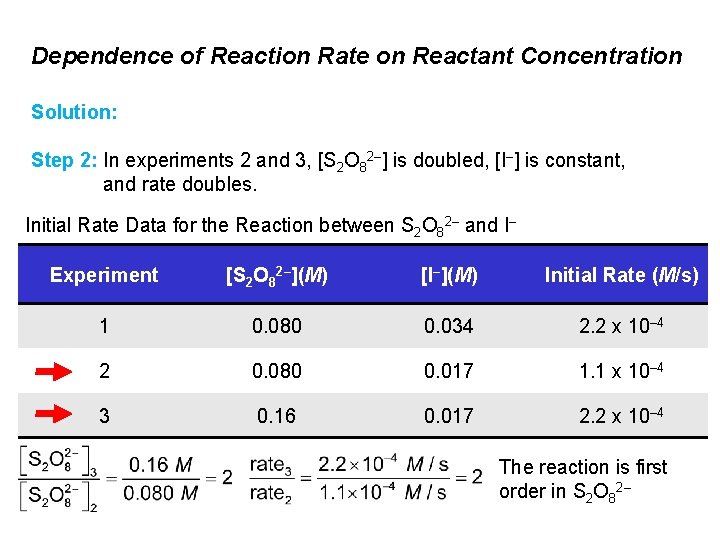
Dependence of Reaction Rate on Reactant Concentration Solution: Step 2: In experiments 2 and 3, [S 2 O 82–] is doubled, [I–] is constant, and rate doubles. Initial Rate Data for the Reaction between S 2 O 82– and I– Experiment [S 2 O 82–](M) [I–](M) Initial Rate (M/s) 1 0. 080 0. 034 2. 2 x 10– 4 2 0. 080 0. 017 1. 1 x 10– 4 3 0. 16 0. 017 2. 2 x 10– 4 The reaction is first order in S 2 O 82–

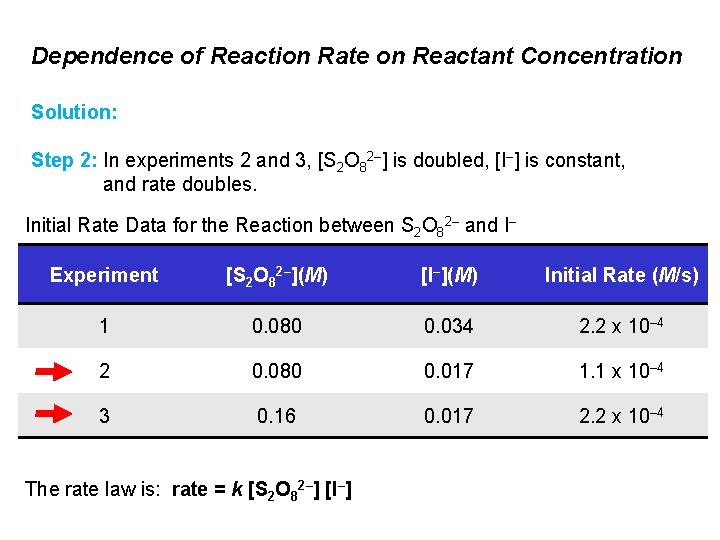
Dependence of Reaction Rate on Reactant Concentration Solution: Step 2: In experiments 2 and 3, [S 2 O 82–] is doubled, [I–] is constant, and rate doubles. Initial Rate Data for the Reaction between S 2 O 82– and I– Experiment [S 2 O 82–](M) [I–](M) Initial Rate (M/s) 1 0. 080 0. 034 2. 2 x 10– 4 2 0. 080 0. 017 1. 1 x 10– 4 3 0. 16 0. 017 2. 2 x 10– 4 The rate law is: rate = k [S 2 O 82–] [I–]

Dependence of Reaction Rate on Reactant Concentration Solution: Step 3: Use the data from any experiment to calculate k. Initial Rate Data for the Reaction between S 2 O 82– and I– Experiment [S 2 O 82–](M) [I–](M) Initial Rate (M/s) 1 0. 080 0. 034 2. 2 x 10– 4 2 0. 080 0. 017 1. 1 x 10– 4 3 0. 16 0. 017 2. 2 x 10– 4

14. 3 Dependence of Reactant Concentration on Time The rate law can be used to determine the rate of a reaction using the rate constant and the reactant concentrations: rate law rate = k[A]x[B]y rate constant A rate law can also be used to determine the concentration of a reactant at a specific time during a reaction.

