11 Reactions of Alkyl Halides Nucleophilic Substitutions and

11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations Based on Mc. Murry’s Organic Chemistry, 6 th edition

Alkyl Halides React with Nucleophiles and Bases n n Alkyl halides are polarized at the carbonhalide bond, making the carbon electrophilic Nucleophiles will replace the halide in C -X bonds of many alkyl halides (reaction as Lewis base) 2
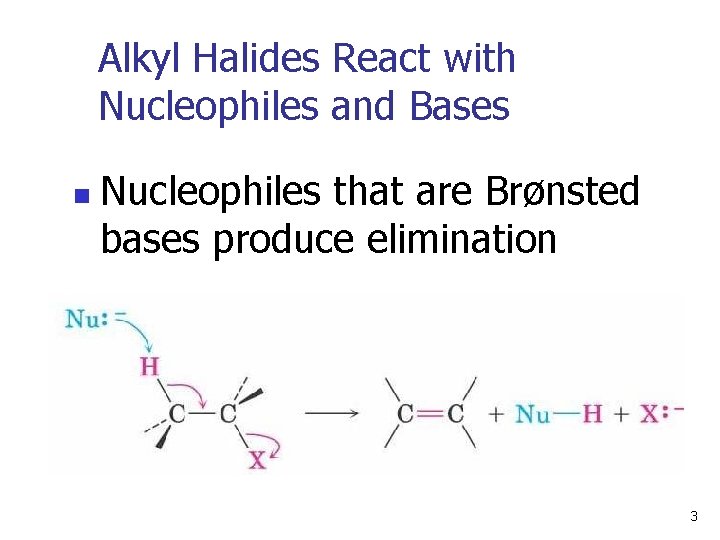
Alkyl Halides React with Nucleophiles and Bases n Nucleophiles that are Brønsted bases produce elimination 3

Substitution vs. Elimination 4

The Nature of Substitution n n Substitution requires that a "leaving group", which is also a Lewis base, departs from the reacting molecule. A nucleophile is a reactant that can be expected to participate as a Lewis base in a substitution reaction. 5

Substitution Mechanisms n S N 1 n n n Two steps with carbocation intermediate Occurs in 3°, allyl, benzyl S N 2 n n Concerted mechanism - without intermediate Occurs in primary, secondary 6

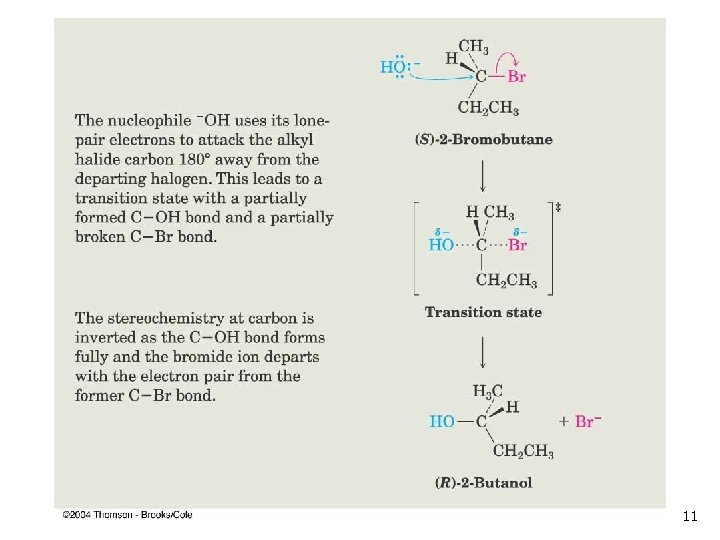
The SN 2 Reaction n Reaction occurs with inversion of configuration at electrophilic C Follows second order reaction kinetics Ingold nomenclature to describe ratedetermining step: n n n S=substitution N (subscript) = nucleophilic 2 = both nucleophile and electrophile in ratedetermining step (bimolecular) 7
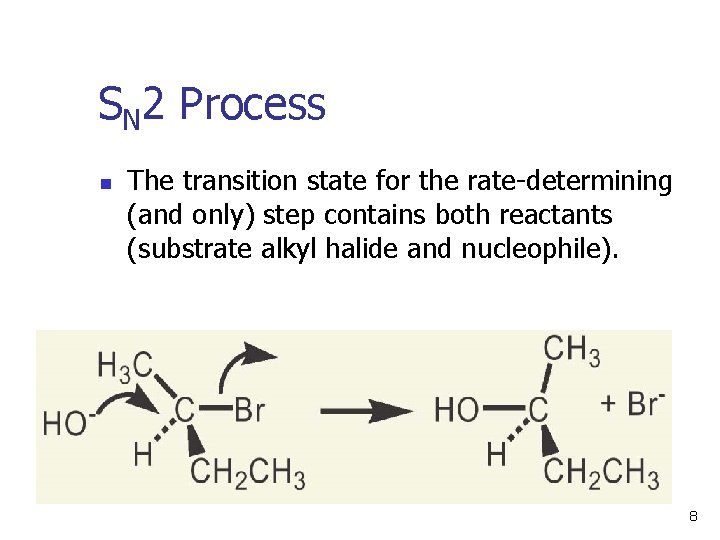
SN 2 Process n The transition state for the rate-determining (and only) step contains both reactants (substrate alkyl halide and nucleophile). 8
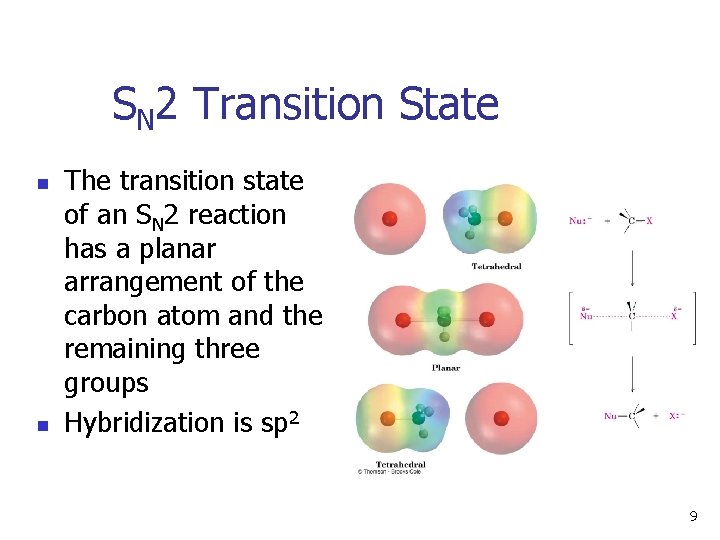
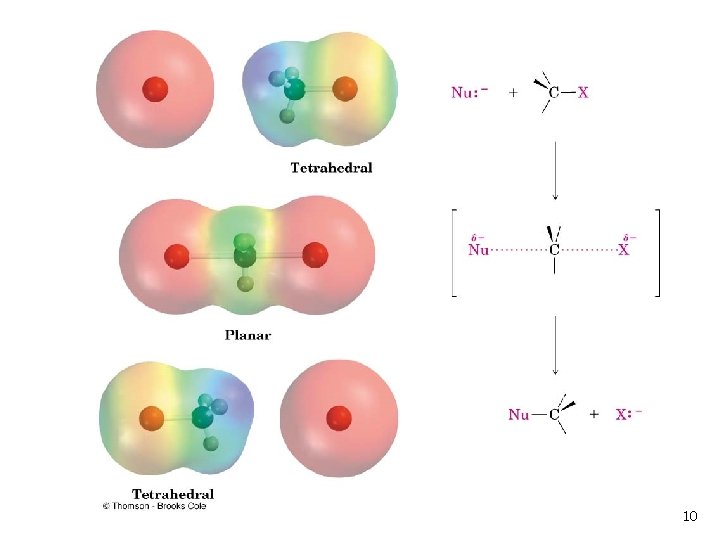
SN 2 Transition State n n The transition state of an SN 2 reaction has a planar arrangement of the carbon atom and the remaining three groups Hybridization is sp 2 9
10
11

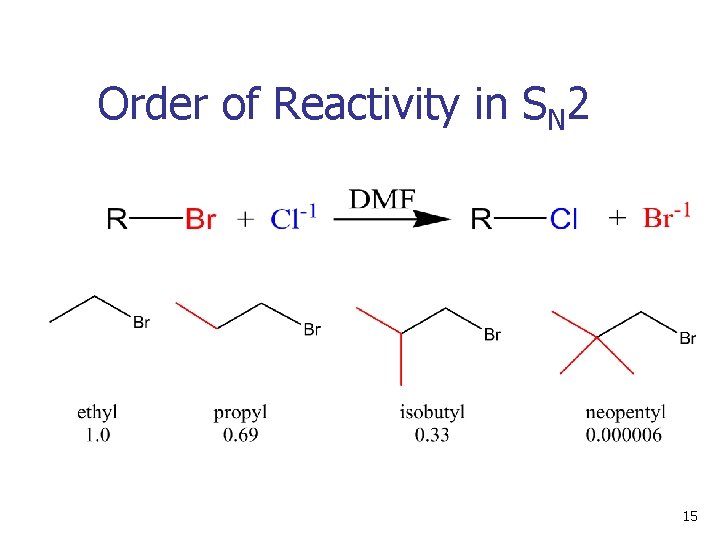
11. 5 Characteristics of the SN 2 Reaction n n n Sensitive to steric effects Methyl halides are most reactive Primary are next most reactive Unhindered secondary halides react under some conditions Tertiary are unreactive by this path No reaction at C=C (vinyl or aryl halides) 12

