1122020 Prof Mazen Qato 1 Objectives At the

11/2/2020 Prof. Mazen Qato 1

Objectives At the end of this sessions students should be able to: 1. List and discuss common routes of drug administration 2. Explain the absorption pattern of common routes of drug administration. 3. Describe various dosage forms and their absorption pattern 4. Discuss noveldrug deliv ery systems like transdermal drug delivery etc…

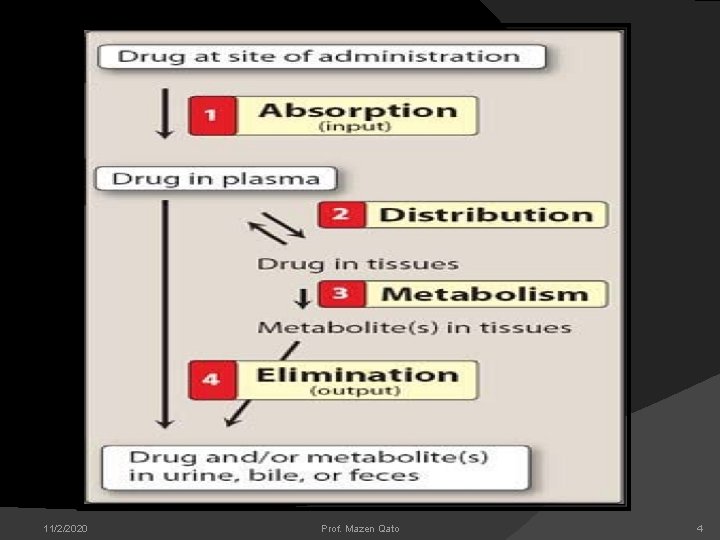
PHARMACOKINETICS The quantitative study of drug movement in, through and out of the body(Effect of Body on Drug, fate of drug). Pharmacokinetic processes include I. Absorption of drug II. Distribution of drug III. Metabolism of drug IV. Excretion of drug
11/2/2020 Prof. Mazen Qato 4

Pharmacokinetics is significant for Route of drug administration Dose of drug Latency of onset Time of peak action Duration of action Frequency of drug administration
11/2/2020 Prof. Mazen Qato 6
11/2/2020 Prof. Mazen Qato 7

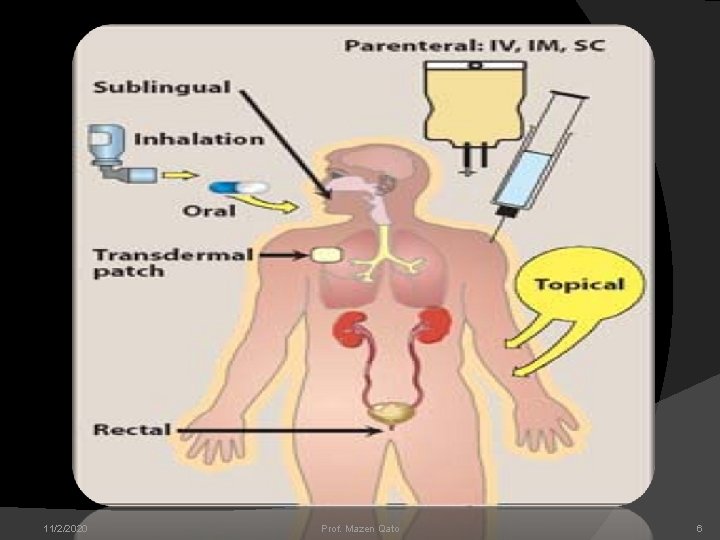
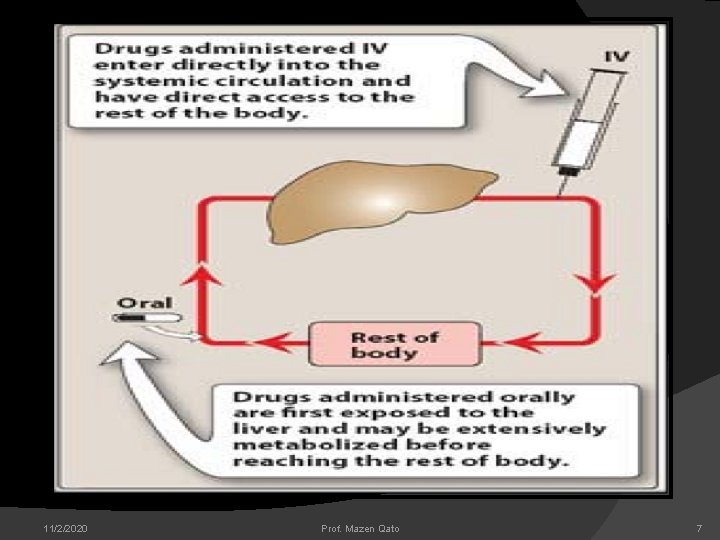
Routes of drug administration Factors governing choice of route i. Physical and chemical properties of drug ii. Site of desired action iii. Rate and extent of absorption of drug from different routes iv. Effect of digestive juices and first pass metabolism. v. Rapidity with which the response is desired vi. Condition of the patient

