1127 Do NOW Complete the following table Element
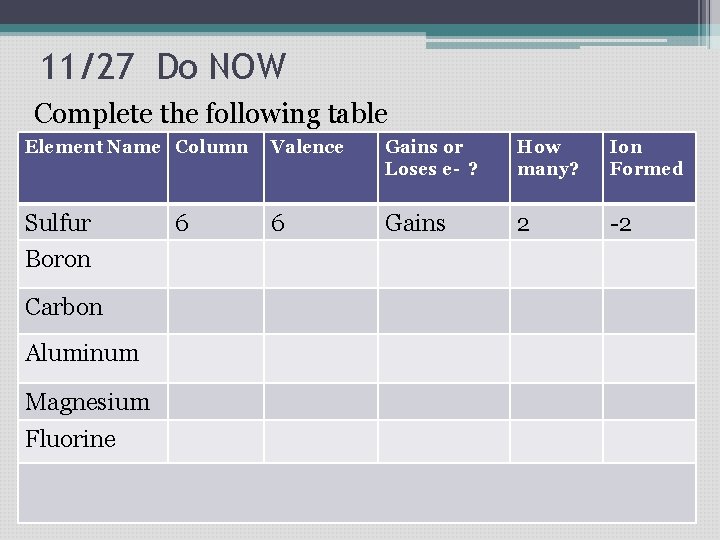
11/27 Do NOW Complete the following table Element Name Column Valence Gains or Loses e- ? How many? Ion Formed Sulfur 6 Gains 2 -2 Boron Carbon Aluminum Magnesium Fluorine 6

11/27 Agenda 1. Do Now 2. Go over Ch 5 Test 3. New Learning: Ionic Bonds HW: 1 -5, 8 -9 (Pg: 6&7 are extra practice) Quiz #1 Tomorrow (Vocab about ions and Dot Diagrams) Quiz #2 Friday Quiz #3 Monday

Chapter 6 Chemical Bonds

6. 1 Ionic Bonding • Stable Electron Configurations ▫ When the highest occupied energy level of an atom is filled with electrons, the atom is stable and will NOT react. �Ex: Noble Gases (8 valence electrons) and Helium (2 v. e. )
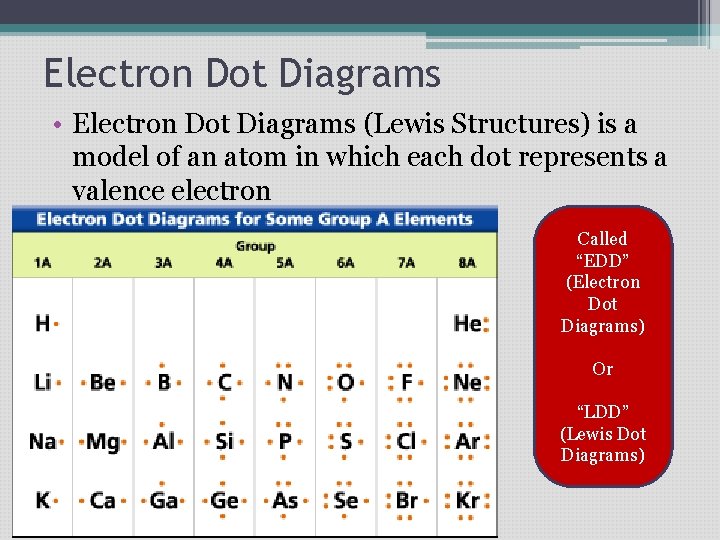
Electron Dot Diagrams • Electron Dot Diagrams (Lewis Structures) is a model of an atom in which each dot represents a valence electron Called “EDD” (Electron Dot Diagrams) Or “LDD” (Lewis Dot Diagrams)

Ionic Bonds • Elements that DO NOT have complete sets of valence electrons will be reactive. • By reacting, they can achieve complete sets. • Some elements achieve stable electron configurations through the transfer of electrons between atoms. PS: What’s Wrong with the Sodium LDD? We can Rearrange them!!