Order of Reactivity in SN 2 n The more alkyl groups connected to the reacting carbon, the slower the reaction 13

Vinyl and Aryl Halides: 14
Order of Reactivity in SN 2 15

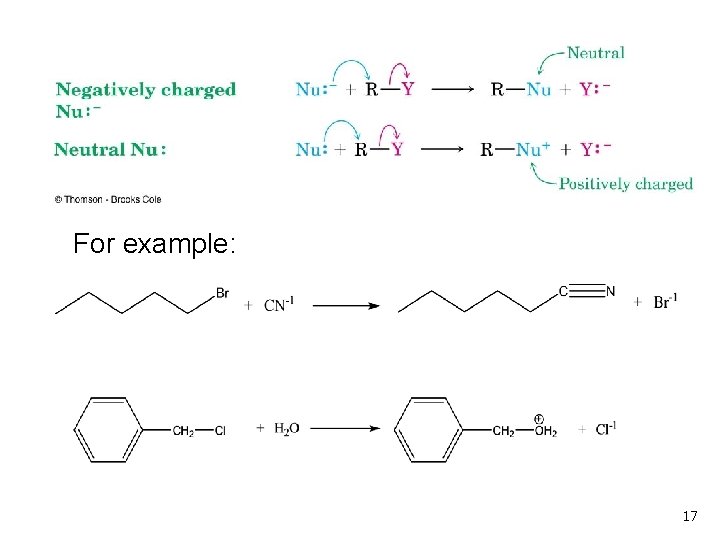
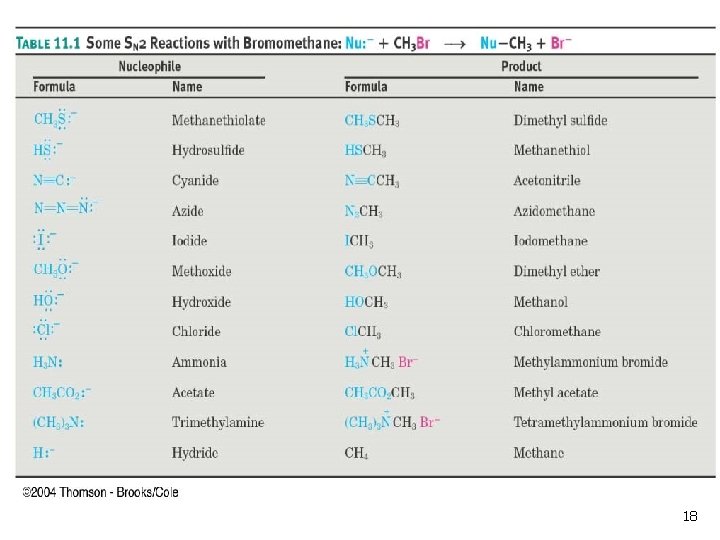
The Nucleophile n n Neutral or negatively charged Lewis base Reaction increases coordination (adds a new bond) at the nucleophile n n n Neutral nucleophile acquires positive charge Anionic nucleophile becomes neutral See Table 11 -1 for an illustrative list 16
For example: 17
18

Relative Reactivity of Nucleophiles n n Depends on reaction and conditions More basic nucleophiles react faster (for similar structures. See Table 11 -2) Better nucleophiles are lower in a column of the periodic table Anions are usually more reactive than neutrals 19
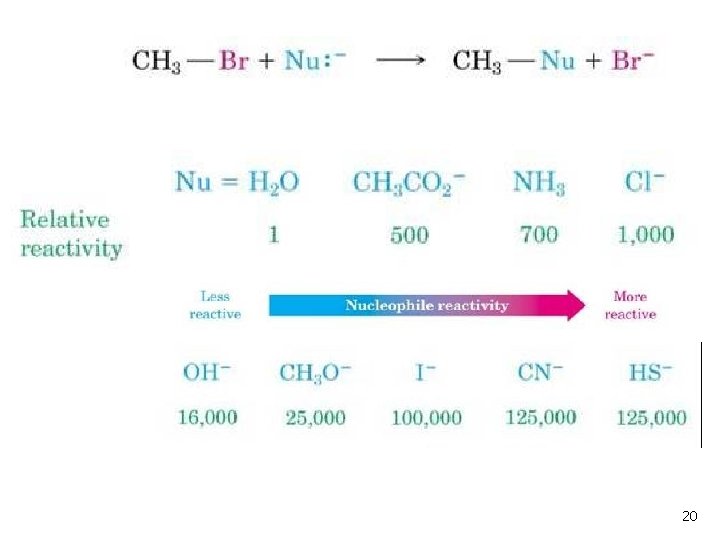
20

The Leaving Group A good leaving group reduces the energy of activation of a reaction n Stable anions that are weak bases (conjugate bases of strong acids) are usually excellent leaving groups n Stronger bases (conjugate bases of weaker acids) are usually poorer leaving groups n 21
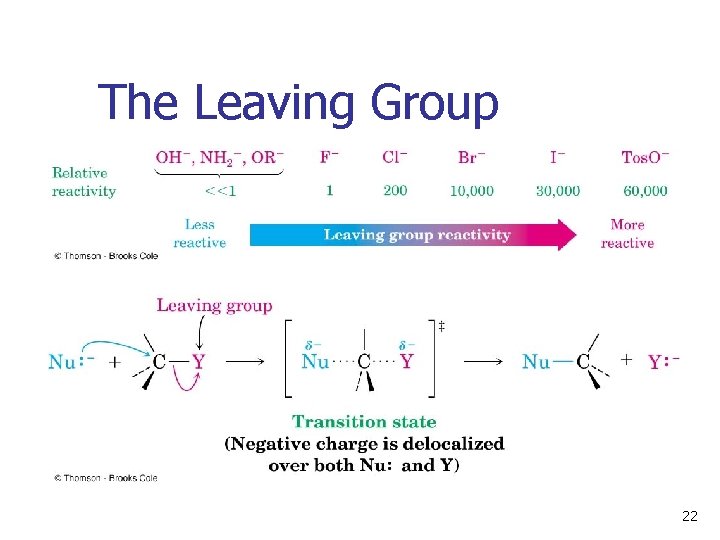
The Leaving Group 22

Poor Leaving Groups n If a group is very basic or very small, it does not undergo nucleophilic substitution. 23

The Solvent n n n Protic solvents (which can donate hydrogen bonds; -OH or –NH) slow SN 2 reactions by associating with reactants (anions). Energy is required to break interactions between reactant and solvent Polar aprotic solvents (no NH, OH, SH) form weaker interactions with substrate and permit faster reaction 24
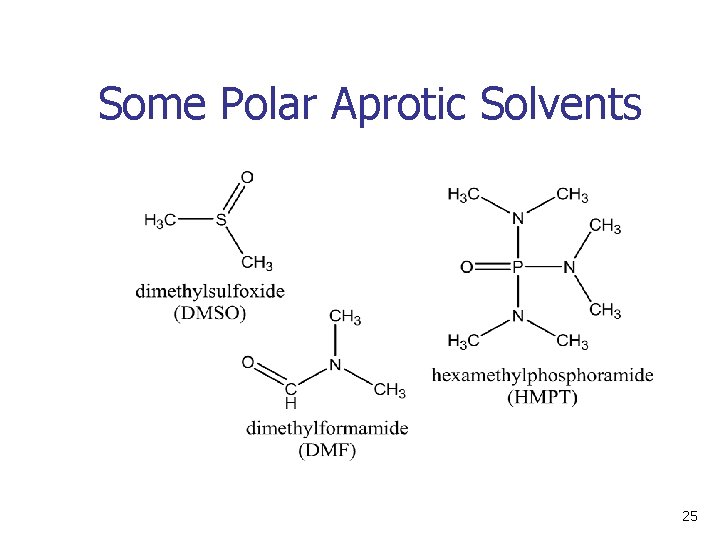
Some Polar Aprotic Solvents 25

Summary of SN 2 Characteristics: n n n Substrate: CH 3 ->1 o>2 o>>3 o (Steric effect) Nucleophile: Strong, basic nucleophiles favor the reaction Leaving Groups: Good leaving groups (weak bases) favor the reaction Solvent: Aprotic solvents favor the reaction; protic reactions slow it down by solvating the nucleophile Stereochemistry: 100% inversion 26