Dependence of Reactant Concentration on Time A first-order reaction is a reaction whose rate depends on the concentration of one of the reactants raised to the first power. C 2 H 6 → 2 ·CH 3 rate = k[C 2 H 6] 2 N 2 O 5(g) → 2 NO 2(g) + O 2(g) rate = k[N 2 O 5]

Dependence of Reactant Concentration on Time In a first-order reaction of the type A → products The rate can be expressed as the rate of change in reactant concentration, as well as in the form of the rate law: rate = k[A] Setting the two expressions equal to each other yields:

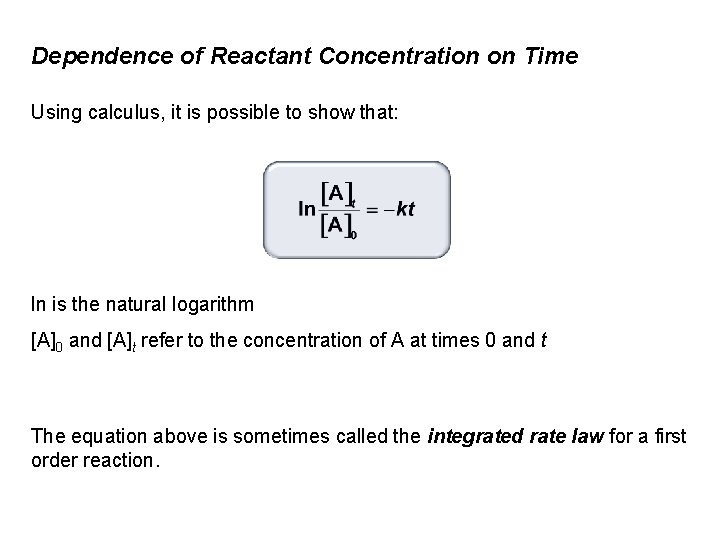
Dependence of Reactant Concentration on Time Using calculus, it is possible to show that: ln is the natural logarithm [A]0 and [A]t refer to the concentration of A at times 0 and t The equation above is sometimes called the integrated rate law for a first order reaction.

Dependence of Reactant Concentration on Time The rate constant for the reaction 2 A → B is 7. 5 x 10– 3 s– 1 at 110°C. The reaction is first order in A. How long (in seconds) will it take for [A] to decrease from 1. 25 M to 0. 71 M? Solution: Step 1: Use the equation below to calculate time in seconds. t = 75 seconds

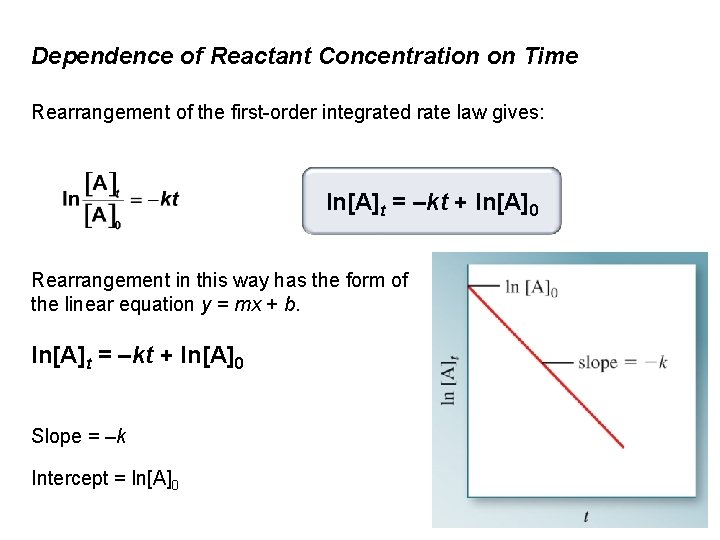
Dependence of Reactant Concentration on Time Rearrangement of the first-order integrated rate law gives: ln[A]t = –kt + ln[A]0 Rearrangement in this way has the form of the linear equation y = mx + b. ln[A]t = –kt + ln[A]0 Slope = –k Intercept = ln[A]0

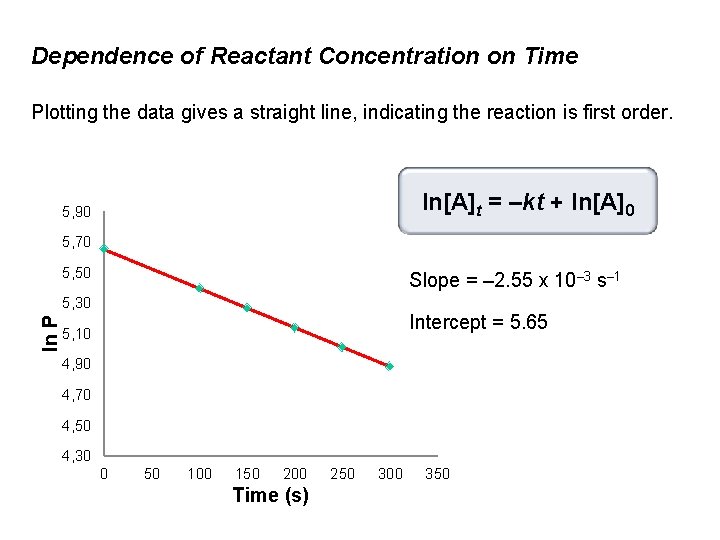
Dependence of Reactant Concentration on Time The rate of decomposition of azomethane is studied by monitoring the partial pressure of the reactant as a function of time. CH 3—N=N—CH 3(g) → N 2(g) + C 2 H 6(g) The data obtained at 300°C are listed in the following table: Time (s) Pazomethane (mm. Hg) 0 284 100 220 150 193 200 170 250 150 300 132

