13 2 Solubility and Factors Affecting Solubility Dr

13. 2 Solubility and Factors Affecting Solubility Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Solubility December 20

Properties of Solution Characteristics: • Distribution of particles is uniform • Components in solution do not separate upon standing • Components cannot be separated by filtration. • Solute / Solvent mixes in ratios - up to the solubility limit. • Solution is almost always transparent. • Compounds of solution may be separated by other methods i. e. , distillation or chromatography. 2 Solubility December 20
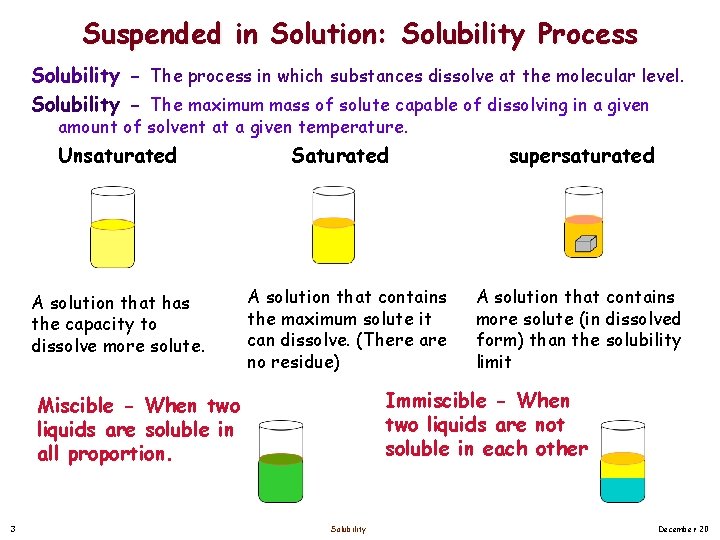
Suspended in Solution: Solubility Process Solubility - The process in which substances dissolve at the molecular level. Solubility - The maximum mass of solute capable of dissolving in a given amount of solvent at a given temperature. Unsaturated A solution that has the capacity to dissolve more solute. Saturated A solution that contains the maximum solute it can dissolve. (There are no residue) A solution that contains more solute (in dissolved form) than the solubility limit Immiscible - When two liquids are not soluble in each other Miscible - When two liquids are soluble in all proportion. 3 supersaturated Solubility December 20

1) Nature of Solute and Solvent Dissolving Process: Why is water soluble in alcohol yet water is insoluble in oil ? Solubility Factor: Solute and Solvent characteristic: • In aqueous solution, water will form strong intermolecular forces with only other polar molecules (the dissolution process) • Oil is a nonpolar substance and therefore will only form strong IMF with other nonpolar substances such as organic compounds. An oil layer floating on water. For a substance to dissolve, the water -water hydrogen bonds must be broken to make a “hole” for each solute particle. However, the water-water interactions will break only if they are replaced by similar strong interactions with the solute. • The result is the immisciblity of water and oil. (Later the Energetics of this process will be discuss ) Organic Chemist saying “Like Dissolves Like” 4 Solubility December 20