Prob. 11. 36 Arrange in order of SN 2 reactivity 27
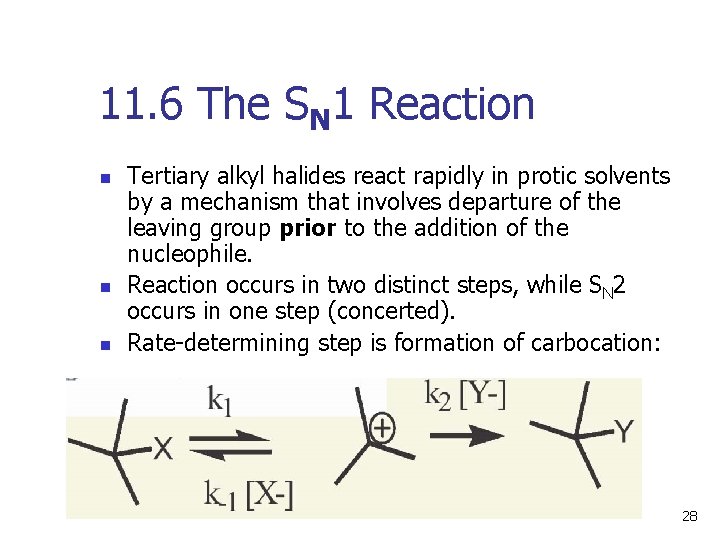
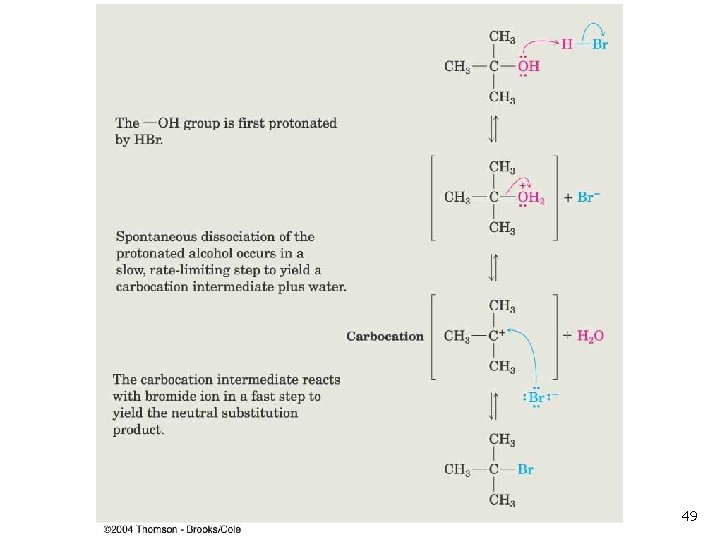
11. 6 The SN 1 Reaction n Tertiary alkyl halides react rapidly in protic solvents by a mechanism that involves departure of the leaving group prior to the addition of the nucleophile. Reaction occurs in two distinct steps, while SN 2 occurs in one step (concerted). Rate-determining step is formation of carbocation: 28
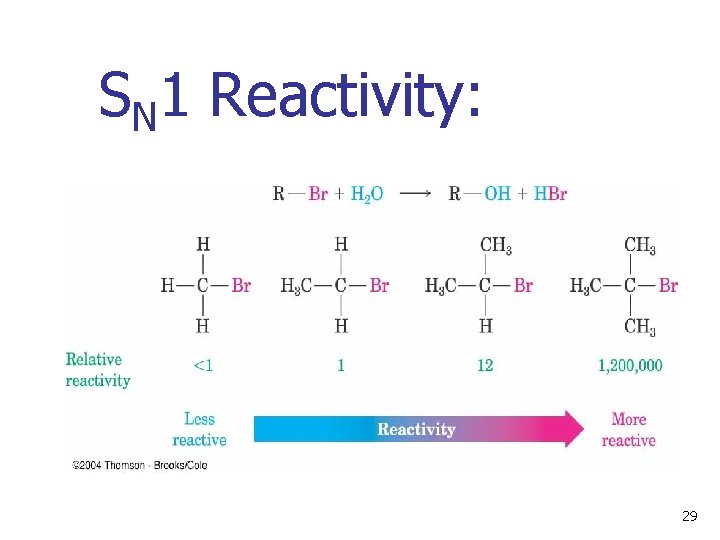
SN 1 Reactivity: 29
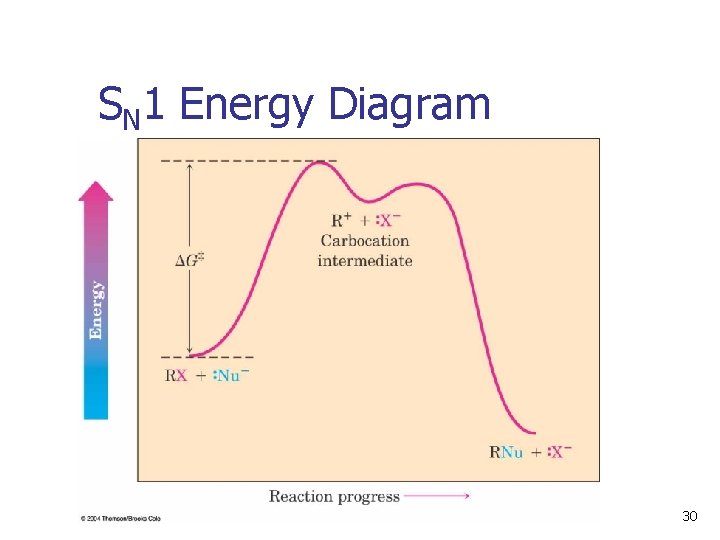
SN 1 Energy Diagram k 1 k-1 k 2 30

Rate-Limiting Step n n The overall rate of a reaction is controlled by the rate of the slowest step The rate depends on the concentration of the species and the rate constant of the step The step with the largest energy of activation is the rate-limiting or rate-determining step. See Figure 11. 9 – the same step is ratedetermining in both directions) 31
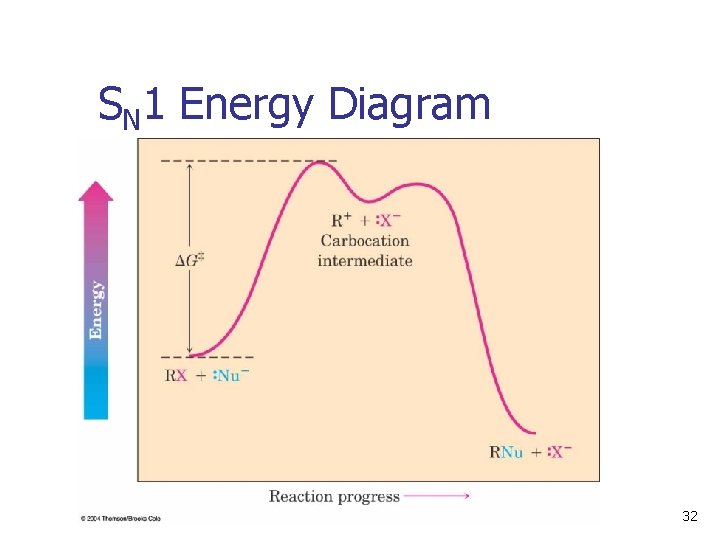
SN 1 Energy Diagram k 1 k-1 k 2 32

33
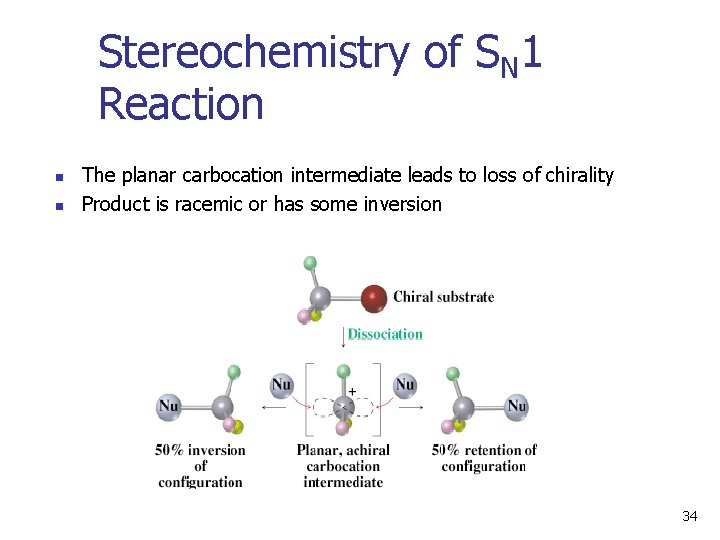
Stereochemistry of SN 1 Reaction n n The planar carbocation intermediate leads to loss of chirality Product is racemic or has some inversion 34
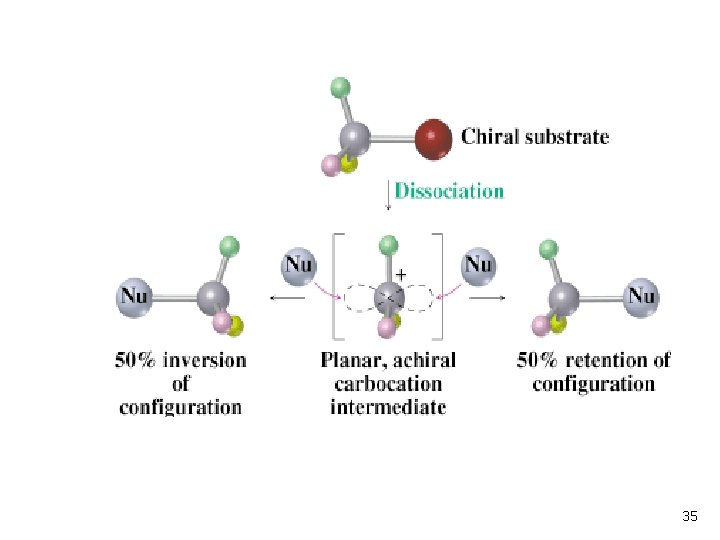
35