PHARMACOKINETICS II DRUG ABSORPTION 11/2/2020 Prof. Mazen Qato 9

Objectives Discuss factors affecting drug absorption 2. Emphasize factors influencing bioavailability and bioequivalence 3. Explain the concept of Henderson Hasselbach equation. 1.

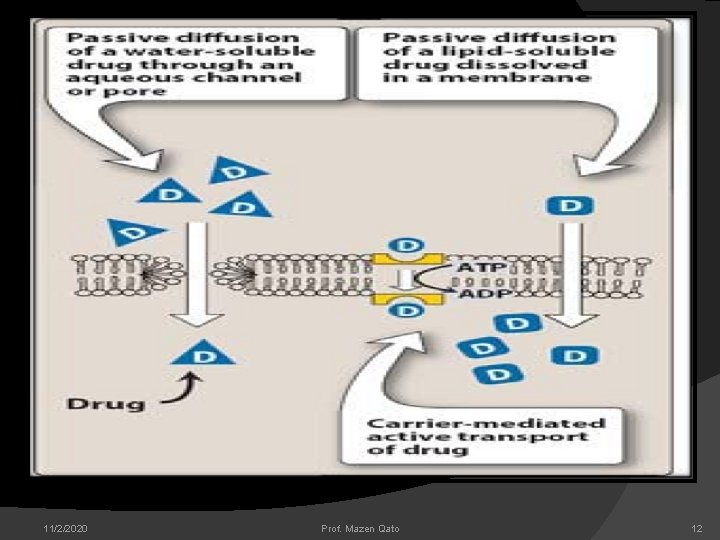
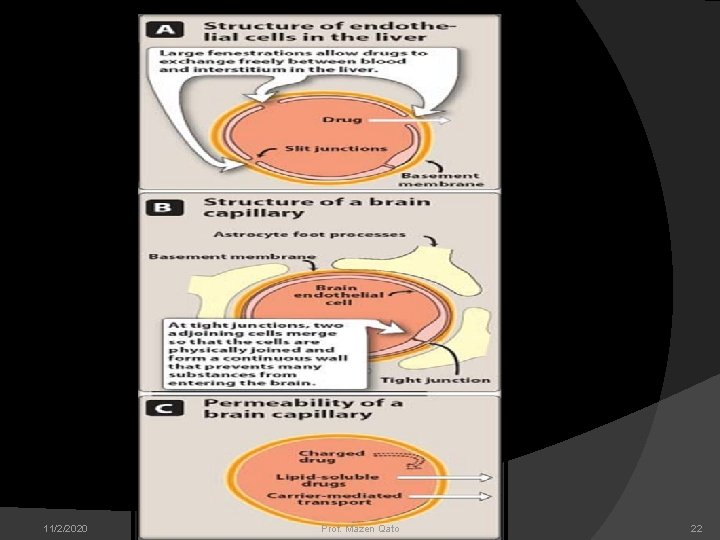
DRUG ABSORPTION Absorption is the movement of drug from its site of administration into the blood stream. Factors affecting drug absorption: Aqueous solubility(transport across membrane) Effect of p. H Area of absorbing surface Vascularity of the absorbing surface Route of administration
11/2/2020 Prof. Mazen Qato 12

