14 3 NOTES Beyond Alkanes Beyond Alkanes Saturated

14. 3 – NOTES Beyond Alkanes

• Beyond Alkanes • Saturated hydrocarbons • Each carbon atom forms as many single covalent bonds as it can • All alkanes are saturated molecules • Points of unsaturation • Double bonds have 1 point(s) of unsaturation • Triple bonds have 2 point(s) of unsaturation • Rings have 1 point(s) of unsaturation
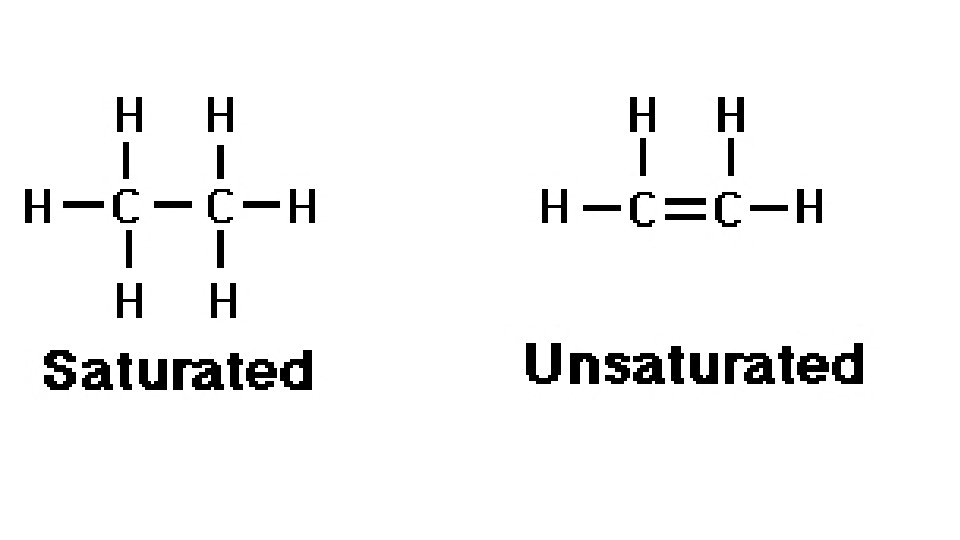
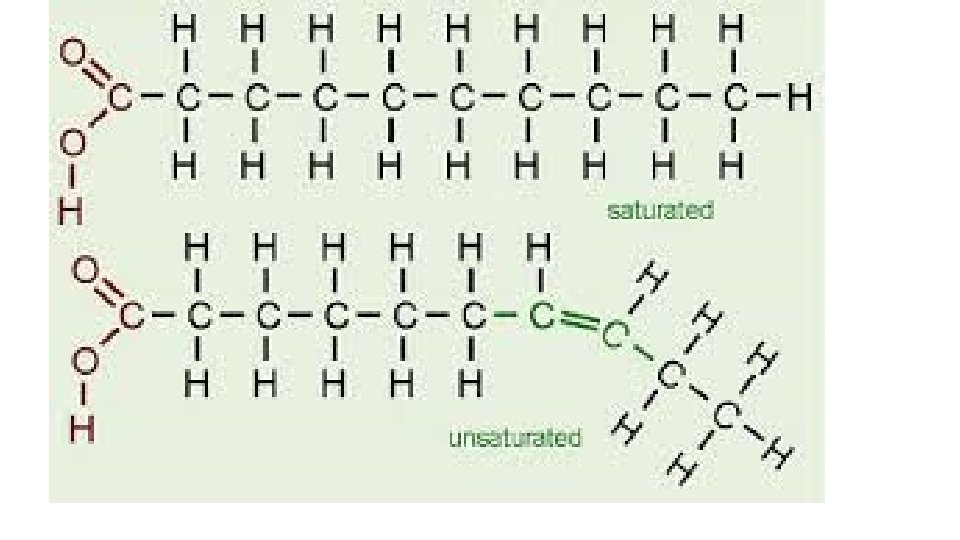