Stereochemistry of SN 1 Reaction • Carbocation is usually biased to react on side opposite leaving group because it is unsymmetrically solvated • The second step may occur with the carbocation loosely associated with leaving group. • The result is racemization with some inversion: 36
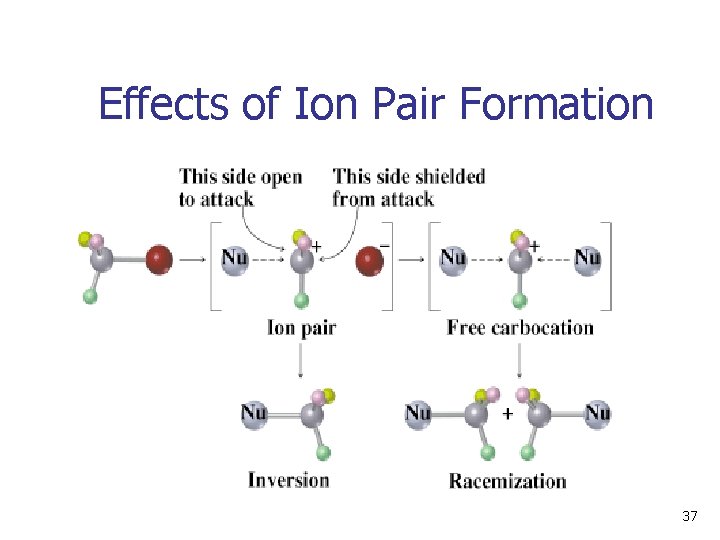
Effects of Ion Pair Formation 37

Prob. 11. 9: What is the % inversion & racemization? 38

11. 9 Characteristics of the SN 1 Reaction n Tertiary alkyl halide is most reactive by this mechanism n Controlled by stability of carbocation 39

Relative Reactivity of Halides: 40

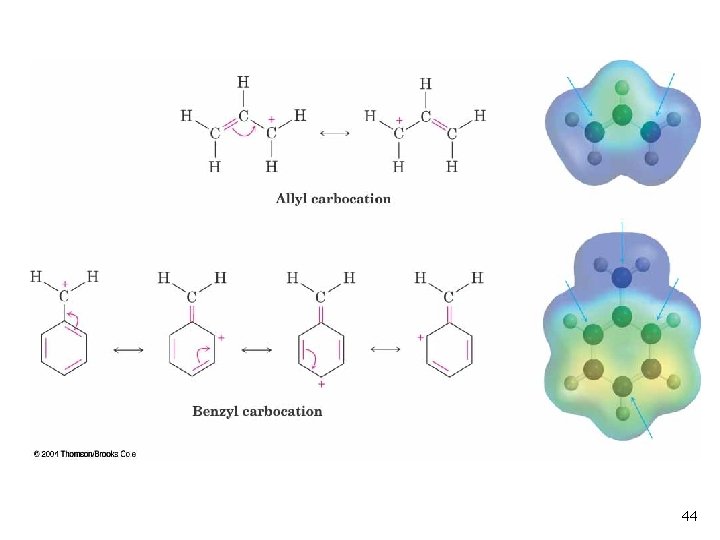
Delocalized Carbocations Delocalization of cationic charge enhances stability n Primary allyl is more stable than primary alkyl n Primary benzyl is more stable than allyl n 41
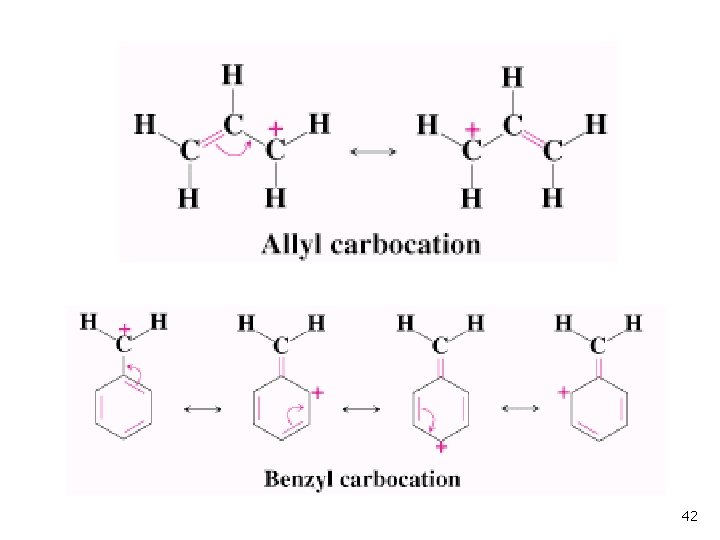
42
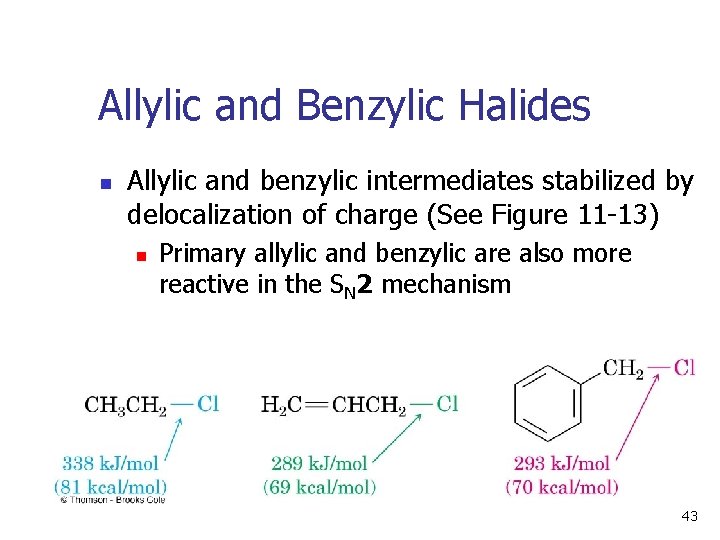
Allylic and Benzylic Halides n Allylic and benzylic intermediates stabilized by delocalization of charge (See Figure 11 -13) n Primary allylic and benzylic are also more reactive in the SN 2 mechanism 43
44

Relative SN 1 rates (formolysis): RCl + HCOO-1 45

Formation of the allylic cation: 46

Effect of Leaving Group on SN 1 n Critically dependent on leaving group n n n Reactivity: the larger halides ions are better leaving groups In acid, OH of an alcohol is protonated and leaving group is H 2 O, which is still less reactive than halide p-Toluensulfonate (Tos. O-) is an excellent leaving group 47

Nucleophiles in SN 1 n n Since nucleophilic addition occurs after formation of carbocation, reaction rate is not normally affected by nature or concentration of nucleophile The nucleophile must be preferably neutral (not basic; example: CH 3 OH rather than CH 3 O-) to prevent competition with elimination reactions. 48
49

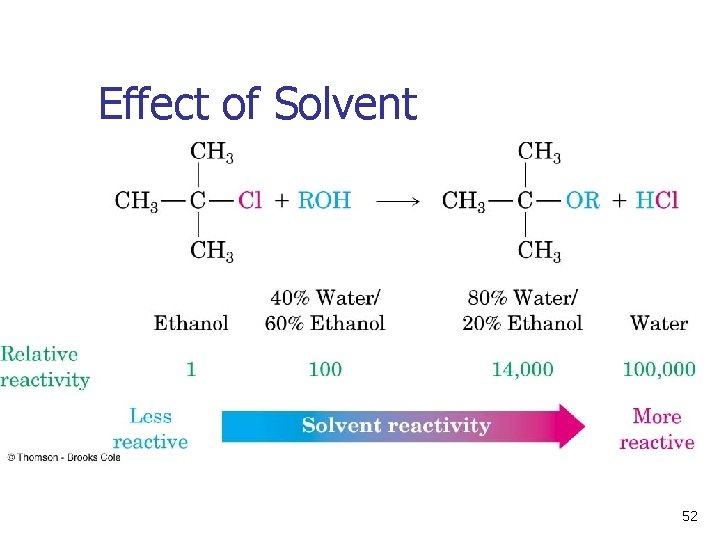
Solvent Is Critical in SN 1 n The solvent stabilizes the carbocation, and also stabilizes the associated transition state. This controls the rate of the reaction. Solvation of a carbocation by water 50