Dependence of Reactant Concentration on Time Plotting the data gives a straight line, indicating the reaction is first order. ln[A]t = –kt + ln[A]0 5, 90 5, 70 5, 50 Slope = – 2. 55 x 10– 3 s– 1 ln P 5, 30 Intercept = 5. 65 5, 10 4, 90 4, 70 4, 50 4, 30 0 50 100 150 200 Time (s) 250 300 350

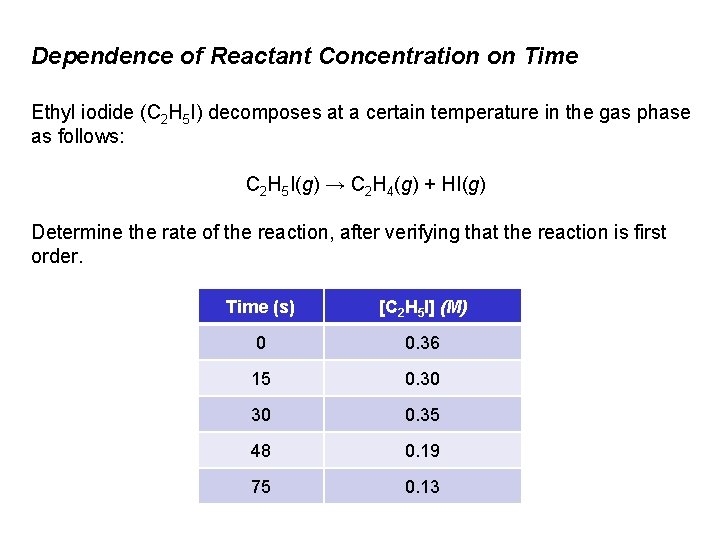
Dependence of Reactant Concentration on Time Ethyl iodide (C 2 H 5 I) decomposes at a certain temperature in the gas phase as follows: C 2 H 5 I(g) → C 2 H 4(g) + HI(g) Determine the rate of the reaction, after verifying that the reaction is first order. Time (s) [C 2 H 5 I] (M) 0 0. 36 15 0. 30 30 0. 35 48 0. 19 75 0. 13
![Dependence of Reactant Concentration on Time Solution Plot lnC 2 H 5 I vs Dependence of Reactant Concentration on Time Solution: Plot ln[C 2 H 5 I] vs](https://slidetodoc.com/presentation_image_h2/98bd42706c89970a835b6a85ccb4e0a6/image-39.jpg)
Dependence of Reactant Concentration on Time Solution: Plot ln[C 2 H 5 I] vs time. If a straight line results, the reaction is first order. The slope is equal to k. 0, 00 Time (s) [C 2 H 5 I] (M) ln[C 2 H 5 I] 0 0. 36 -1. 02 15 0. 30 -1. 20 30 0. 35 -1. 39 48 0. 19 -1. 66 -2, 00 75 0. 13 -2. 04 -2, 50 Slope = – 1. 3 x 10– 2 s– 1; k = 1. 3 x 10– 2 s– 1 ln [C 2 H 5 I] 0 20 40 -0, 50 -1, 00 -1, 50 Time (s) 60 80

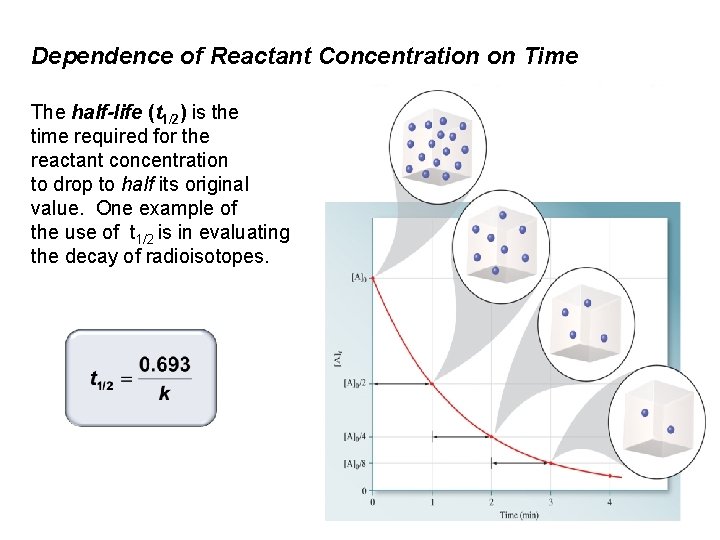
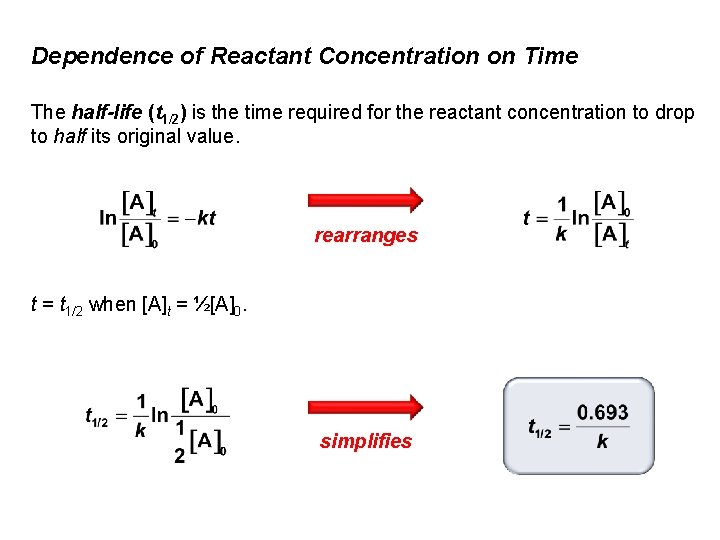
Dependence of Reactant Concentration on Time The half-life (t 1/2) is the time required for the reactant concentration to drop to half its original value. One example of the use of t 1/2 is in evaluating the decay of radioisotopes.