Dissolution of Solid Solute What is the driving force which causes solutes to dissolve to form solutions? Covalent versus Ionic solute 1. Covalent solutes dissolve by H-bonding to water or by 2. Ionic solutes dissolve by dissociation into their ions. LDF Ionic Covalent Picture of Ethanol and Na. Cl dissolving 5 Solubility December 20
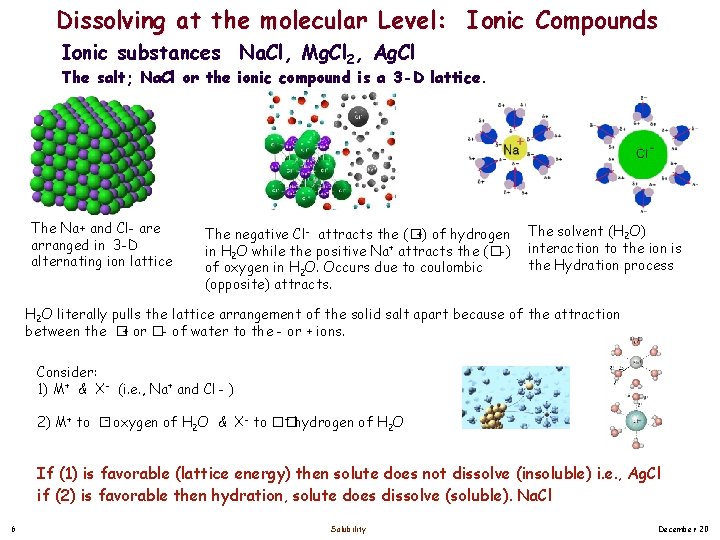
Dissolving at the molecular Level: Ionic Compounds Ionic substances Na. Cl, Mg. Cl 2, Ag. Cl The salt; Na. Cl or the ionic compound is a 3 -D lattice. The Na+ and Cl- are arranged in 3 -D alternating ion lattice The negative Cl- attracts the (�+) of hydrogen in H 2 O while the positive Na+ attracts the (�-) of oxygen in H 2 O. Occurs due to coulombic (opposite) attracts. The solvent (H 2 O) interaction to the ion is the Hydration process H 2 O literally pulls the lattice arrangement of the solid salt apart because of the attraction between the � + or �- of water to the - or + ions. Consider: 1) M+ & X- (i. e. , Na+ and Cl - ) + hydrogen of H O 2) M+ to �- oxygen of H 2 O & X- to �� 2 If (1) is favorable (lattice energy) then solute does not dissolve (insoluble) i. e. , Ag. Cl if (2) is favorable then hydration, solute does dissolve (soluble). Na. Cl 6 Solubility December 20
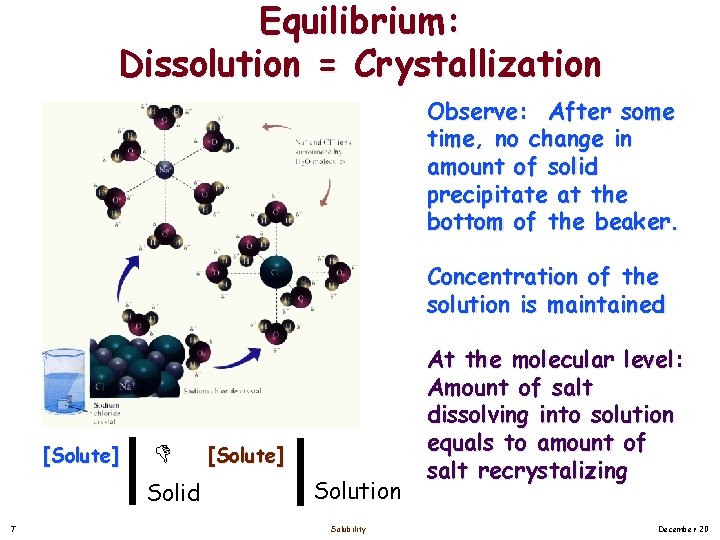
Equilibrium: Dissolution = Crystallization Observe: After some time, no change in amount of solid precipitate at the bottom of the beaker. Concentration of the solution is maintained [Solute] Solid 7 [Solute] Solution Solubility At the molecular level: Amount of salt dissolving into solution equals to amount of salt recrystalizing December 20

Dynamic Equilibrium Situation in which changes occur at equal rates so there is no apparent net change. Le. Chatelier Principle A change (stress) on a system at equilibrium will cause the system to self adjust itself to reduce the stress until a new equilibrium is re-established. Example: Traffic at a toll bridge Vapor Pressure Sugar dissolving 8 Solubility December 20

Ionic Vs Covalent Compounds: Electrolyte Vs. Nonelectrolyte Substance when dissolve can break-up to ions or stay intact. i. e. , Na. Cl and sugar. Type: % ionization: Solubility (in H 2 O) Electrolyte: conducts electricity. Strong electrolyte 100 % ionization Weak electrolyte less 100% ionization very soluble slightly to very soluble Nonelectrolyte: Do not conduct electricity no conduction 9 0 % ionization insoluble or soluble Solubility December 20
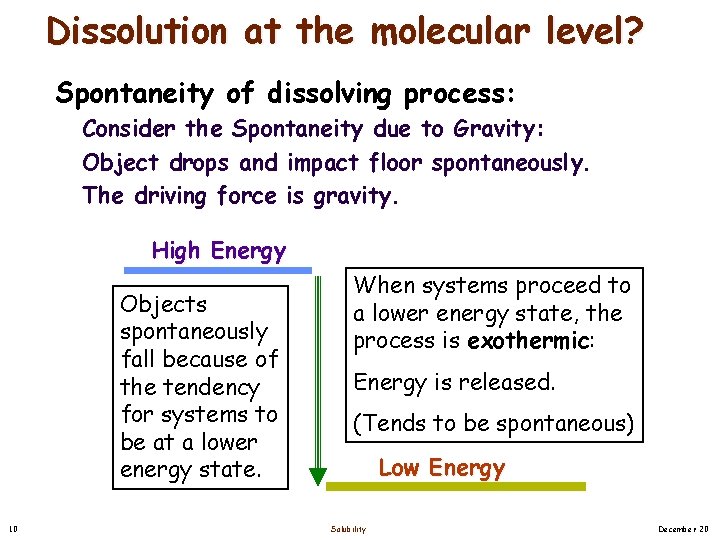
Dissolution at the molecular level? Spontaneity of dissolving process: Consider the Spontaneity due to Gravity: Object drops and impact floor spontaneously. The driving force is gravity. High Energy Objects spontaneously fall because of the tendency for systems to be at a lower energy state. 10 When systems proceed to a lower energy state, the process is exothermic: Energy is released. (Tends to be spontaneous) Low Energy Solubility December 20