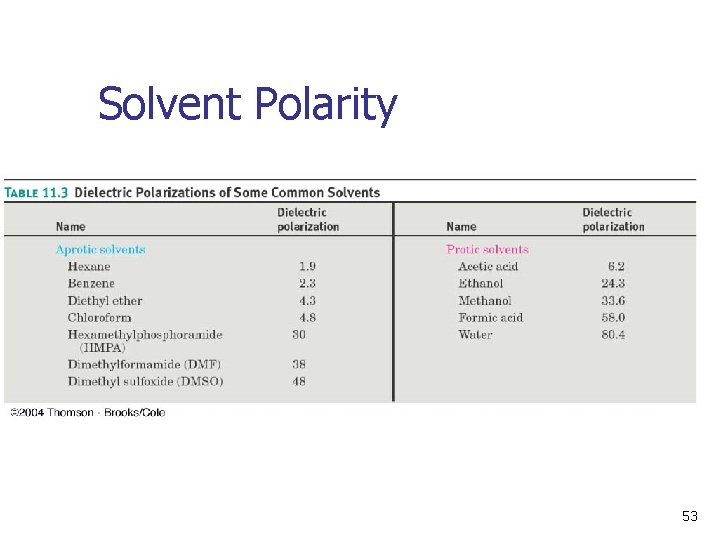
Polar Solvents Promote Ionization n n Polar, protic and unreactive Lewis base solvents facilitate formation of R+ Solvent polarity is measured as dielectric polarization (P) (Table 11 -3) 51
Effect of Solvent 52
Solvent Polarity 53

Summary of SN 1 Characteristics: n n n Substrate: Benzylic~allylic>3 o >2 o Nucleophile: Does not affect reaction (although strong bases promote elimination) Leaving Groups: Good leaving groups (weak bases) favor the reaction Solvent: Polar solvents favor the reaction by stabilizing the carbocation. Stereochemistry: racemization (with some inversion) 54
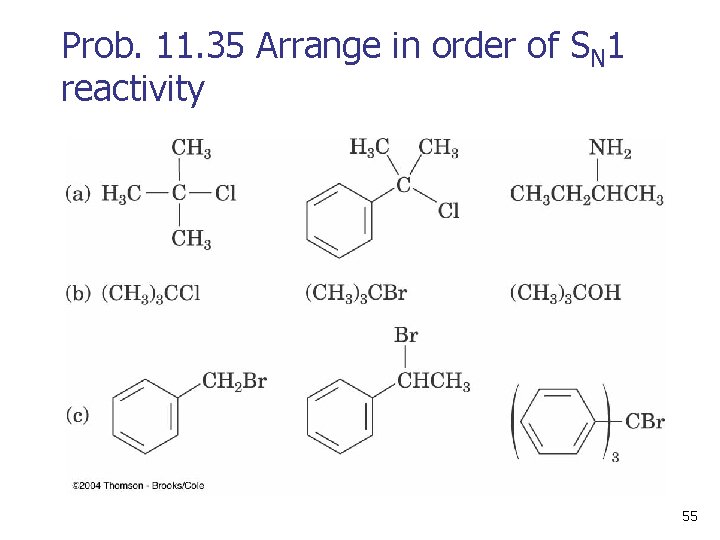
Prob. 11. 35 Arrange in order of SN 1 reactivity 55
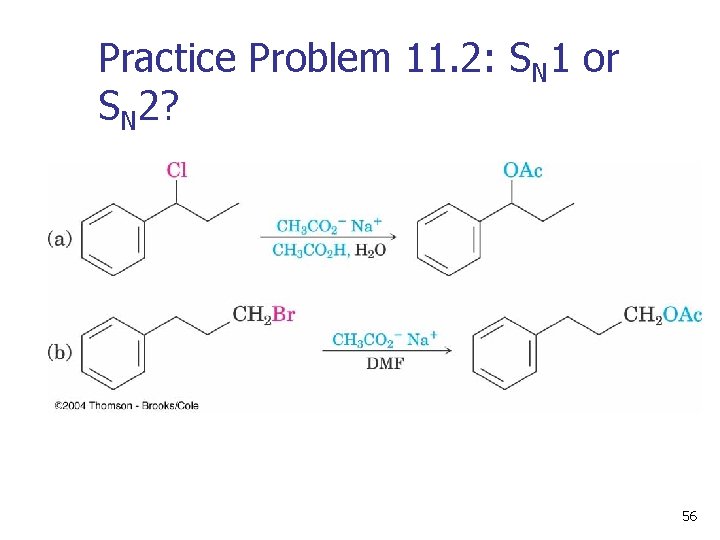
Practice Problem 11. 2: SN 1 or SN 2? 56
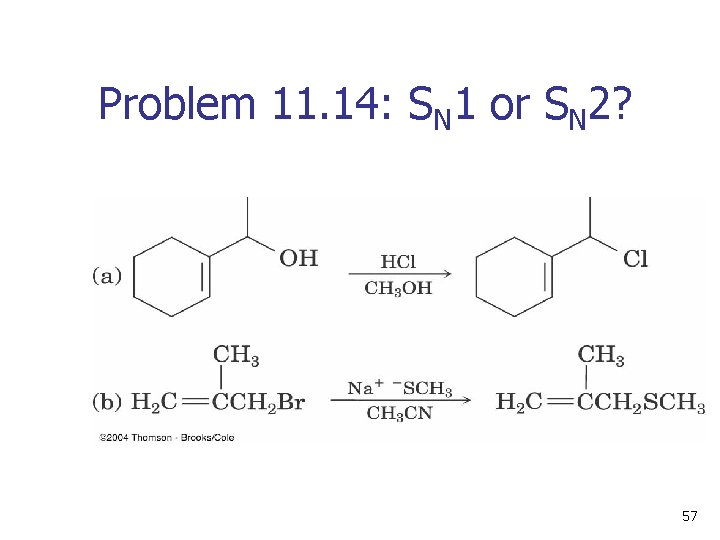
Problem 11. 14: SN 1 or SN 2? 57

11. 10 Alkyl Halides: Elimination n Elimination is an alternative pathway to substitution Elimination is formally the opposite of addition, and generates an alkene It can compete with substitution and decrease yield, especially for SN 1 processes 58

Zaitsev’s Rule for Elimination Reactions (1875) n In the elimination of HX from an alkyl halide, the more highly substituted alkene product predominates 59

Mechanisms of Elimination Reactions n n Ingold nomenclature: E – “elimination” E 1 (1 st order): X- leaves first to generate a carbocation n n a base abstracts a proton from the carbocation E 2 (2 nd order): Concerted transfer of a proton to a base and departure of leaving group 60
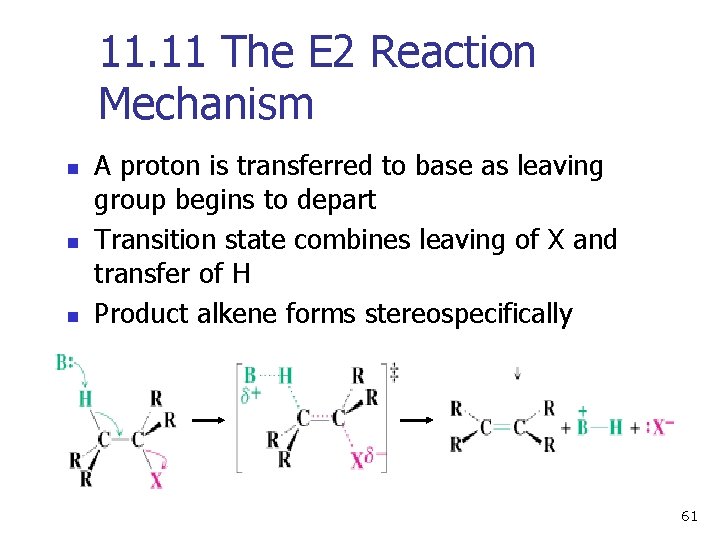
11. 11 The E 2 Reaction Mechanism n n n A proton is transferred to base as leaving group begins to depart Transition state combines leaving of X and transfer of H Product alkene forms stereospecifically 61
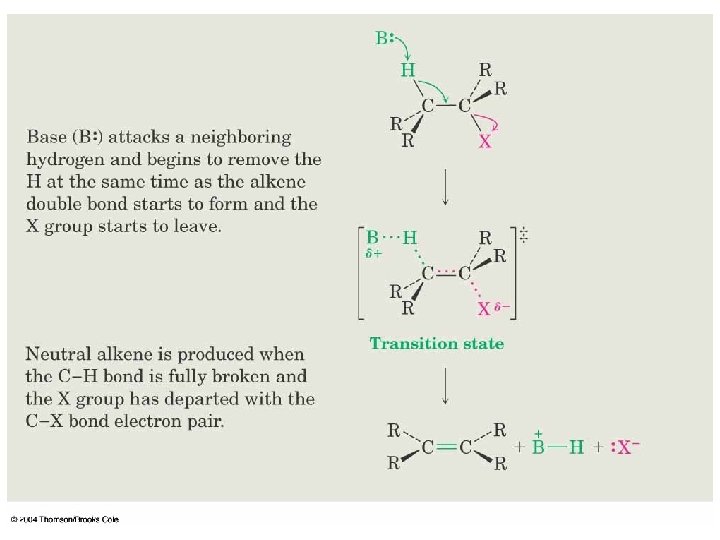
62