11/2/2020 Prof. Mazen Qato 13
11/2/2020 Prof. Mazen Qato 14

11/2/2020 Prof. Mazen Qato 15

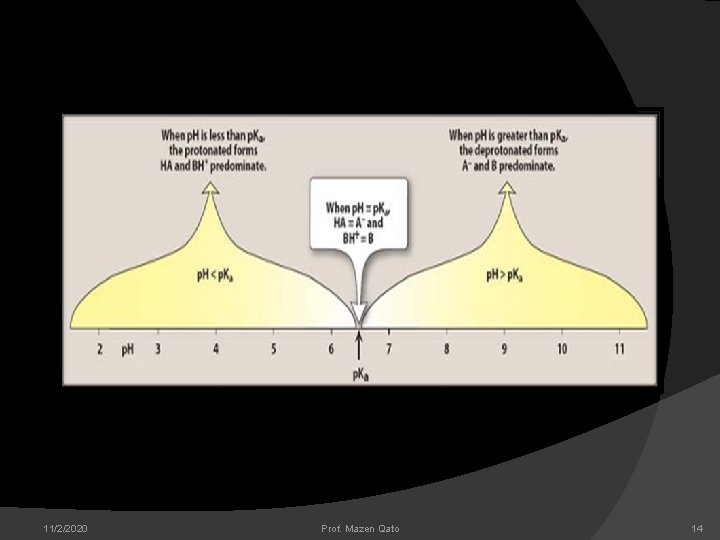
Pharmacological implications of Henderson Hasselbach’s equation: Most drugs are weak electrolytes, that is, their ionization is p. H dependant. Weakly acidic drugs which form salts with cations ( example: sodium phenobarbitone, sodium sulphadiazine , potassium penicillin V) ionise more at alkaline p. H.

Weakly basic drugs which form salts with anions(example: atropine sulphate, ephedrine hydrochloride, chloroquine phosphate) ionize more at acidic ph. Ions being lipid insoluble do not diffuse across a biological membrane. Acidic drug example aspirin(p. Ka =3. 5) are largely unionized at acidic gastric p. H and are absorbed from the stomach.

Basic drugs like atropine(p. Ka =10) are largely unionized and absorbed only when they reach small intestine. Acidic drug are ionized more in alkaline urine – do not diffuse in the kidney and are excreted faster. Accordingly, basic drugs are excreted faster if urine is acidified. This principle is used in treatment of drug overdose.

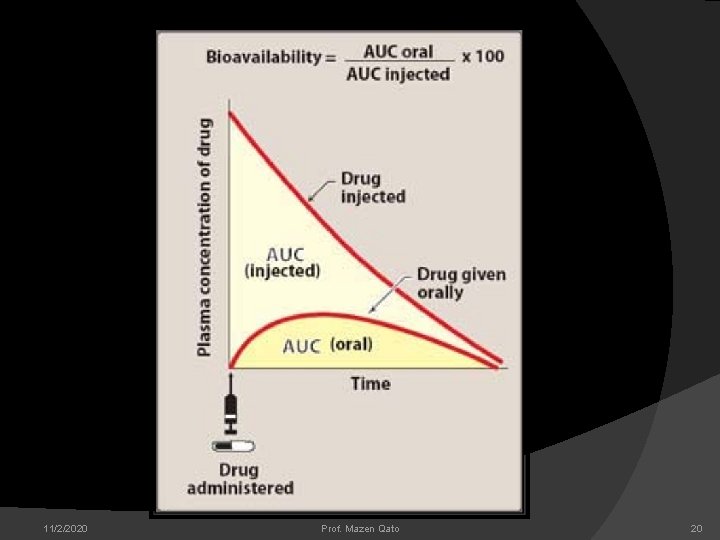
Bioavailability It is a measure of the fraction of administered dose of a drug that reaches the systemic circulation in the unchanged form. Bioavailability of a drug injected intravenously(IV) is 100%. Calculated from comparing plasma level of a drug after a particular route of administration with plasma drug level achieved by IV injection.
11/2/2020 Prof. Mazen Qato 20

Factors that influence bioavailability of a drug 1. First pass hepatic metabolism 2. Solubility of a drug 3. Chemical stability 4. Nature of drug formulation. Bioavailibility variation assumes practical significance for drugs with low safety margin (digoxin) or where dosage needs precise control.
11/2/2020 Prof. Mazen Qato 22
- Slides: 22