Driving Force for Dissolution Reaction exothermic �H (-) Downhill �� Spontaneous Reaction exothermic �H (+) Uphill �Spontaneous ? ? ? 11 Solubility December 20
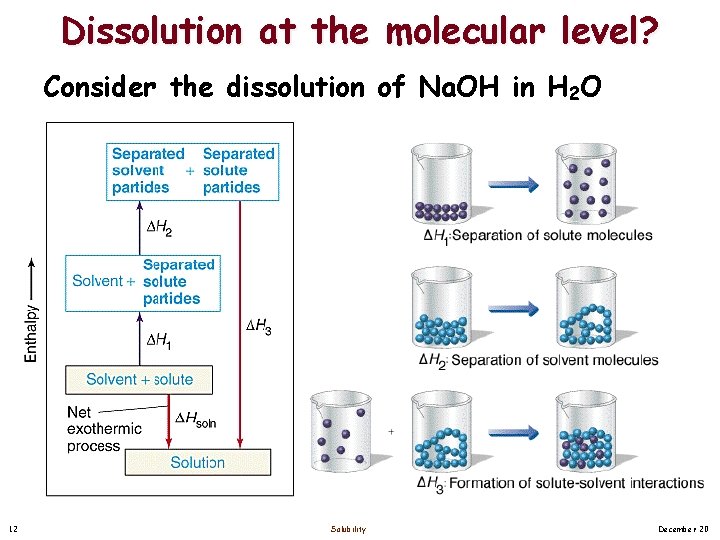
Dissolution at the molecular level? Consider the dissolution of Na. OH in H 2 O 12 Solubility December 20
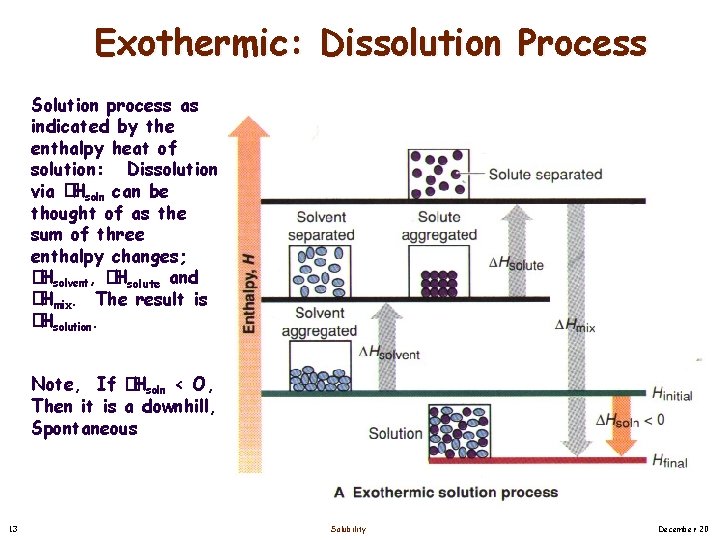
Exothermic: Dissolution Process Solution process as indicated by the enthalpy heat of solution: Dissolution via �Hsoln can be thought of as the sum of three enthalpy changes; � Hsolvent, �Hsolute and � Hmix. The result is � Hsolution. Note, If �Hsoln < O, Then it is a downhill, Spontaneous 13 Solubility December 20
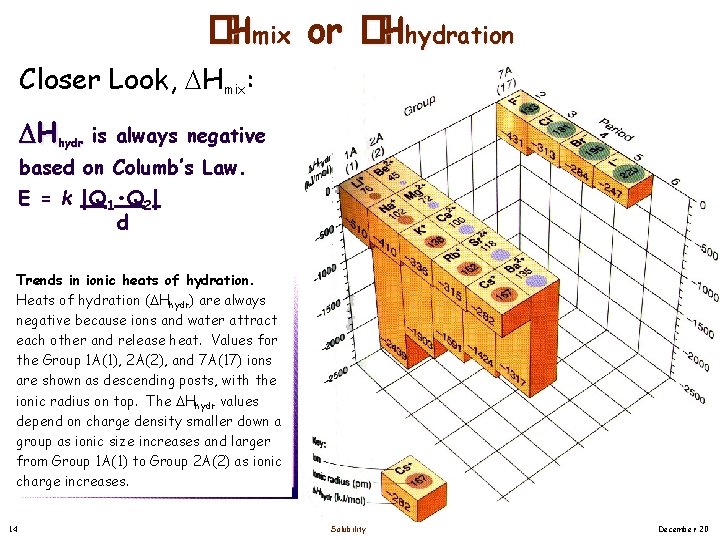
�Hmix or �Hhydration Closer Look, Hmix: Hhydr is always negative based on Columb’s Law. E = k |Q 1 • Q 2| d Trends in ionic heats of hydration. Heats of hydration ( Hhydr) are always negative because ions and water attract each other and release heat. Values for the Group 1 A(1), 2 A(2), and 7 A(17) ions are shown as descending posts, with the ionic radius on top. The Hhydr values depend on charge density smaller down a group as ionic size increases and larger from Group 1 A(1) to Group 2 A(2) as ionic charge increases. 14 Solubility December 20
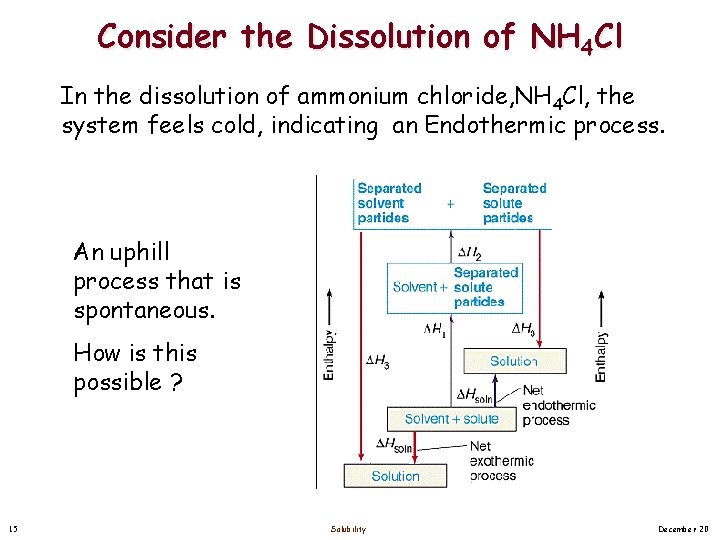
Consider the Dissolution of NH 4 Cl In the dissolution of ammonium chloride, NH 4 Cl, the system feels cold, indicating an Endothermic process. An uphill process that is spontaneous. How is this possible ? 15 Solubility December 20
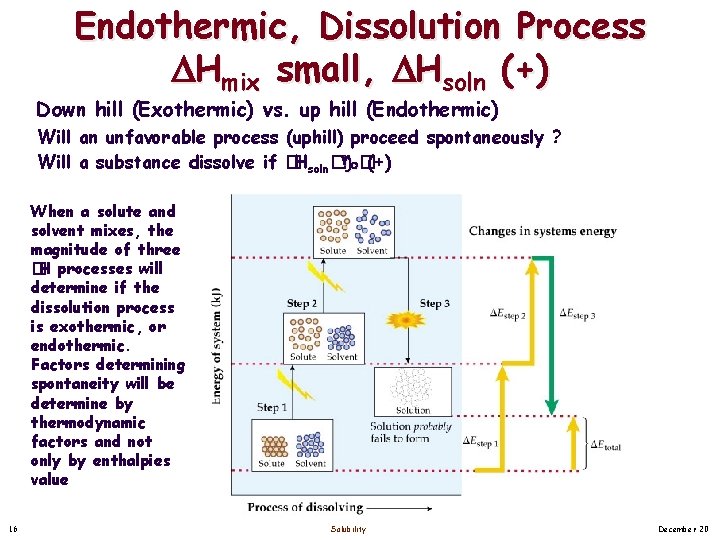
Endothermic, Dissolution Process Hmix small, Hsoln (+) Down hill (Exothermic) vs. up hill (Endothermic) Will an unfavorable process (uphill) proceed spontaneously ? Will a substance dissolve if � Hsoln� �(+) When a solute and solvent mixes, the magnitude of three �H processes will determine if the dissolution process is exothermic, or endothermic. Factors determining spontaneity will be determine by thermodynamic factors and not only by enthalpies value 16 Solubility December 20