Dependence of Reactant Concentration on Time The half-life (t 1/2) is the time required for the reactant concentration to drop to half its original value. rearranges t = t 1/2 when [A]t = ½[A]0. simplifies

Dependence of Reactant Concentration on Time Calculate the half-life of the decomposition of azomethane, k = 2. 55 x 10– 3. Solution: Step 1: Use the equation below to calculate half-life:

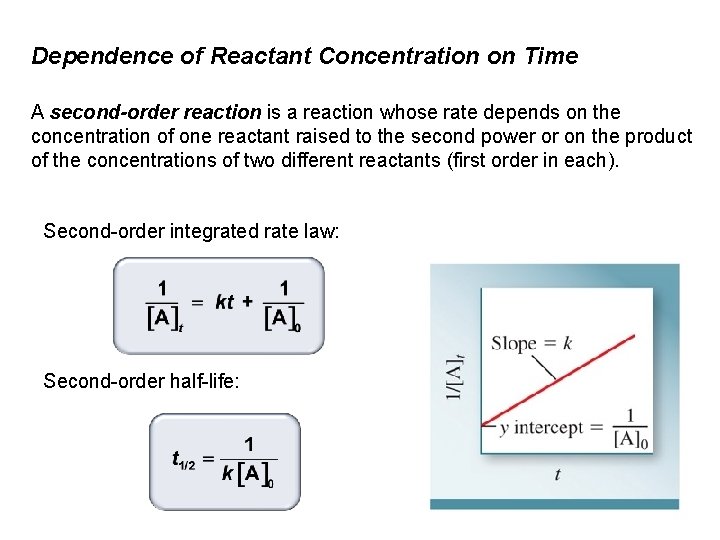
Dependence of Reactant Concentration on Time A second-order reaction is a reaction whose rate depends on the concentration of one reactant raised to the second power or on the product of the concentrations of two different reactants (first order in each). Second-order integrated rate law: Second-order half-life:

Dependence of Reactant Concentration on Time The rate of a zero-order reaction is a constant. Third-order and higher are rare.

14. 4 Dependence of Reaction Rate on Temperature Most reactions happen faster at higher temperature. Chemical reactions generally occur as a result of collisions between reacting molecules. According to collision theory of chemical kinetics, the reaction rate is directly proportional to the number of molecular collisions per second:

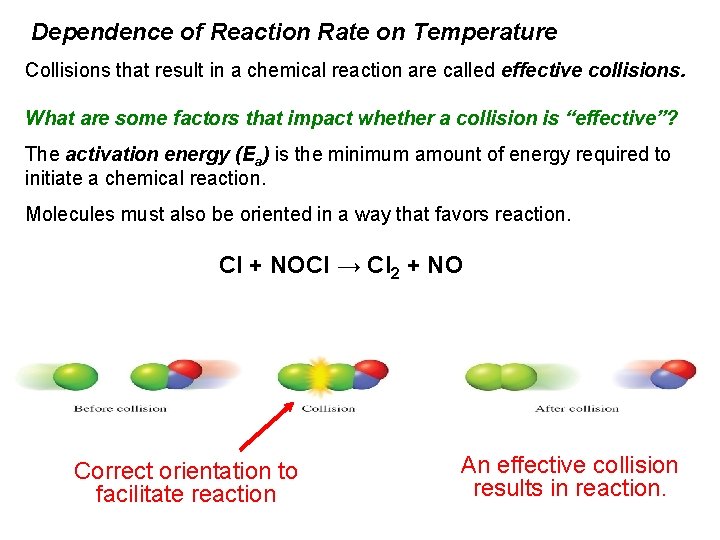
Dependence of Reaction Rate on Temperature Collisions that result in a chemical reaction are called effective collisions. What are some factors that impact whether a collision is “effective”? The activation energy (Ea) is the minimum amount of energy required to initiate a chemical reaction. Molecules must also be oriented in a way that favors reaction. Cl + NOCl → Cl 2 + NO Correct orientation to facilitate reaction An effective collision results in reaction.