E 2 Reaction Kinetics One step (concerted): rate law dependent on base and alkyl halide n Rate = k[R-X][B] n Reaction goes faster with stronger base, better leaving group n 63
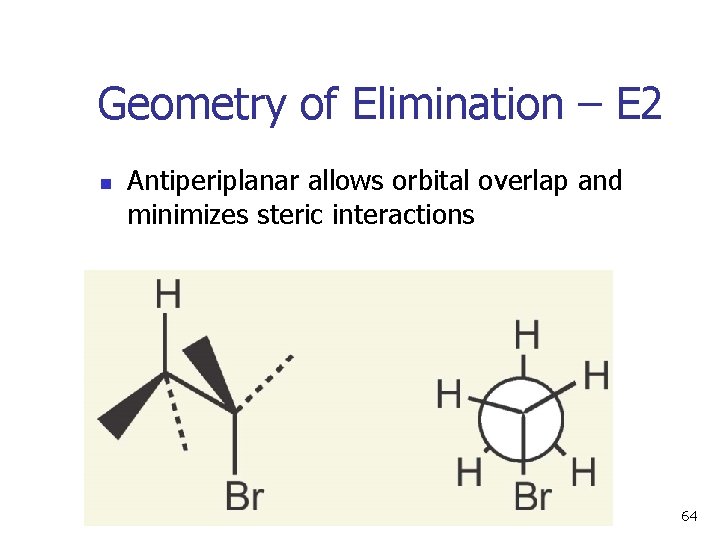
Geometry of Elimination – E 2 n Antiperiplanar allows orbital overlap and minimizes steric interactions 64
65
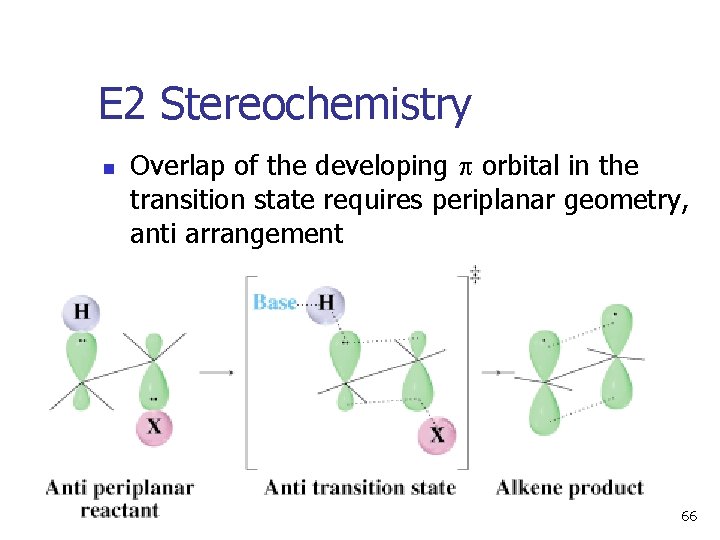
E 2 Stereochemistry n Overlap of the developing orbital in the transition state requires periplanar geometry, anti arrangement 66
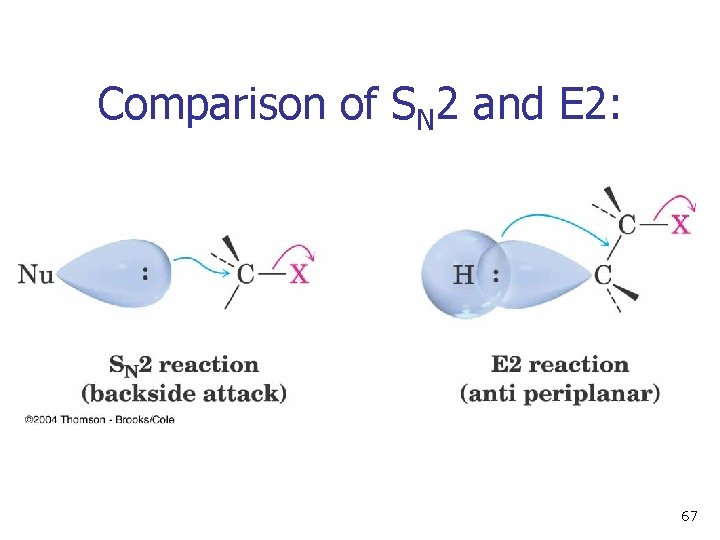
Comparison of SN 2 and E 2: 67
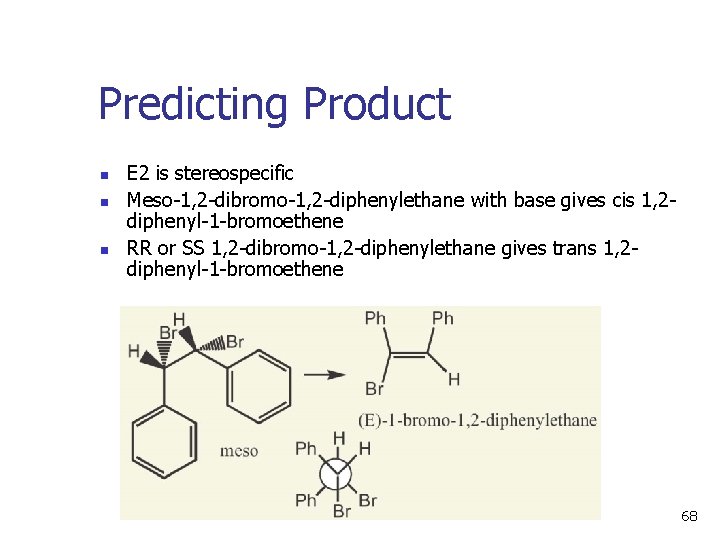
Predicting Product n n n E 2 is stereospecific Meso-1, 2 -dibromo-1, 2 -diphenylethane with base gives cis 1, 2 diphenyl-1 -bromoethene RR or SS 1, 2 -dibromo-1, 2 -diphenylethane gives trans 1, 2 diphenyl-1 -bromoethene 68
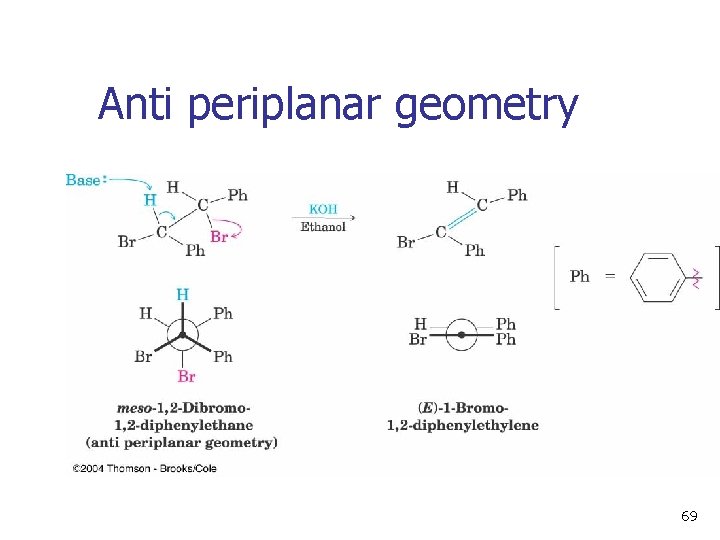
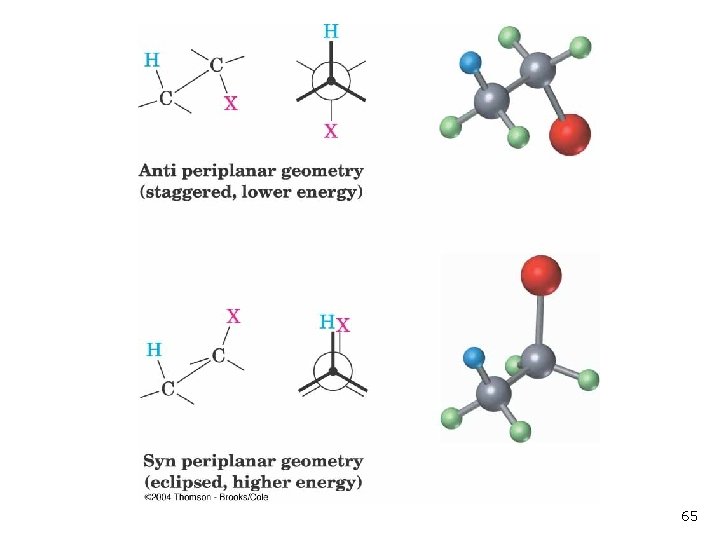
Anti periplanar geometry 69

