13 2 NOTES Solutions Basics Endo Exo Solvation

13. 2 – NOTES Solutions Basics

Endo Exo Solvation Demo

• Solution = Homogeneous Mixture • Solute: the substance being dissolved • Solvent: the substance that the solute is dissolving into (almost always water!)

• Different Kinds of Solutions • Solid dissolved in a Liquid: Soluble • Liquid dissolved in a Liquid: Miscible • Gas dissolved in a Liquid: ex. Soda, dissolved O 2 in water sources • Often these solutions effervesce when opened or exposed, gas particles escape quickly. • Liquid dissolved in a Gas: perfumes, aerosols • Gas dissolved in a Gas: air, baking smells • Mixing a gas with another gas ALWAYS produces a homogenous mixture because all gases expand to fill their container, and so will be evenly distributed when mixed. • Solid dissolved in a Solid*: alloys, but must be melted to combine, then resolidifed

• Solvation (Dissolution) in Water • Water is known as the Universal Solvent. Why? It’s polar, lots of solutes are polar • Dissolve vs. Dissociate • To dissolve means to become surrounded by solvent particles. • To dissociate means to break into pieces (ions) when dissolved.

• Ionic Compounds in Water • All ionic compounds dissociate into ions when dissolved, then become surrounded by water molecules. • When ionic compounds dissolve, bonds are broken.

• Molecular Compounds in Water • Almost all molecular compounds do not dissociate when they dissolve, but they become surrounded by water molecules. • When molecular compounds dissolve, intermolecular forces are broken.

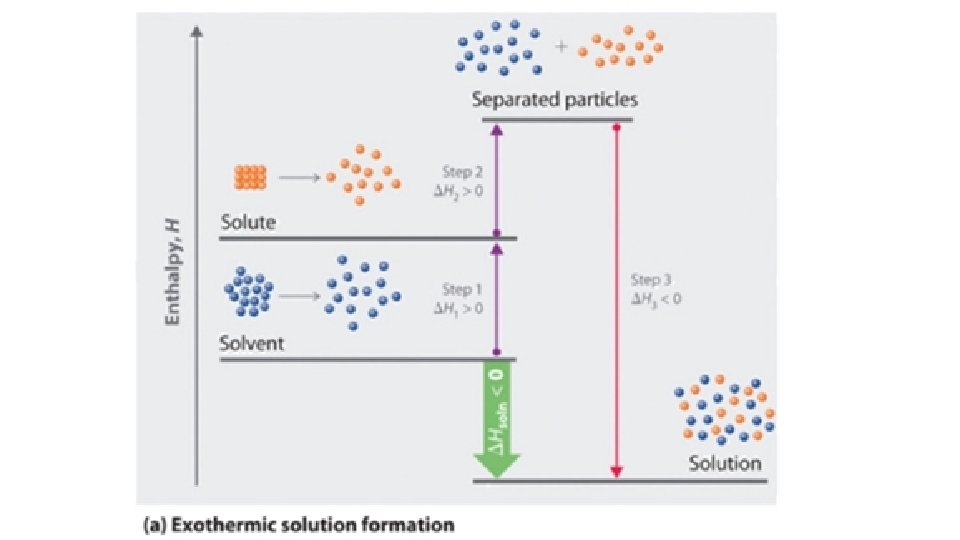
• Heat of Solution: Energy required or released when a substance dissolves • Solvation occurs in 3 Steps: • Solute particles must separate from each other (IMFs or bonds must be broken) – endothermic; ΔH is positive • Solvent particles must separate from each other to make room for the solute particles to move between them (IMFs or bonds must be broken) – endothermic; ΔH is positive • Solute and Solvent particles must attract each other (IMFs or bonds must form!) – exothermic; ΔH is negative • The sum of the three steps (measured in Joules) gives the overall
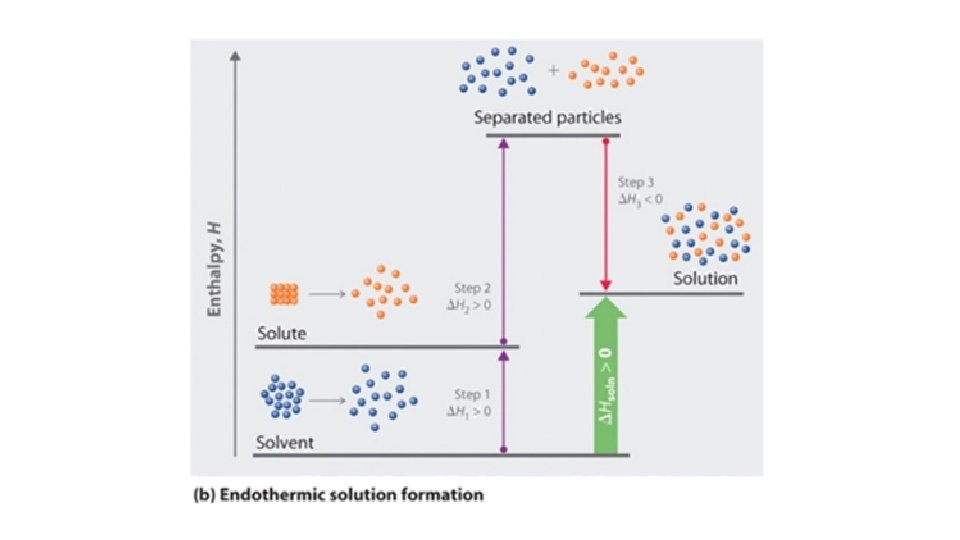

• The sum of the three steps (measured in Joules) gives the overall Heat of Solution • If the sum is positive: endothermic • When the solute dissolves, the temperature of the mixture will go down (feels cold !) • These solutes are more soluble at high temps. • If the sum is negative: exothermic • When the solute dissolves, the temperature of the mixture will go up (feels hot !) • These solutes are more soluble at low temps.