Second Law of Thermodynamics Why does ink spread or gas expand or your room gets chaotic? Greater probability of disorder than order. One of the Basic Law of Nature 2 nd Law of Thermodynamics: Process in which disorder increases occur spontaneously 17 Solubility December 20
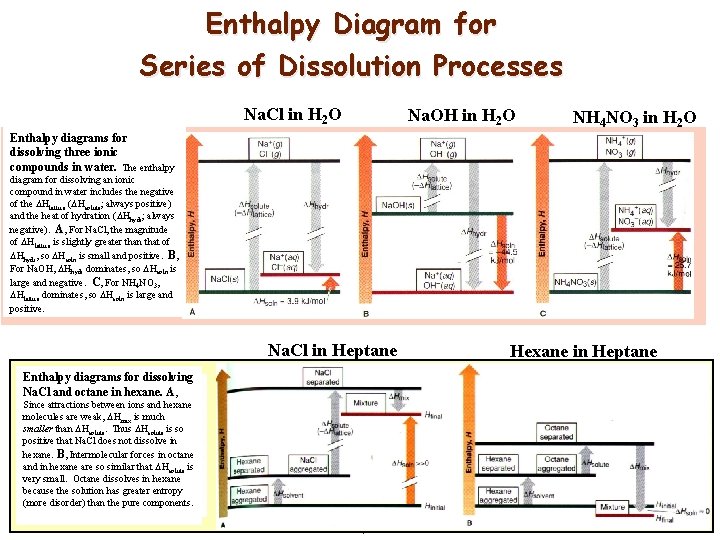
Enthalpy Diagram for Series of Dissolution Processes Na. Cl in H 2 O Na. OH in H 2 O NH 4 NO 3 in H 2 O Enthalpy diagrams for dissolving three ionic compounds in water. The enthalpy diagram for dissolving an ionic compound in water includes the negative of the Hlattice ( Hsolute; always positive) and the heat of hydration ( Hhydr; always negative). A, For Na. Cl, the magnitude of Hlattice is slightly greater than that of Hhydr, so Hsoln is small and positive. B, For Na. OH, Hhydr dominates, so Hsoln is large and negative. C, For NH 4 NO 3, Hlattice dominates, so Hsoln is large and positive. Na. Cl in Heptane Hexane in Heptane Enthalpy diagrams for dissolving Na. Cl and octane in hexane. A, Since attractions between ions and hexane molecules are weak, Hmix is much smaller than Hsolute. Thus Hsolute is so positive that Na. Cl does not dissolve in hexane. B, Intermolecular forces in octane and in hexane are so similar that Hsolute is very small. Octane dissolves in hexane because the solution has greater entropy (more disorder) than the pure components. 18 Solubility December 20