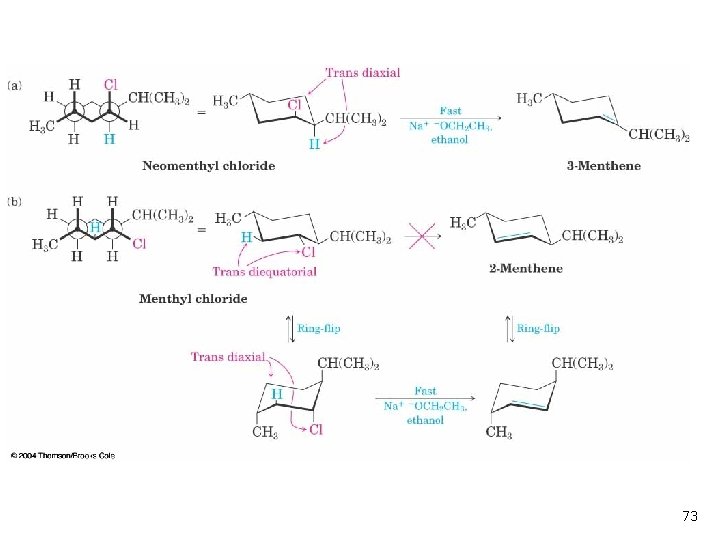
11. 12 Elimination From Cyclohexanes n n Abstracted proton and leaving group should align trans-diaxial to be anti periplanar (app) in approaching transition state (see Figures 11 -19 and 11 -20) Equatorial groups are not in proper alignment 70
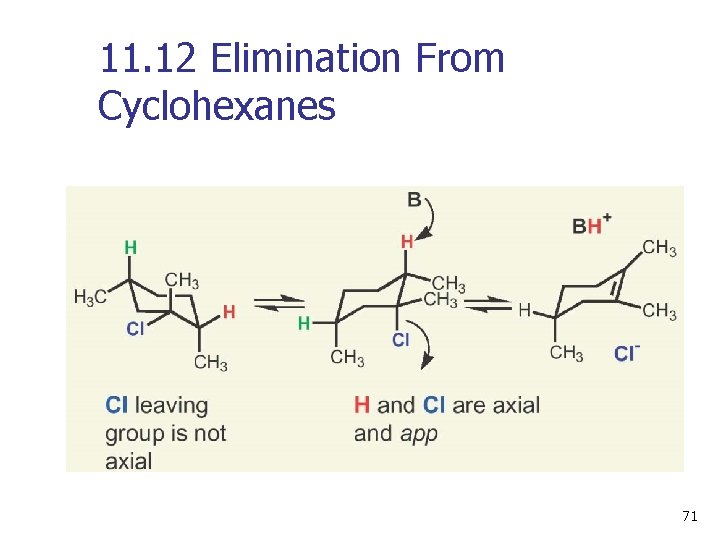
11. 12 Elimination From Cyclohexanes 71
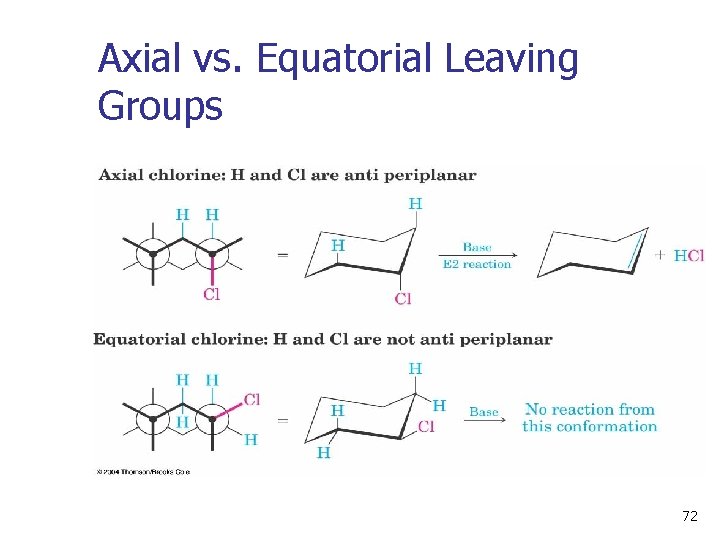
Axial vs. Equatorial Leaving Groups 72
73
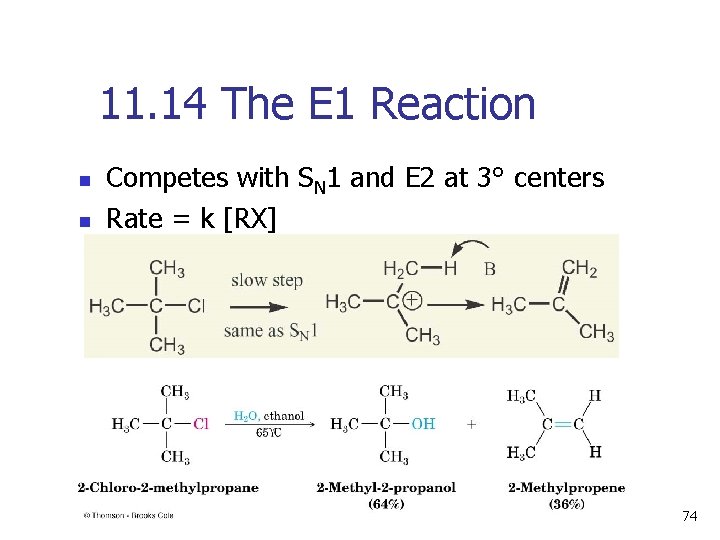
11. 14 The E 1 Reaction n n Competes with SN 1 and E 2 at 3° centers Rate = k [RX] 74

Stereochemistry of E 1 Reactions n n E 1 is not stereospecific and there is no requirement for alignment Product has Zaitsev orientation because the step that controls product formation is loss of proton after formation of carbocation 75

Comparing E 1 and E 2 Strong base is needed for E 2 but not for E 1 n E 2 is stereospecifc, E 1 is not n E 1 gives Zaitsev orientation; E 2 may not due to stereospecificity n E 1 is favored in protic solvents; competes with SN 1 n 76
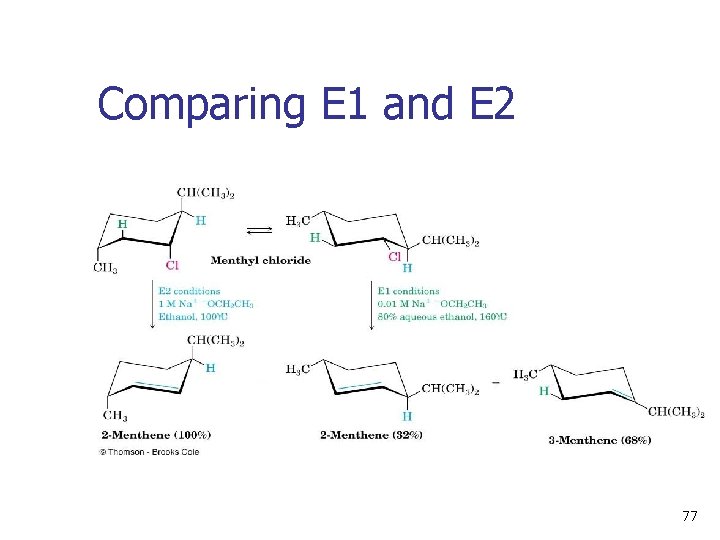
Comparing E 1 and E 2 77
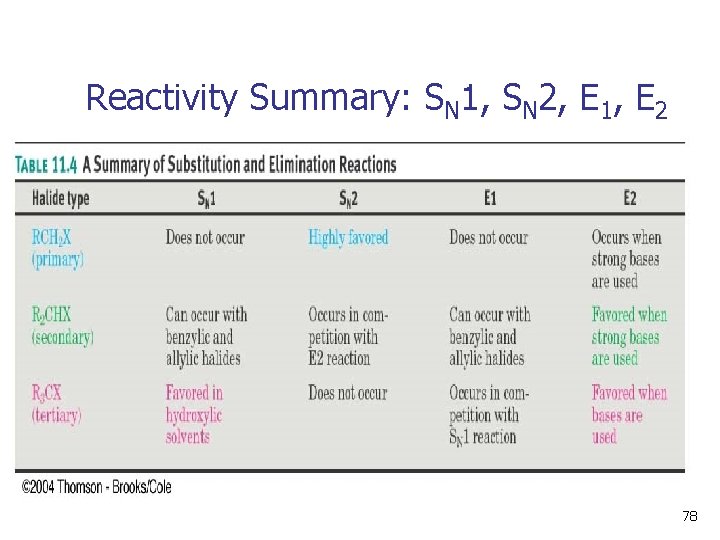
Reactivity Summary: SN 1, SN 2, E 1, E 2 78