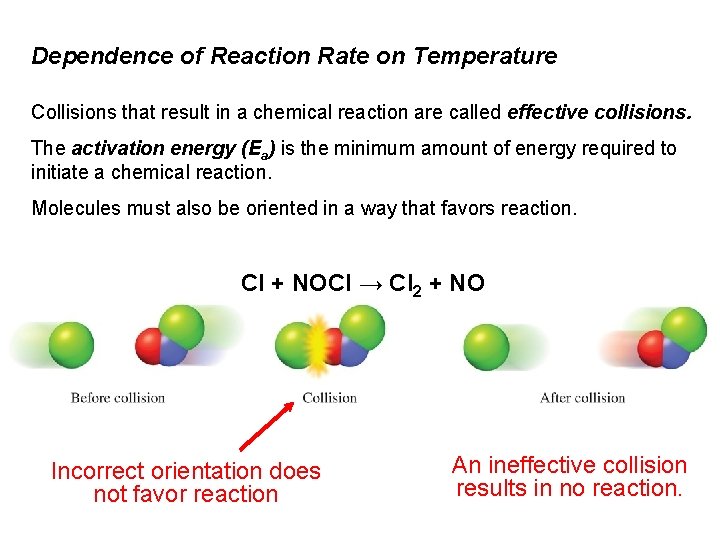
Dependence of Reaction Rate on Temperature Collisions that result in a chemical reaction are called effective collisions. The activation energy (Ea) is the minimum amount of energy required to initiate a chemical reaction. Molecules must also be oriented in a way that favors reaction. Cl + NOCl → Cl 2 + NO Incorrect orientation does not favor reaction An ineffective collision results in no reaction.

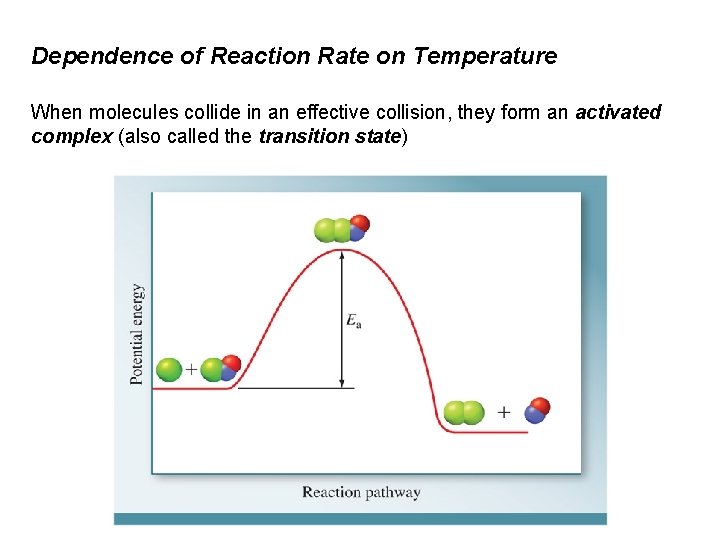
Dependence of Reaction Rate on Temperature When molecules collide in an effective collision, they form an activated complex (also called the transition state)

Dependence of Reaction Rate on Temperature The dependence of the rate constant on temperature can be expressed by the Arrhenius equation. A represents the collision frequency and is called the frequency factor. Ea is the activation energy (in k. J/mol) R is the gas constant (8. 314 J/mol K) T is the absolute temperature e is the base of the natural logarithm

Dependence of Reaction Rate on Temperature Taking the natural log of both sides, the Arrhenius equation may be written as: Rearrangement gives the linear form of the Arrhenius equation:

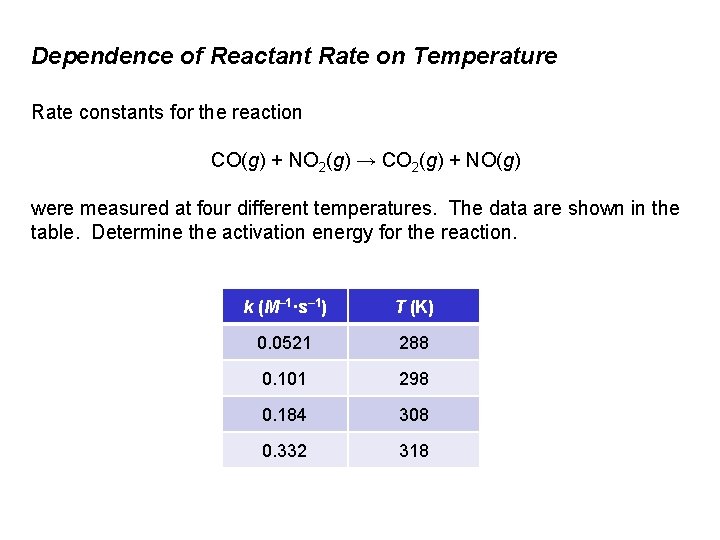
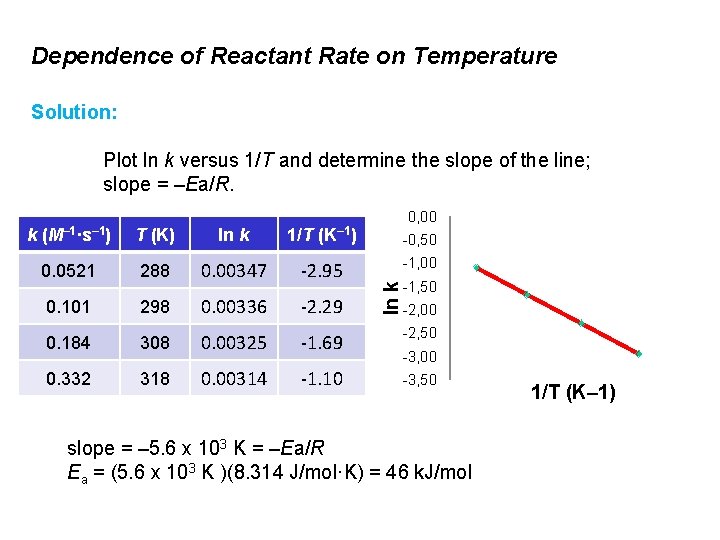
Dependence of Reactant Rate on Temperature Rate constants for the reaction CO(g) + NO 2(g) → CO 2(g) + NO(g) were measured at four different temperatures. The data are shown in the table. Determine the activation energy for the reaction. k (M– 1·s– 1) T (K) 0. 0521 288 0. 101 298 0. 184 308 0. 332 318

Dependence of Reactant Rate on Temperature Solution: Plot ln k versus 1/T and determine the slope of the line; slope = –Ea/R. 1/T 0, 00 (K– 1) T (K) ln k 0. 0521 288 0. 00347 -2. 95 0. 101 298 0. 00336 -2. 29 0. 184 308 0. 00325 -1. 69 -2, 50 0. 332 318 0. 00314 -1. 10 -3, 50 -0, 50 -1, 00 ln k k (M– 1·s– 1) -1, 50 -2, 00 -3, 00 slope = – 5. 6 x 103 K = –Ea/R Ea = (5. 6 x 103 K )(8. 314 J/mol·K) = 46 k. J/mol 1/T (K– 1)

Dependence of Reactant Rate on Temperature A two point form of the Arrhenius equation may be written: If the rate constants at two different temperatures are known, it is possible to calculate the activation energy. If the activation energy and the rate constant at one temperature are known, it is possible to determine the rate constant at any other temperature.

14. 5 Reaction Mechanisms A balanced chemical equation does not indicate how a reaction actually takes place. The sequence of steps that sum to give the overall reaction is called the reaction mechanism. Step 1: A + B → C Step 2: C + B → D Overall reaction: A + 2 B → D

Reaction Mechanism Chemical species that appear in the reaction mechanism, but not in the overall chemical equation are called intermediates. Step 1: NO + NO N 2 O 2 Step 2: N 2 O 2 + O 2 2 NO 2 Overall reaction: 2 NO + O 2 2 NO 2

Reaction Mechanism Each step in a reaction mechanism represents an elementary reaction, one that occurs in a single collision of the reactant molecules. The molecularity of an elementary reaction is essentially the number of reactant molecules involved in the collision. unimolecular (one reactant molecule) A → products rate = k[A] first order bimolecular (two reactant molecules) A + B → products rate = k[A][B] second order A + A → products rate = k[A]2 second order termolecular (three reactant molecules)

Reaction Mechanism In a reaction mechanism consisting of more than elementary step, the rate law for the overall process is given by the rate-determining step. The rate determining step is the slowest step in the sequence. A proposed mechanism must satisfy two requirements: The sum of the elementary reaction must be the overall balanced equation for the reaction. The rate determining step must have the same rate law as that determined from the experimental data.

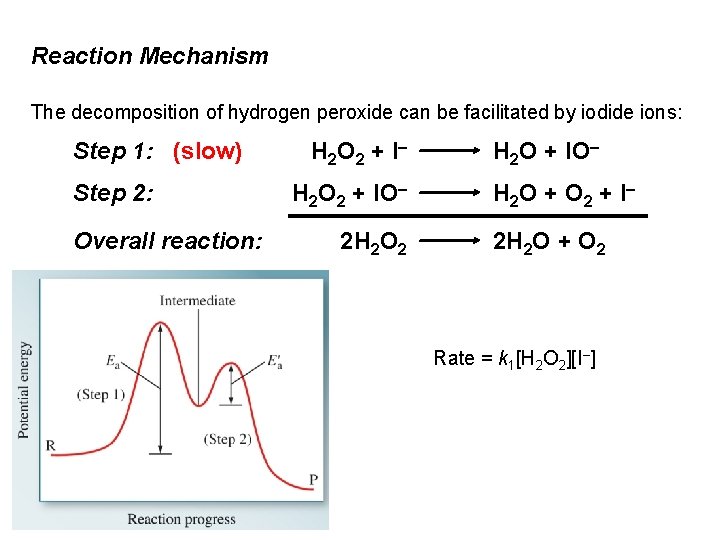
Reaction Mechanism The decomposition of hydrogen peroxide can be facilitated by iodide ions: Step 1: (slow) Step 2: Overall reaction: H 2 O 2 + I – H 2 O 2 + IO– 2 H 2 O 2 H 2 O + IO– H 2 O + O 2 + I – 2 H 2 O + O 2 Rate = k 1[H 2 O 2][I–]