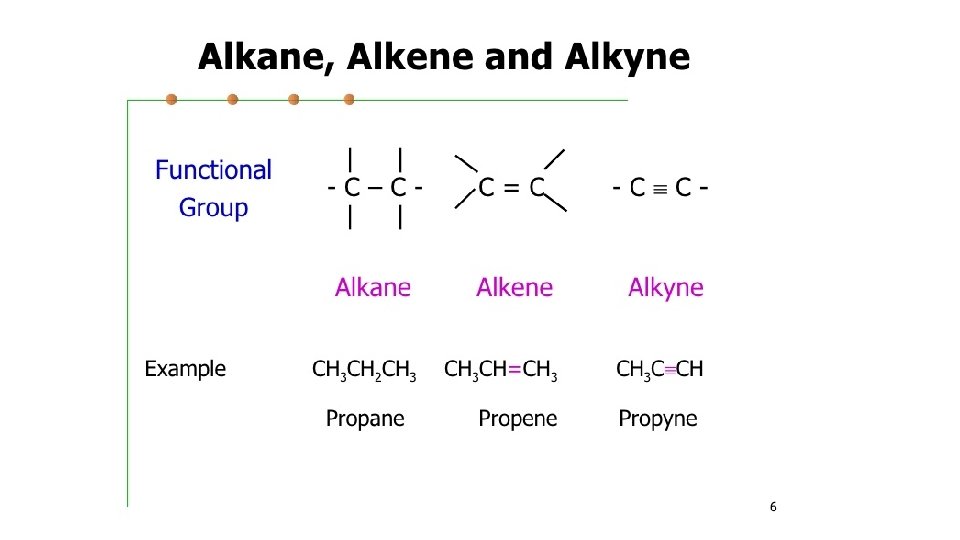
• Alkenes • Hydrocarbon with at least 1 double bond • • Unsaturated hydrocarbons • • More chemically reactive than alkanes because they are able to break the double bond to bond with other atoms • • General formula- Cn. H 2 n •
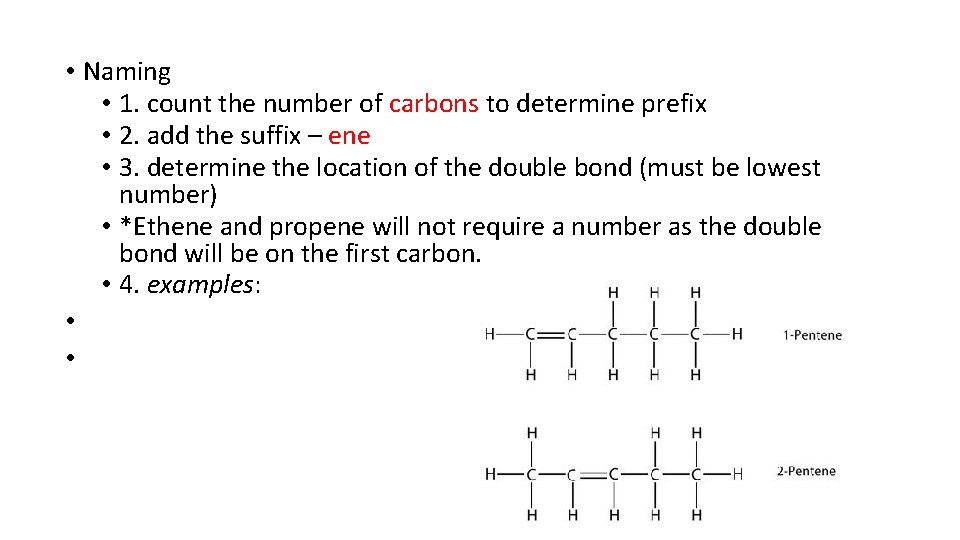
• Naming • 1. count the number of carbons to determine prefix • 2. add the suffix – ene • 3. determine the location of the double bond (must be lowest number) • *Ethene and propene will not require a number as the double bond will be on the first carbon. • 4. examples: • •

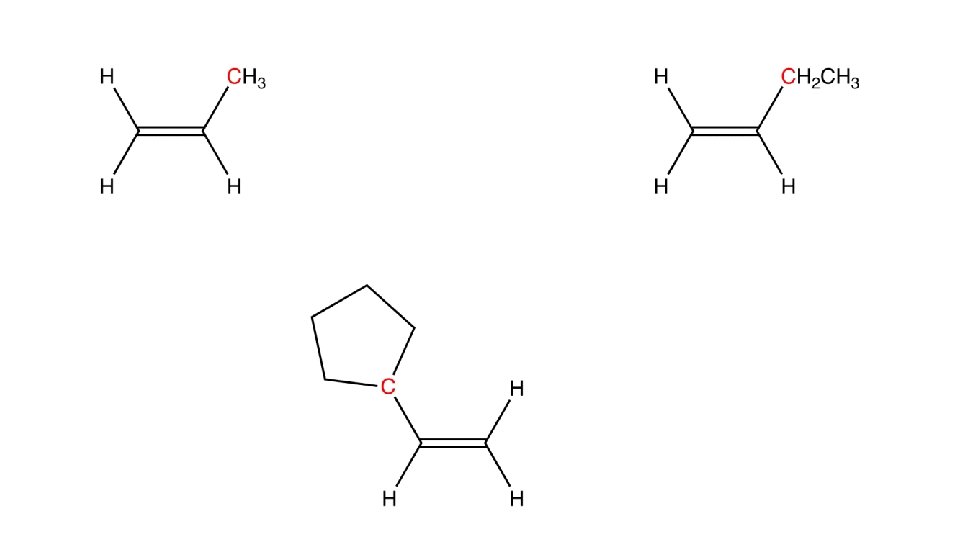
• Substituted alkenes • They contain one or more other atoms besides C and H • O, N, Cl, S • Changes reactivity • Two different substituted alkenes may share molecular formulas, but the arrangement/structural formula of the molecule may be very different