General Pattern by Substrate 79
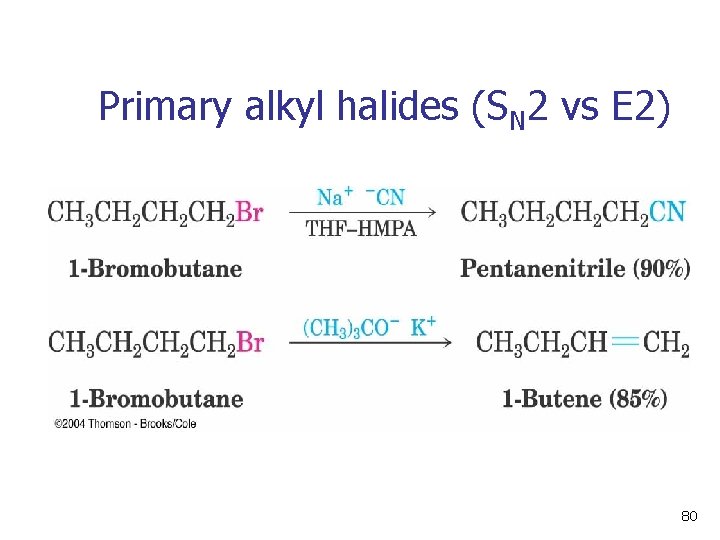
Primary alkyl halides (SN 2 vs E 2) 80
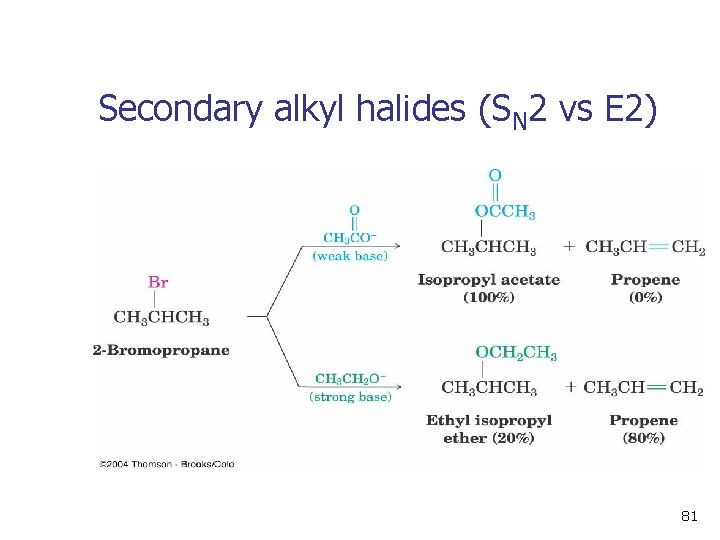
Secondary alkyl halides (SN 2 vs E 2) 81
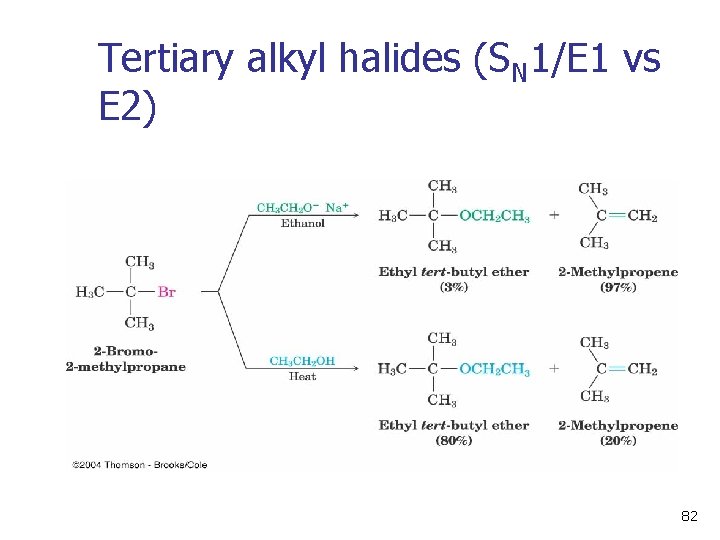
Tertiary alkyl halides (SN 1/E 1 vs E 2) 82
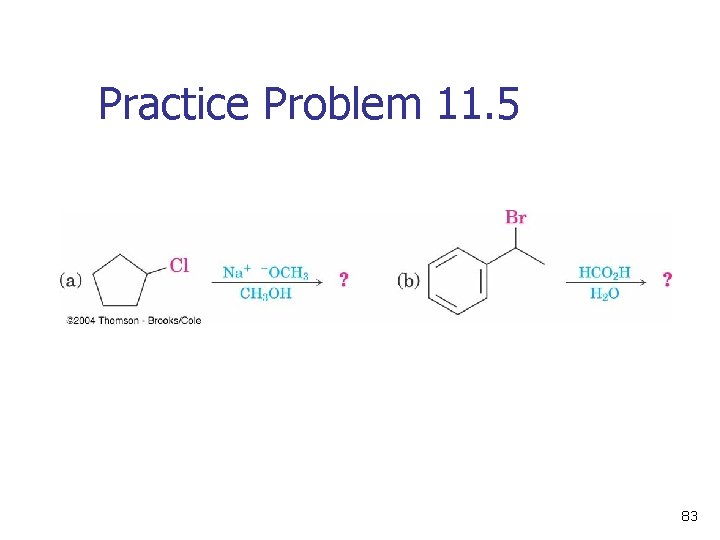
Practice Problem 11. 5 83
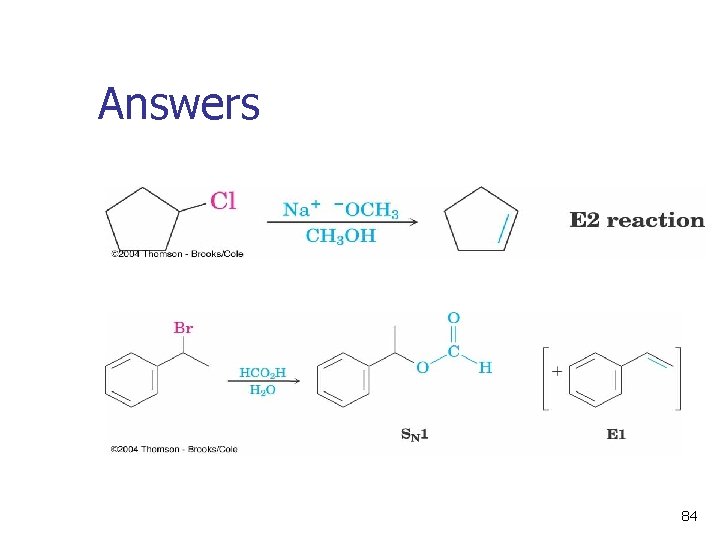
Answers 84
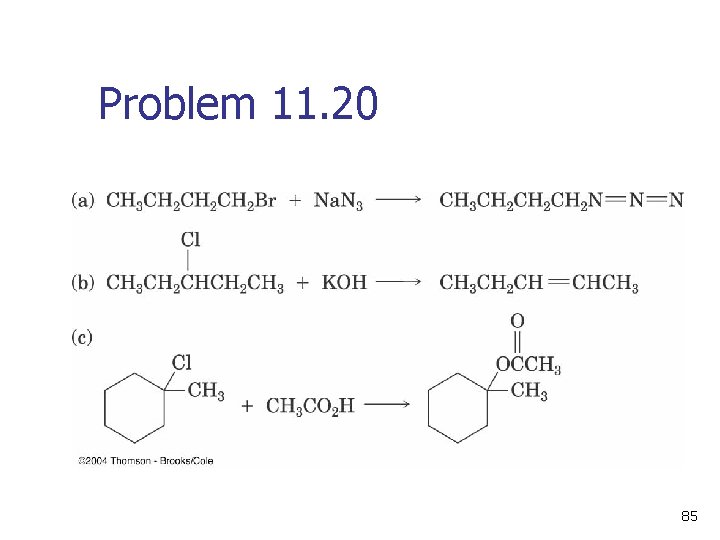
Problem 11. 20 85
- Slides: 85