Formation of Ions • When an atom gains or loses an electron, the protons do not equal the electrons. The atom is no longer neutral. • Ion- Atom that has a net positive or negative charge. ▫ Anion- Ion with a negative charge. �To name an anion, you change the suffix to –ide and add ion. �Ex: Chloride ion ▫ Cation- Ion with a positive charge. �To name a cation, you add ion to the end. �Ex: Sodium ion

11/28 Do NOW 1. Write the LDD for the following atoms P Be I B 2. Write the LDD for the following ions P Be I 3. Name the 4 ions above B

11/28 Agenda 1. Do Now 2. Continue Ionic Bonds 1. LDD Practice 2. Drawing Ionic bonds practice HW: 1 -5, 8 -9 (Pg: 6&7 are extra practice) Quiz #1 Tomorrow (Vocab about ions and Dot Diagrams) Quiz #2 Friday Quiz #3 Monday

Formation of Ionic Bonds • Chemical Bond- force that holds atoms or ions together. • Ionic Bond- force that holds anions and cations together. ▫ An ionic bond forms when electrons are transferred from one atom to another • Ionization Energy- the amount of energy needed to remove an electron. ▫ The lower the ionization energy, the easier it is to remove an electron. �It’s easier to remove an electron from a metal than from a nonmetal

Pg in HW packet Ionic Compounds • A chemical formula is a notation that shows what elements a compound contains and the ratio of the atoms or ions of these elements in the compound. ▫ Ex: Mg. Cl 2

11/29 Do NOW

11/29 Agenda 1. Do Now 2. Naming Ionic Bonds 3. Practice: pages 3, 4, 5 HW: Pgs 1 -9 Quiz #1 tomorrow ( Ionic Bonding vocab, LDD’s, ionic names, ionic formulas)

If charges don’t balance, you’ll need to add more atoms Ionic Compound Diagrams to try: • K and Cl Li and S • Sr and Cl Al and N • *Ga and S

6. 3 Naming Compounds and Writing Formulas • Naming Ionic Compounds ▫ Binary Ionic Compounds – Made from only 2 elements – 1 metal and 1 nonmetal �Cation first followed by anion. �Change the ending of the anion to -ide Skip Forward in your notes

6. 3 Naming Compounds and Writing Formulas Skip Forward in your notes • Writing Formulas for Ionic Compounds ▫ Place the symbol of the cation first, followed by the symbol for the anion. ▫ Use subscripts to show the ratio of the ions in the compound (all compounds must be neutral) �Use crisscross method!! �Ex: Strontium chloride Magnesium oxide Aluminum nitride Aluminum oxide ▫ **Coefficient – the # in front of the compound �Tells you how many compounds you have.

Subscript and Coefficient Practice • Do Pg 2 • Pg 3 Fe Fe(OH)3 3 Fe(OH)3 13 NH 4 NO 3 • Finish Pgs 3 -8 for HW O H N C

11/30 Do NOW Complete the following info: Ex: Mg and Cl Steps: 1. Draw the LDD of each atom B and Cl 2. Draw arrows showing movement of electrons 3. Write the Diagram 4. Write the Formula 5. Give the name 1 2 Mg. Cl 2 Magnesium Chloride Ions Stage!

11/30 Agenda 1. Do Now and check HW 2. Go over HW 3. Properties of Ionic Bonds 4. “Putting it all together” 5. Quiz HW: 1) Correct your HW mistakes Quiz #2 Monday/Tues (Covalent Bonds: vocab and LDD’s) ** Clinic today – Test make ups ONLY!!

Ionic Bonds make Crystal Lattices • Remember that orderly, 3 -D structure of solids? ▫ In Na. Cl, each chloride ion is surrounded by 6 sodium ions and each sodium ion is surrounded by 6 chloride ions. ▫ The opposite charges keep ions in their fixed positions in a rigid framework, or lattice. • Crystals- Solids whose particles are arranged in a lattice structure.