Factors Affecting Solubility 1. Nature of Solute / Solvent. - Like dissolves like (IMF) 2. Temperate Factor i) Solids/Liquids- Solubility increases with Temperature solute / solvent. Increase K. E. increases motion and collision between ii) gas - Solubility decreases with Temperature Increase K. E. result in gas escaping to atmosphere. 3. Pressure Factor i) Solids/Liquids - Very little effect Solids and Liquids are already lose together, extra pressure will not increase solubility. ii) gas - Solubility increases with Pressure. Increase pressure squeezes gas solute into solvent. 19 Solubility December 20

2 i) Temperature on Solubility: Solids & Liquids Temperature - (Solid and Liquid) Consider the extent in which sugar or Na. Cl dissolves in water. What are the conditions which will increase the solubility of sugar or salt in water. [Solute] Solid Solution As the temperature increase, both solute and solvent will be moving faster, this will result in the mixing of both substance to be more effective. 20 Solubility December 20
![Equilibrium Revisited [Solute] Solid [Solute] Solution Observe: After some time, no change in amount Equilibrium Revisited [Solute] Solid [Solute] Solution Observe: After some time, no change in amount](http://slidetodoc.com/presentation_image_h/e53ceb35ff54cc79285dcad57fb930d4/image-21.jpg)
Equilibrium Revisited [Solute] Solid [Solute] Solution Observe: After some time, no change in amount of solid precipitate at the bottom of the beaker. Concentration of the solution is maintained Equilibrium Situation in which changes occur at equal rates so there is no apparent net change. Le. Chatelier Principle A Change (i. e. , stress) on a system at equilibrium will cause the system to self adjust itself to reduce the stress until a new equilibrium is re-establish. 21 Solubility December 20