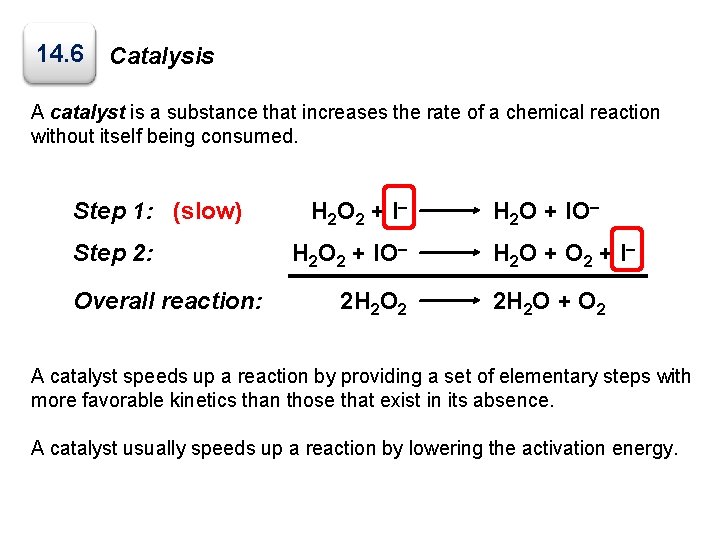
14. 6 Catalysis A catalyst is a substance that increases the rate of a chemical reaction without itself being consumed. Step 1: (slow) Step 2: Overall reaction: H 2 O 2 + I – H 2 O 2 + IO– 2 H 2 O 2 H 2 O + IO– H 2 O + O 2 + I – 2 H 2 O + O 2 A catalyst speeds up a reaction by providing a set of elementary steps with more favorable kinetics than those that exist in its absence. A catalyst usually speeds up a reaction by lowering the activation energy.

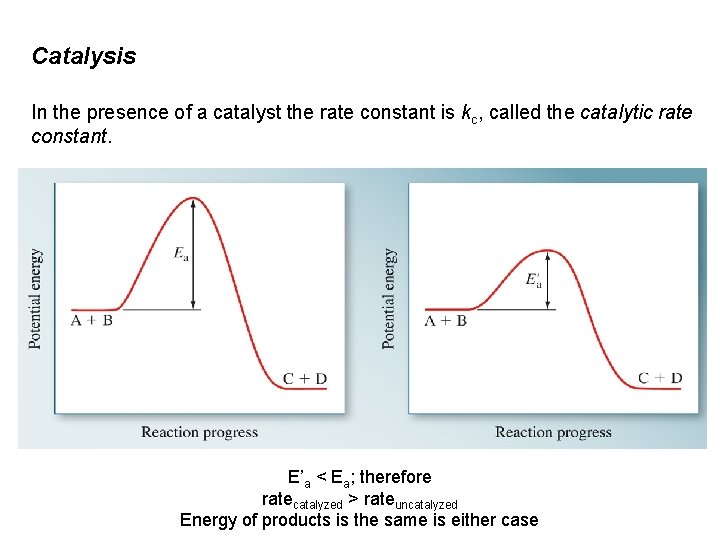
Catalysis In the presence of a catalyst the rate constant is kc, called the catalytic rate constant. E’a < Ea; therefore ratecatalyzed > rateuncatalyzed Energy of products is the same is either case

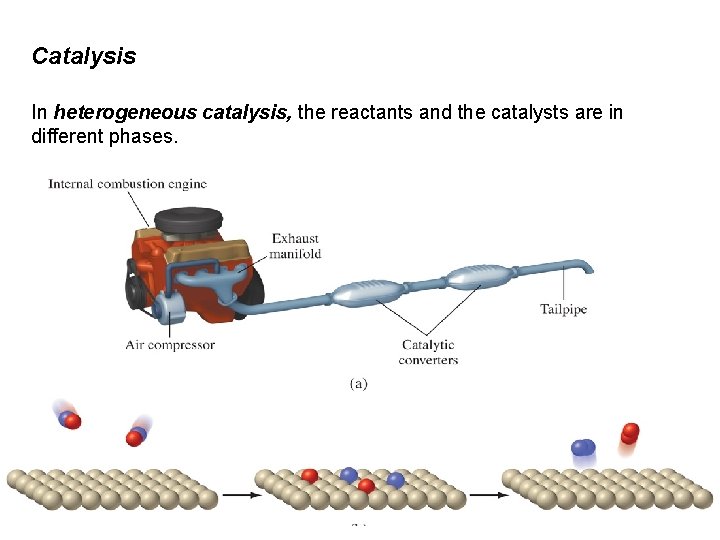
Catalysis In heterogeneous catalysis, the reactants and the catalysts are in different phases.