Properties of Ionic Compounds • The properties of an ionic compound can be explained by the strong attractions among ions. 1. High melting point 2. Poor conductor of electricity when solid, good conductor when melted. 3. Shatter when struck by a hammer
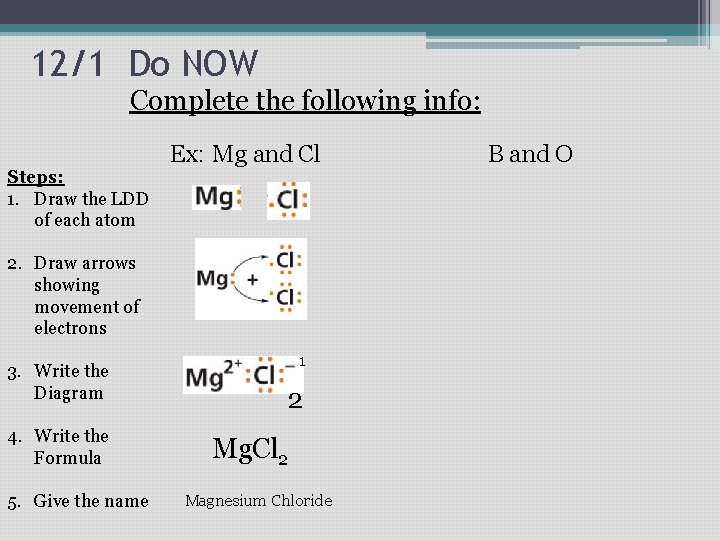
12/1 Do NOW Complete the following info: Ex: Mg and Cl Steps: 1. Draw the LDD of each atom 2. Draw arrows showing movement of electrons 3. Write the Diagram 4. Write the Formula 5. Give the name 1 2 Mg. Cl 2 Magnesium Chloride B and O

12/1 Agenda 1. Do Now 2. Go over Quiz 3. Quiz #2 – practice and grade 4. Start covalent bonds HW: None Quiz #2 Monday/ (Basic Covalent Bonds stuff: vocab and LDD’s)

Putting it ALL together Complete the following info: Ex: Mg and Cl Steps: 1. Draw the LDD of each 2. Draw arrows showing movement of electrons 3. Write the Diagram 4. Write the Formula 5. Give the name 1 2 Mg. Cl 2 Magnesium Chloride Rb and Se Al and Br

We’re now LEAVING Ionic Bonds (Giving/Taking Electrons) ENTERING Covalent Bonds (Sharing Electrons)

Ionic or Covalent? ? Ionic Na. Cl Place the following compounds Covalent NH 4 Na. Cl Mg. Cl 2 NH 4 O 2 Mg. Cl 2 Al 2 O 3 O 2 CH 4 H 2 O Al 2 O 3 Na. Br H 2 O Br 2 Na. Br CO 2 Br 2 CO 2 So, what criteria makes ionic and covalent compounds different?

12/4 Do Now 1. Write the electron dot diagram for the following atoms OR Ions Sulfide ions Sulfur 2. Give the formula for the following ionic compounds AND name them Magnesium and Bromine Aluminum and phosphorous 3. Label the following compounds “I” for Ionic or “C” for Covalent H 2 O Mg. Cl 2 CO 2 Li 2 O