• Factors that Influence Solubility • Solubility: Maximum amount of solute that can dissolve in a certain volume of solvent (at a certain Temperature and Pressure) • How much solute can be held? At some point, the solvent becomes “full” and no more solute can possibly dissolve.

• Unsaturated Solution: contains less than the maximum amount of possible solute; more can be dissolved! • Saturated Solution: contains the maximum amount of possible solute; the solution is full!; no more can be dissolved! • Supersaturated Solution: contains more solute than expected at a certain temperature; made from a careful process. • Make a saturated solution. • Heat (or cool) until the extra solute dissolves. • Allow the solution to come to room temperature slowly. • The additional solute will remain dissolved until the solution is disturbed. • If disturbed, all extra solute will precipitate quickly!

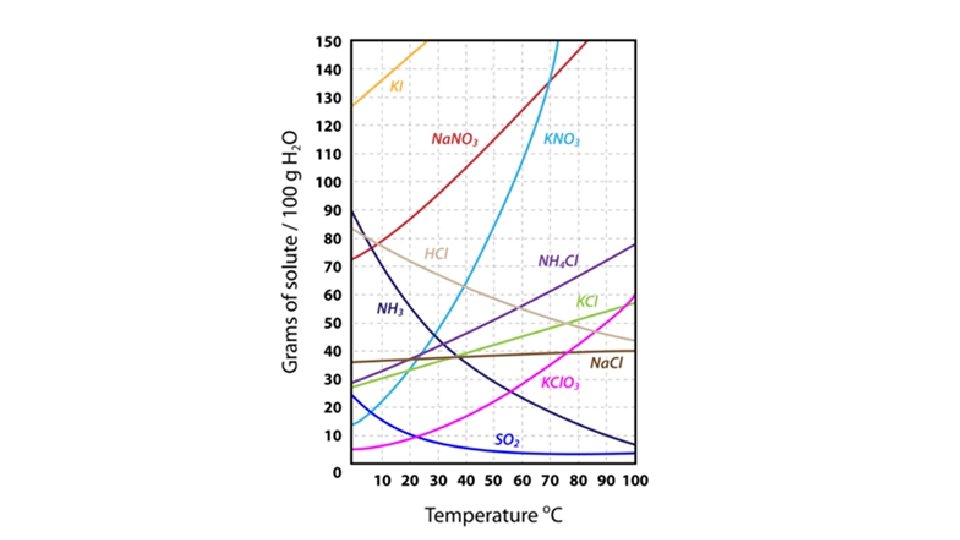
• Effect of Temperature • Solubility Curves: graphs that show the relationship between temperature and solubility • Most solutes are more soluble at high temperatures. • Depends on the Heat of Solution for the substance. • Endothermic Heat + Solute → Solution • Exothermic Solute → Solution + Heat • All gases are more soluble at low temperatures.

• Effect of Pressure • Liquid and Solid solutes’ solubilities are not effected by changes in pressure. Why? Solids and liquids do not compress • Gases are more soluble at high pressures.

• Effect of Surface Area • The higher the surface area (the larger the particle size), the faster a solute will dissolve. • Smaller solute particles have greater contact with the solvent • Sugar cubes are designed to dissolve slowly, so that a hint of sweetness is present. Granulated sugar should dissolve faster, and powdered sugar even faster! Rank the four seltzer tablets seen to the left in terms of rate of solution:

• Agitation -(Stirring) can also speed up the dissolving process • Increases solute/solvent contact

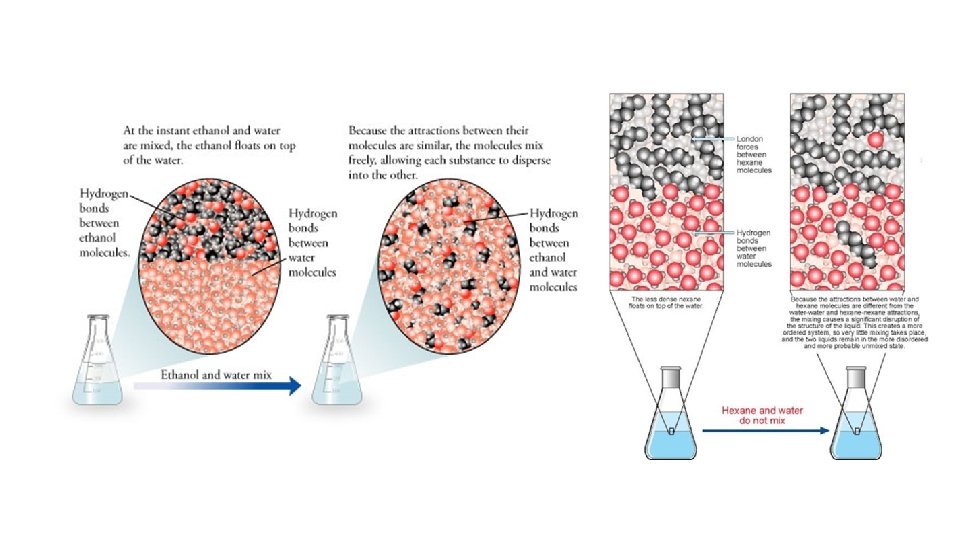
• Effect of Polarity • Polar solutes are soluble in polar solvents (H 2 O) • Nonpolar solutes are soluble in nonpolar solvents (CCl 4, benzene) • If polarities do not match, a suspension is formed: a heterogeneous mixture that separates!

- Slides: 21