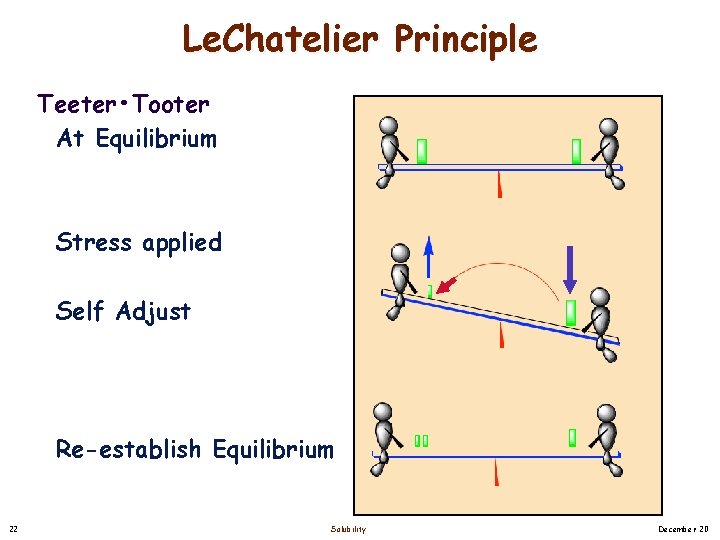
Le. Chatelier Principle Teeter • Tooter At Equilibrium Stress applied Self Adjust Re-establish Equilibrium 22 Solubility December 20
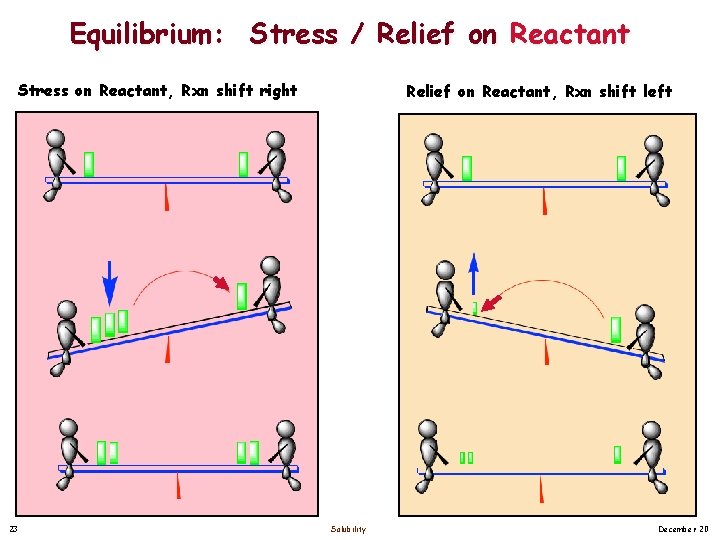
Equilibrium: Stress / Relief on Reactant Stress on Reactant, Rxn shift right 23 Relief on Reactant, Rxn shift left Solubility December 20
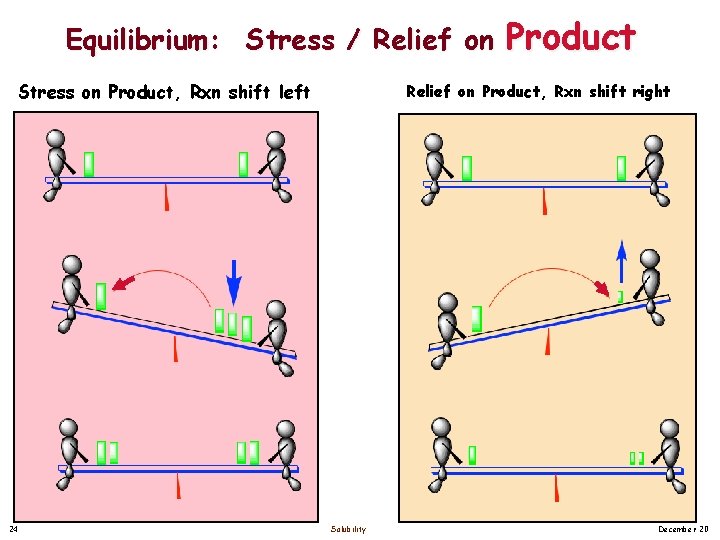
Equilibrium: Stress / Relief on Stress on Product, Rxn shift left 24 Product Relief on Product, Rxn shift right Solubility December 20
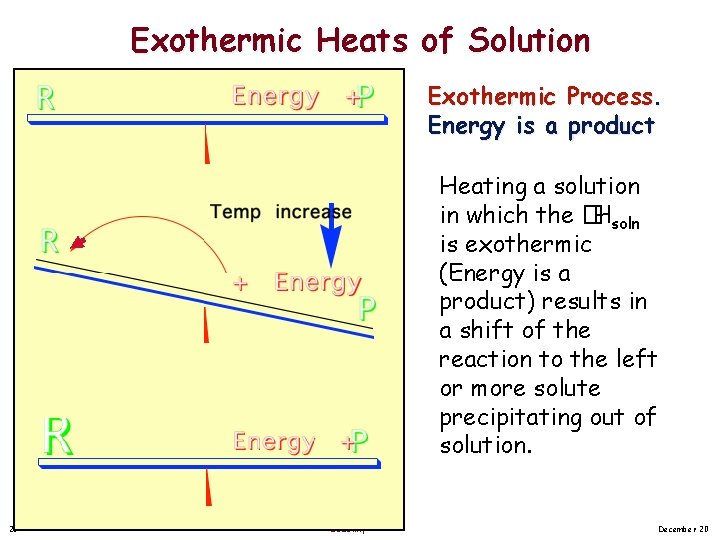
Exothermic Heats of Solution Exothermic Process. Energy is a product Heating a solution in which the �Hsoln is exothermic (Energy is a product) results in a shift of the reaction to the left or more solute precipitating out of solution. 25 Solubility December 20
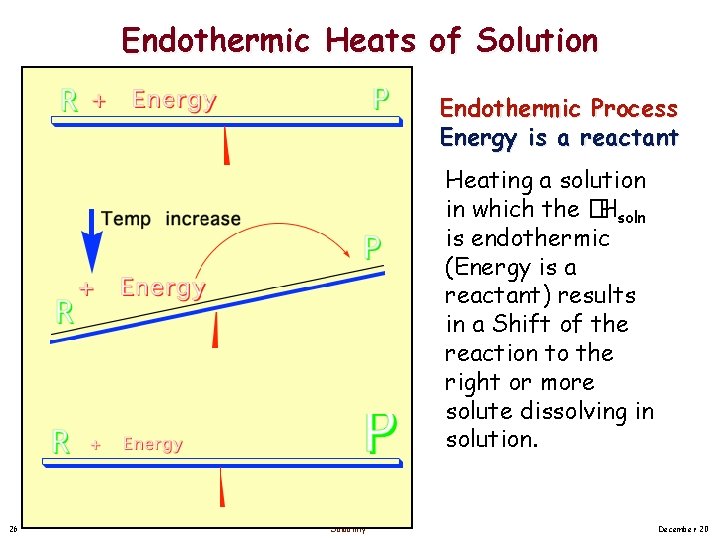
Endothermic Heats of Solution Endothermic Process Energy is a reactant Heating a solution in which the �Hsoln is endothermic (Energy is a reactant) results in a Shift of the reaction to the right or more solute dissolving in solution. 26 Solubility December 20
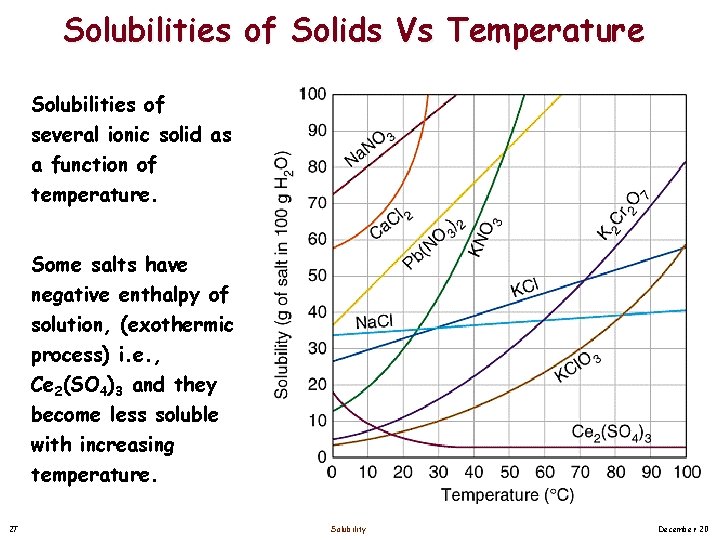
Solubilities of Solids Vs Temperature Solubilities of several ionic solid as a function of temperature. Some salts have negative enthalpy of solution, (exothermic process) i. e. , Ce 2(SO 4)3 and they become less soluble with increasing temperature. 27 Solubility December 20