12/4 Agenda 1. Do Now 2. Move Seats 3. Begin Covalent bonds 4. Check for understanding HW: 10, 12, 13 Quiz #2 Wednesday- (Covalent Bonds: vocab, LDD’s, and formulas) Objective: Identify and label covalent bonds and diatomic molecules
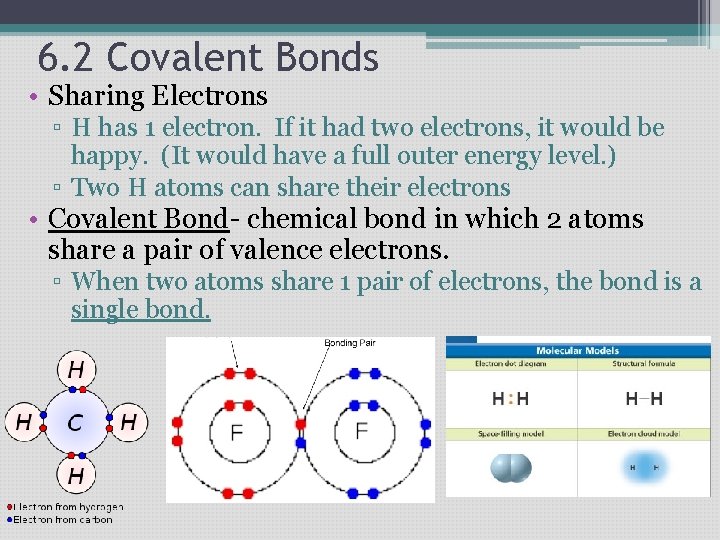
6. 2 Covalent Bonds • Sharing Electrons ▫ H has 1 electron. If it had two electrons, it would be happy. (It would have a full outer energy level. ) ▫ Two H atoms can share their electrons • Covalent Bond- chemical bond in which 2 atoms share a pair of valence electrons. ▫ When two atoms share 1 pair of electrons, the bond is a single bond.

Molecules of Elements • Reminder: • Two hydrogen atoms bonded together form a unit called a molecule • Molecule- atoms joined together by one or more covalent bonds • The attractions between the shared electrons and the protons in each nucleus hold the atoms together in a covalent bond. • This happens when 2 atoms both want electrons

Diatomic Molecules & Multiple Covalent Bonds • Diatomic Molecules- occurs in many nonmetals. It’s two atoms of the same element. ▫ Ex: Halogen Group ▫ HONCl. Br. IF • Multiple Covalent Bonds Other non-metals can form Diatomic molecules too… • Ex: Nitrogen has 5 valence electrons. It needs to share 3 pairs of electrons in order to be happy.

Check for understanding 1. Star the Covalent bonds 2. Circle the Diatomic molecules 1. Mg. Cl 2 6. Rb 3 P 2 11. Cl 2 2. CH 4 7. N 2 12. H 2 SO 4 3. Li 2 S 8. Br 2 13. HNO 3 4. H 2 O 5. O 2 9. Sr. O 10. Na. Cl 14. P 2 15. Na. OH


12/5 Do Now For the compounds below: 1. Circle the Covalent Bonds 2. Label each Covalent compound “D” for Diatomic or “M” for Multiple Covalent Bonds 1. Be. I 2 4. Ca 3 S 2 7. I 2 2. NH 4 5. P 2 8. Na 2 O 3. H 2 6. HPO 3

12/5 Agenda 1. 2. 3. 4. Do Now and check HW Review HW 2 Types of Covalent Bonds Polar vs Non Polar Exit Ticket - HW: pg 14 – skip #25 and #26 - Quiz #2 Tomorrow - (Covalent Bonds: Vocab, Polar v Nonpolar) - Lab Thursday! Ionic Bonds Dress accordingly Objective: Identify and label polar covalent vs non polar covalent bonds by both picture and formula
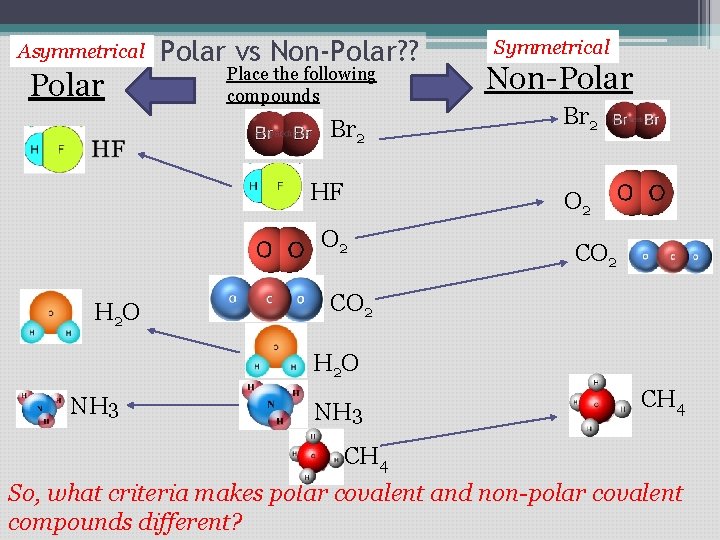
Asymmetrical Polar vs Non-Polar? ? Place the following compounds Br 2 HF Non-Polar Br 2 O 2 H 2 O Symmetrical CO 2 H 2 O NH 3 CH 4 So, what criteria makes polar covalent and non-polar covalent compounds different?