• Alkynes • Hydrocarbon with a triple bond • • General formula- • Cn. H 2 n-2

• Naming • 1. count the number of carbons to determine prefix • 2. add the suffix –yne • 3. determine the location of the triple bond (must be lowest number) • *ethyne and propyne will not require a number as the triple bond will be on the first carbon. • 4. examples:

• More Builder Molecules • Some carbons are joined in rings, rather than in straight lines • Cycloalkanes • Saturated hydrocarbons made of singly bonded carbons in a ring form • • General formula- Cn. H 2 n
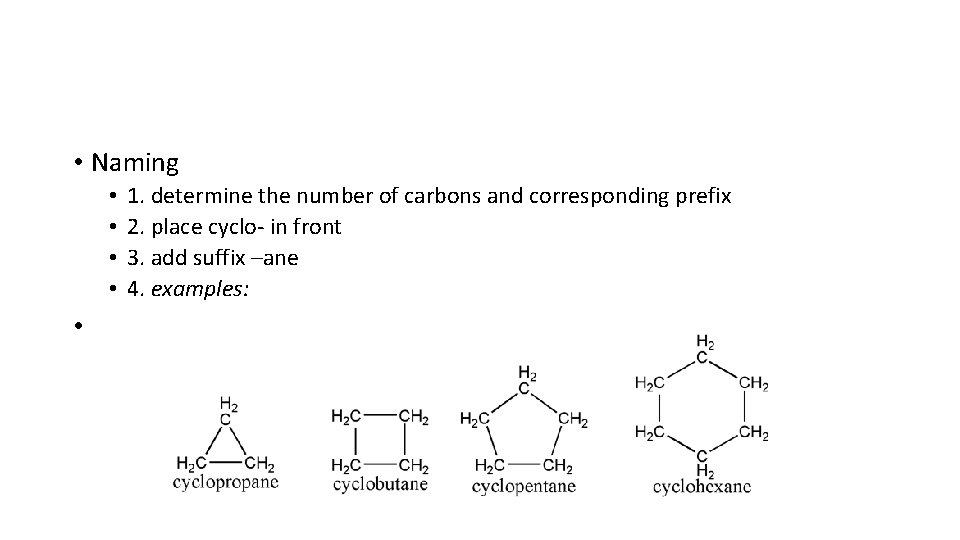
• Naming • • • 1. determine the number of carbons and corresponding prefix 2. place cyclo- in front 3. add suffix –ane 4. examples:

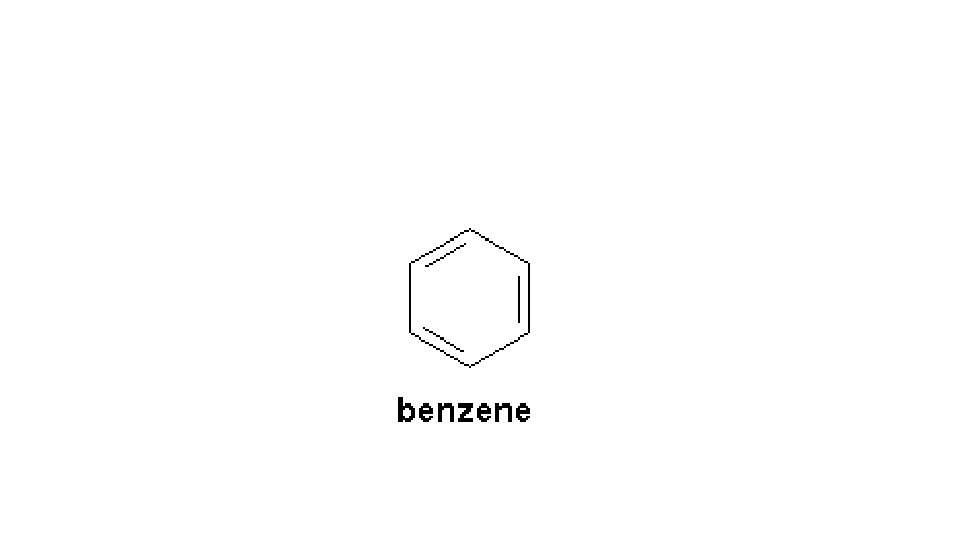
• Aromatic compounds • Aromatics involve double bonds within the ring • Behave like they DON”T have double bonds- not reactive like noncyclical double bonded compounds • Discovered that the double bond is shared among all carbons equally. • Shown with a circle in the center representing the double bonding • Very different from cycloalkanes • The most common aromatic compound is benzene, C 6 H 6 • Structure (next slide) • • • Uses: perfumes, dyes
- Slides: 15