2 ii) Temperature & Solubility: Gases Temperature - (Gas) Consider the extent in which O 2 or CO 2 dissolves in water. What are the conditions which will increase the solubility of gas in water. [Solute] gas As the temperature increase, both solute and solvent will be moving faster, the gas solute however will now have enough energy to leave the liquid interface. 28 Solubility [Solute] Solution December 20
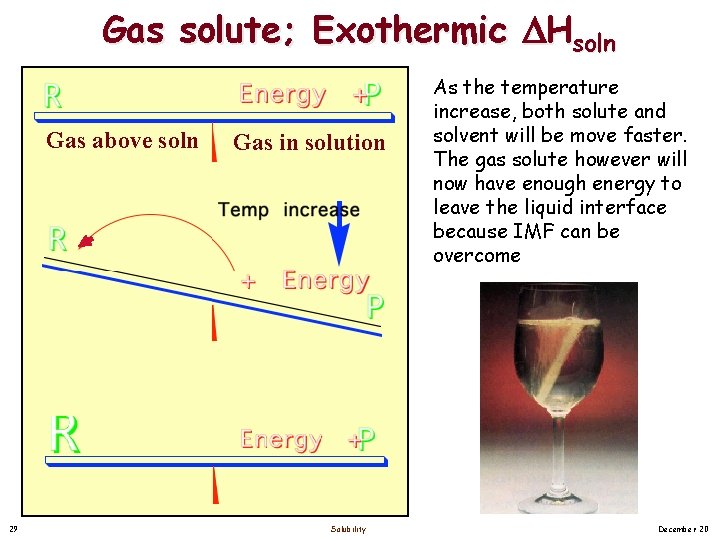
Gas solute; Exothermic Hsoln Gas above soln 29 Gas in solution Solubility As the temperature increase, both solute and solvent will be move faster. The gas solute however will now have enough energy to leave the liquid interface because IMF can be overcome December 20