Unequal Sharing of Electrons What causes this Asymmetry? • Polar Covalent Bonds Asymmetrical ▫ Electrons are NOT shared equally. Ex: H-Cl ▫ **Supplemental: Electrons will hang out by Cl, giving it a slight negative charge. The other atom will have a slight positive charge. • Note: In general, elements on the right of the periodic table have a greater attraction for electrons. ▫ Ex: Fluorine has the strongest attraction. Shared electrons in a hydrogen chloride molecule spend less time near the hydrogen atom than near the chlorine atom.
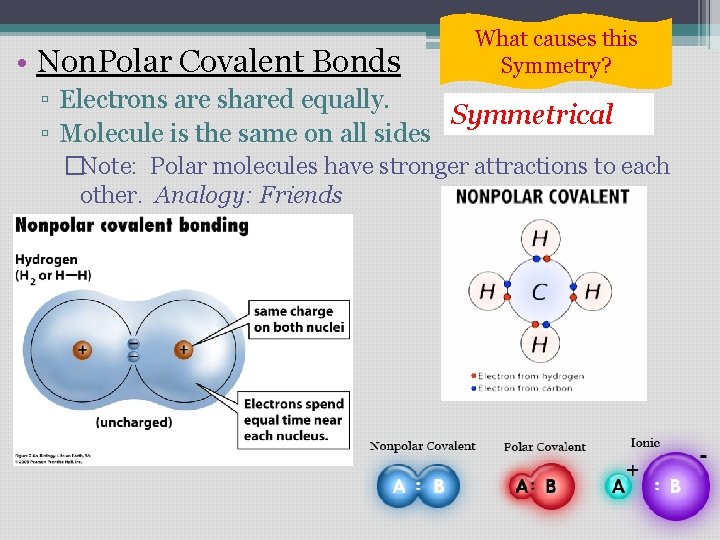
• Non. Polar Covalent Bonds What causes this Symmetry? ▫ Electrons are shared equally. Symmetrical ▫ Molecule is the same on all sides �Note: Polar molecules have stronger attractions to each other. Analogy: Friends

Exit Ticket 1) Label the following as: non-polar covalent (NPC) , polar covalent (PC), or ionic (I). - Now try most of these without pictures 2) On the back of your Exit Ticket, rank yourself 1 -5… (5 being “I can easily do that”) a) I can tell ionic bonds from covalent bonds b) I can identify diatomic molecules c) I can identify Polar vs Non polar compounds


12/6 Do NOW 1. Define “Diatomic Molecule” 2. Give 3 examples of diatomic molecules 3. Explain the difference between ionic and covalent compounds

12/6 Agenda 1. 2. 3. 4. Do Now Go over some HW Review Diatomic and Ionic v Covalent Continue Covalent Bonds - Naming Covalent compounds: Pg 24 - Drawing Covalent Compounds: 19 -23 HW: 1) Pg 24, 19, 20 Quiz #3 Moved to Tomorrow! (Covalent Bonds: vocab and LDD’s)


12/7 Do NOW Draw the EDD Form of the following compounds PCl 3 Challenge: H 2 SO 4 Draw the Structural Formula following compounds HNO Challenge: HSi. Cl 3