Catalysis In homogeneous catalysis, the reactants and catalyst are dispersed in a single phase, usually liquid. Advantages: Reactions can be carried out under atmospheric conditions. Can be designed to function selectively Are generally cheaper.

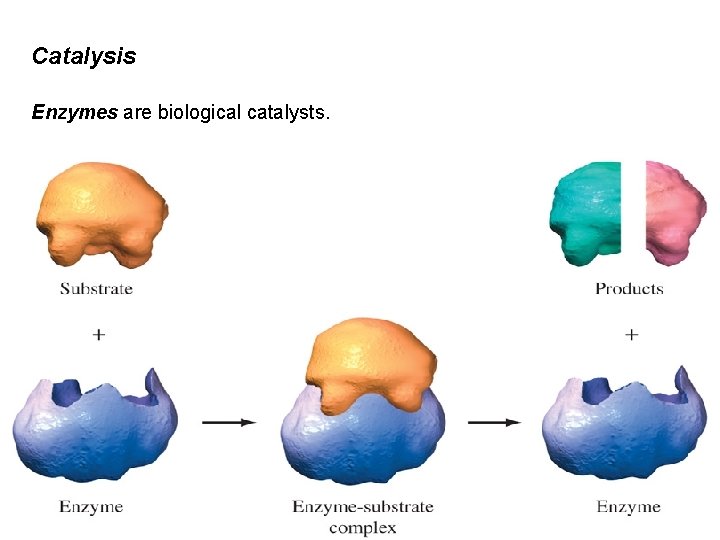
Catalysis Enzymes are biological catalysts.

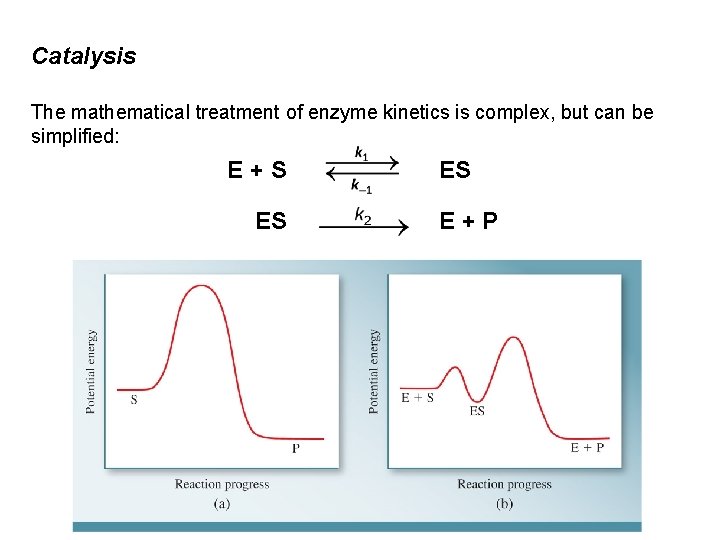
Catalysis The mathematical treatment of enzyme kinetics is complex, but can be simplified: E+S ES ES E+P

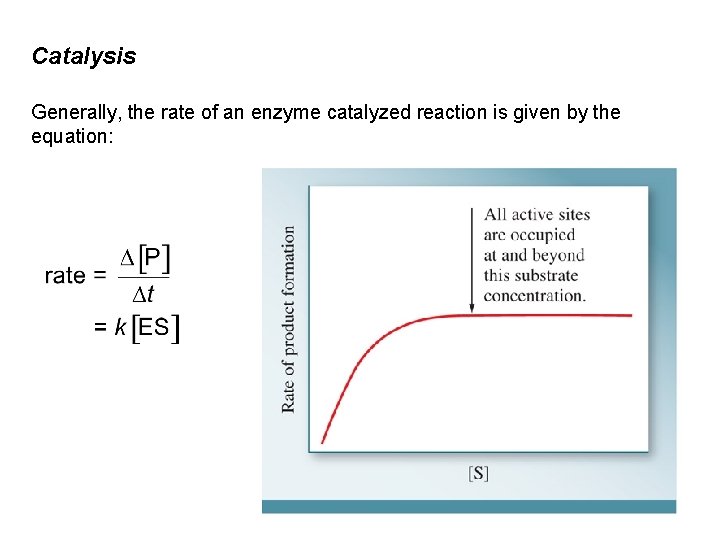
Catalysis Generally, the rate of an enzyme catalyzed reaction is given by the equation:

14 Key Points Average Reaction Rates Instantaneous Rate Stoichiometry and Reaction Rate The Rate Law Experimental Determination of the Rate Law First-Order Reactions Second-Order Reactions Collision Theory Elementary Reactions Rate-Determining Step Experimental Support for Reaction Mechanisms Heterogeneous Catalysis Homogeneous Catalysis Enzymes: Biological Catalysts