Disaster: (1700 dead) from Gas Solubility In the African nation of Cameroon in 1986 a huge bubble of CO 2 gas escaped from Lake Nyos and moved down a river valley at 20 m/s (about 45 mph). Because CO 2 is denser than air, it hugged the ground and displaced the air in its path. More than 1700 people suffocated. The CO 2 came from springs of carbonated groundwater at the bottom of the lake. Because the lake is so deep, the CO 2 mixed little with the upper layers of water, and the bottom layer became supersaturated with CO 2. Lake Nyos in Cameroon, the site of a natural disaster. In 1986 a huge bubble of CO 2 escaped from the lake and asphyxiated more than 1700 people. 30 Solubility When this delicate situation was changed, perhaps because of an earthquake or landslide, the CO 2 came out of the lake water just like it does when a can of soda is opened. December 20

3 i) Pressure on Solubility: Solids / Liquid Pressure - (Solid and Liquid) The solubility of solids and liquids are hardly affected by pressure. Solids and liquids are already very close to each other. An increase in pressure will not affect solubility 31 Solubility December 20

3 ii) Pressure on Solubility: Gas Pressure - (Gas) Solubility of gas is greatly affected by pressure Gas solute is very sensitive to pressure. Gas particles are separated by large void space, an increase in pressure will increase these particles to come closer togethereby increasing the solubility of the gas. Divers must be careful when diving to great depth because the potential of dissolved N 2 gas in blood. Clinical term is the Bends. 32 Solubility December 20
![Pressure Affect: Teeter Totter Analogy [Solute] Pressure Sensitive gas Solution In utilizing Le. Chatelier Pressure Affect: Teeter Totter Analogy [Solute] Pressure Sensitive gas Solution In utilizing Le. Chatelier](http://slidetodoc.com/presentation_image_h/e53ceb35ff54cc79285dcad57fb930d4/image-33.jpg)
Pressure Affect: Teeter Totter Analogy [Solute] Pressure Sensitive gas Solution In utilizing Le. Chatelier Principle to determine the direction of solubility for a gaseous solute with variation in pressure, the first thing that must be establish is which side is more sensitive to pressure. In our case the gas is more sensitive than the solution. 33 Solubility December 20

Henry’s Law At pressure of few atmosphere or less, solubility of gas solute follows Henry Law which states that the amount of solute gas dissolved in solution is directly proportional to the amount of pressure above the solution. c = k • P where, c = solubility of the gas (usually molality) k = Henry’s Law Constant, (solute-solvent pair) P = partial pressure of gas over solution Henry’s Law Constant (25°C) 34 Gas k N 2 O 2 CO 2 8. 42 • 10 -7 1. 66 • 10 -6 4. 48 • 10 -5 M/mm. Hg Solubility December 20

Henry’s Law: Example The solubility of ethylene (C 2 H 4) in water at 20°C and a pressure of 0. 300 atm is 1. 27 • 10 -4 m. a) calculate the Henry’s Law constant, k in (m/torr) b) How many grams of ethylene are dissolved in one kilogram of water at 20°C at 500 torr. ? Detm’ k from c = k • P, k= c / P k= 1. 27 • 10 -4 m a) = 1. 27 • 10 -4 m 0. 300 atm (760 torr/1 atm) k = 5. 57 • 10 -7 m / torr 228 torr c = k • P k is constant which depends only on T c = 5. 57 • 10 -7 m / torr • 500 torr = 2. 79 • 10 -4 m /kg H 2 O b) 35 mass = 2. 79 • 10 -4 mol • 28. 0 g / mol mass = 7. 80 • 10 -3 g Solubility December 20
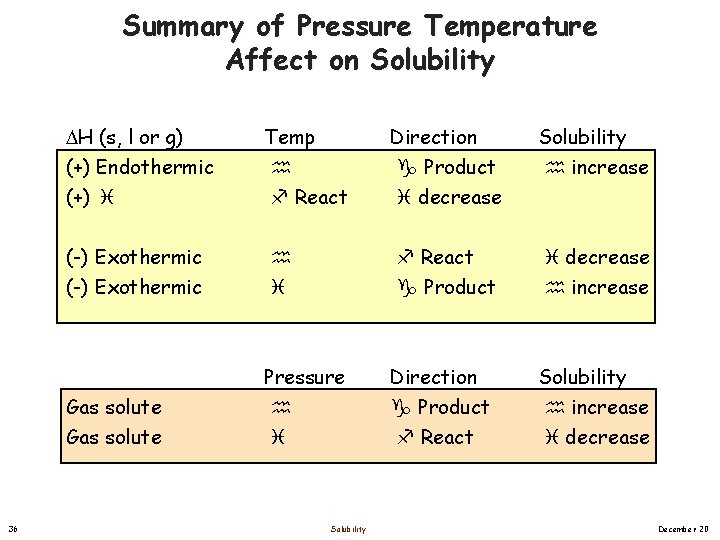
Summary of Pressure Temperature Affect on Solubility 36 H (s, l or g) (+) Endothermic (+) Temp React Direction Product decrease Solubility increase (-) Exothermic React Product decrease increase Gas solute Pressure Direction Product React Solubility increase decrease Solubility December 20
- Slides: 36