12/7 Agenda 1. Do Now 2. Polar Vs Nonpolar 3. Continue Covalent Bonds - Naming Covalent compounds: Pg 24 - Drawing Covalent Compounds: 19 -23 HW: 1) Pg 24, 19, 20

6. 3 Naming Compounds and Writing Formulas Naming Molecular Compounds 1. The more metallic element appears first. 2. The name of the second element changes its ending to –ide. Ex: carbon dioxide 3. **Use the Greek prefixes to reflect the number of atoms of each element. Ex: dinitrogen tetraoxide N 2 O 4 4. If mono appears in the first element, the prefix is dropped. Ex: NO 2 mononitrogen dioxide becomes nitrogen dioxide.

6. 3 Naming Compounds and Writing Formulas Writing Molecular Formulas 1. Write the symbols for the elements in the order the elements appear in the name 2. Prefixes indicate the number of atoms of each element – they become subscripts �If there is no prefix, there is only 1 atom �Ex: Diphosphorus tetrafluoride P 2 F 4

Polar or Non-Polar
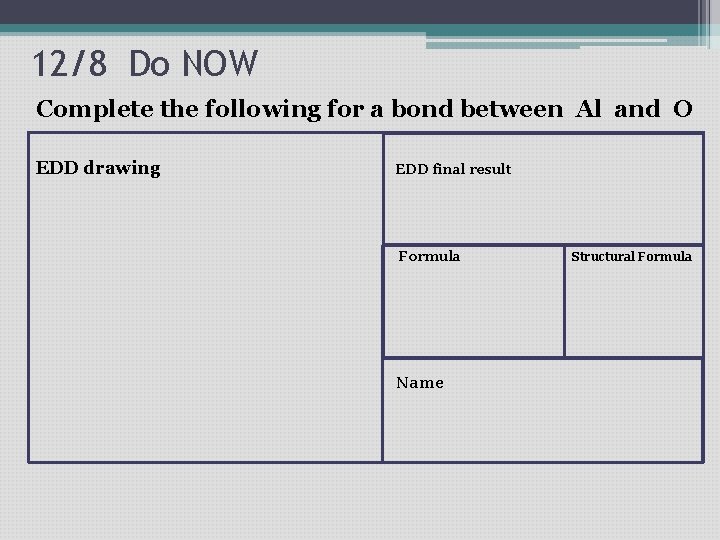
12/8 Do NOW Complete the following for a bond between Al and O EDD drawing EDD final result Formula Name Structural Formula

12/8 Agenda 1. Do Now and check HW 2. Review the HW 3. Quiz 4. “Reminders” 1. Organization Chart 2. The “Central Atom” 5. Begin HW HW: Pgs 21 -23 EVENS Tests will be next week!

The “Central Atom” in Covalent Bonds • In CCl 4, carbon is the “central atom” • In NF 3, nitrogen is the “central atom” What do you think is meant by the “central atom”? Identify the central atom in the following: a) CO 2 b) PH 3 c) Si. O 2
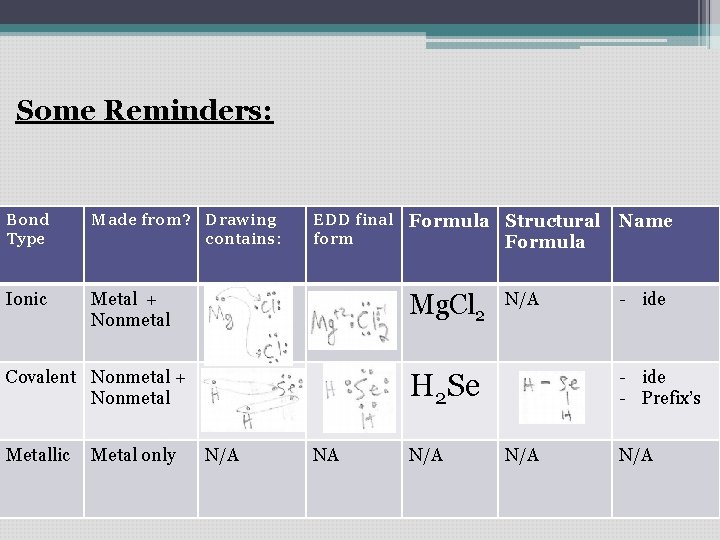
Some Reminders: Bond Type Made from? Drawing contains: Ionic Metal + Nonmetal EDD final Formula Structural form Formula Arrows showing give/take Mg. Cl 2 Covalent Nonmetal + Circles Nonmetal showing sharing e-’s Metallic Metal only N/A N/A - ide - Prefix’s H 2 Se NA Name N/A

The “Central Atom” in Covalent Bonds • In CCl 4, carbon is the “central atom” • In NF 3, nitrogen is the “central atom” What do you think is meant by the “central atom”? Identify the central atom in the following: a) CO 2 b) PH 3 c) Si. O 2
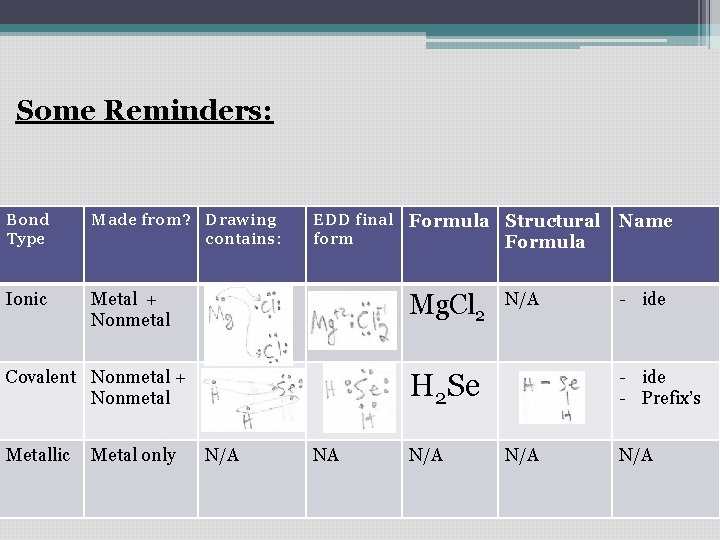
Some Reminders: Bond Type Made from? Drawing contains: Ionic Metal + Nonmetal EDD final Formula Structural form Formula Arrows showing give/take Mg. Cl 2 Covalent Nonmetal + Circles Nonmetal showing sharing e-’s Metallic Metal only N/A N/A - ide - Prefix’s H 2 Se NA Name N/A

12/11 Do NOW 1. Draw the EDD Form of the following compounds PCl 3 Mg. Cl 2 2. Name the compounds above.

12/11 Agenda 1. Do Now 2. Review HW 3. Review Quiz 4. Metallic bonds HW: 1) Pg 25 skip 1, 3, 6 2) Pgs 26, 27 Ionic Test tomorrow
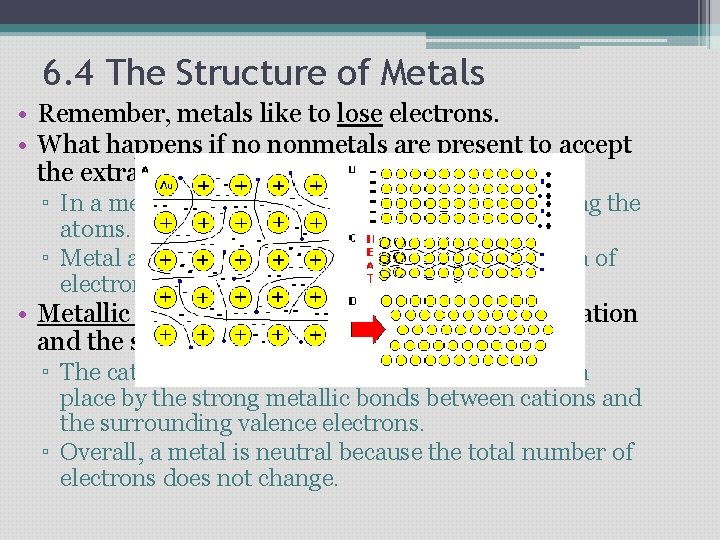
6. 4 The Structure of Metals • Remember, metals like to lose electrons. • What happens if no nonmetals are present to accept the extra electrons? ▫ In a metal, valence electrons are free to move along the atoms. ▫ Metal atoms become cations surrounded by a “sea of electrons” • Metallic Bond- the attraction between a metal cation and the shared electrons that surround it. ▫ The cations in a metal form a lattice that is held in place by the strong metallic bonds between cations and the surrounding valence electrons. ▫ Overall, a metal is neutral because the total number of electrons does not change.
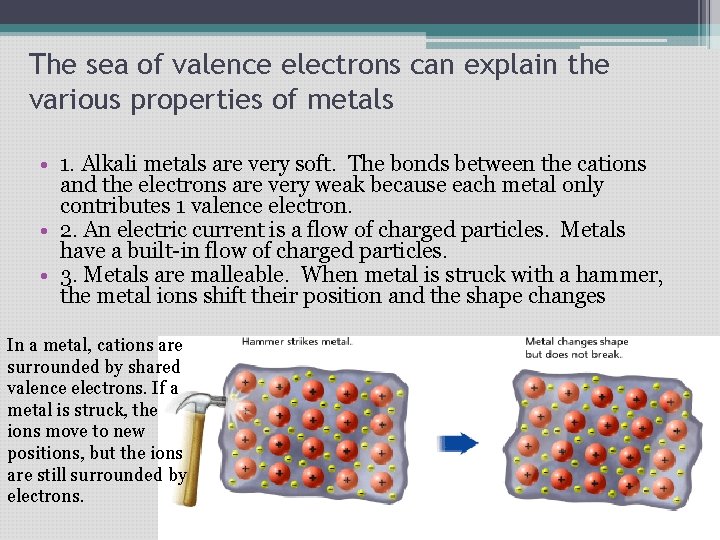
The sea of valence electrons can explain the various properties of metals • 1. Alkali metals are very soft. The bonds between the cations and the electrons are very weak because each metal only contributes 1 valence electron. • 2. An electric current is a flow of charged particles. Metals have a built-in flow of charged particles. • 3. Metals are malleable. When metal is struck with a hammer, the metal ions shift their position and the shape changes In a metal, cations are surrounded by shared valence electrons. If a metal is struck, the ions move to new positions, but the ions are still surrounded by electrons.

Alloys • Gold is a soft metal. It becomes harder when mixed with Ag, Cu, Ni, or Zn • Alloy – A mixture of two or more elements, at least one of which is a metal. ▫ Alloys have the characteristic properties of metals. ▫ EX: �Gold that is 100 percent pure is labeled 24 -karat gold. �Gold jewelry that has a 12 -karat label is only 50 percent gold. �Jewelry that has an 18 -karat label is 75 percent gold.

Alloys • Bronze – (copper and tin) - Bronze is hard and durable enough to be used for propellers on ships and for statues. • Stainless Steel – sulfur, manganese, phophorus, silicon, and chromium • Steel cable used in bridges are strong enough to resist forces that might stretch the cables or cause them to break. • Aluminum-magnesium alloy is used to make planes so they are light weight like aluminum and won’t bend easily

Begin the Review… Use the word bank to complete the statements: Covalent Bond Polar Covalent 1) 2) 3) 4) 5) 6) 7) 8) 9) Ionic Bond Non-Polar Covalent Metallic Bonding is found between cations and anions This is found between atoms of nonmetals This is found between atoms of metals This is the bond that produces electrical conductivity This is the force that holds crystals of table salt together CH 4 is this kind of compound NCl 3 is this kind of compound The force that holds the hydrogen to the oxygen in water This is between 2 nitrogen atoms of nitrogen

- Slides: 64