11 9 Buffers A buffer solution maintains the

11. 9 Buffers A buffer solution maintains the p. H by neutralizing small amounts of added acid or base. An acid must be present to react with any OH− added, and a base must be present to react with any H 3 O+ added. Learning Goal Describe the role of buffers in maintaining the p. H of a solution; calculate the p. H of a buffer. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Buffers When an acid or a base is added to water, the p. H changes drastically. In a buffer solution, the p. H is maintained; p. H does not change when acids or bases are added. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

How Buffers Work Buffers work because • they resist changes in p. H from the addition of an acid or a base. • in the body, they absorb H 3 O+ or OH− from foods and cellular processes to maintain p. H. • they are important in the proper functioning of cells and blood. • they maintain a p. H close to 7. 4 in blood. A change in the p. H of the blood affects the uptake of oxygen and cellular processes. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Components of a Buffer A buffer solution • contains a combination of acid–base conjugate pairs, a weak acid and a salt of its conjugate base, such as HC 2 H 3 O 2(aq) and C 2 H 3 O 2−(aq) • has equal concentrations of a weak acid and its salt. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

How Buffers Work In the buffer with acetic acid (HC 2 H 3 O 2) and sodium acetate (Na. C 2 H 3 O 2), • the salt produces acetate ions and sodium ions. Na. C 2 H 3 O 2(aq) C 2 H 3 O 2−(aq) + Na+(aq) • the salt is added to provide a higher concentration of the conjugate base C 2 H 3 O 2− than from the weak acid alone. HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2−(aq) + H 3 O+(aq) Large amount General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Large amount © 2016 Pearson Education, Inc.

Function of a Weak Acid in a Buffer If a small amount of base is added to this same buffer solution, it is neutralized by the acetic acid, HC 2 H 3 O 2, which shifts the equilibrium in the direction of the products acetate ion and water. HC 2 H 3 O 2(aq) + OH−(aq) C 2 H 3 O 2−(aq) + H 2 O(l) Equilibrium shifts in the direction of the products. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Function of Conjugate Base in a Buffer When a small amount of acid is added, the additional H 3 O+ combines with the acetate ion, C 2 H 3 O 2−, causing the equilibrium to shift in the direction of the reactants, acetic acid and water. The acetic acid produced contributes to the available weak acid. HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2− (aq) + H 3 O+(aq) Equilibrium shifts in the direction of the reactants. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Working Buffers The buffer described here consists of about equal concentrations of acetic acid (HC 2 H 3 O 2) and its conjugate base, acetate ion (C 2 H 3 O 2−). • Adding H 3 O+ to the buffer reacts with the salt, C 2 H 3 O 2−, whereas adding OH− neutralizes the acid HC 2 H 3 O 2. • The p. H of the solution is maintained as long as the added amounts of acid or base are small compared to the concentrations of the buffer components. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating the p. H of a Buffer By rearranging the Ka expression to give [H 3 O+], we can obtain the ratio of the acetic acid/acetate buffer and calculate the p. H. Solving for H 3 O+ gives Weak acid Conjugate base Core Chemistry Skill Calculating the p. H of a Buffer General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check The Ka for acetic acid, HC 2 H 3 O 2, is 1. 8 × 10– 5. What is the p. H of a buffer prepared with 1. 0 M HC 2 H 3 O 2 and 1. 0 M C 2 H 3 O 2−? HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2−(aq) + H 3 O+(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution The Ka for acetic acid, HC 2 H 3 O 2, is 1. 8 × 10– 5. What is the p. H of a buffer prepared with 1. 0 M HC 2 H 3 O 2 and 1. 0 M C 2 H 3 O 2−? HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2−(aq) + H 3 O+(aq) STEP 1 State the given and needed quantities. ANALYZE THE PROBLEM Given [HC 2 H 3 O 2] = 1. 0 M [C 2 H 3 O 2−] = 1. 0 M Equation HC 2 H 3 O 2(aq) + H 2 O(l) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Need p. H of solution C 2 H 3 O 2−(aq) + H 3 O+(aq) © 2016 Pearson Education, Inc.

Solution The Ka for acetic acid, HC 2 H 3 O 2, is 1. 8 × 10– 5. What is the p. H of a buffer prepared with 1. 0 M HC 2 H 3 O 2 and 1. 0 M C 2 H 3 O 2−? HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2−(aq) + H 3 O+(aq) STEP 2 Write the Ka expression and rearrange for [H 3 O+]. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution The Ka for acetic acid, HC 2 H 3 O 2, is 1. 8 10– 5. What is the p. H of a buffer prepared with 1. 0 M HC 2 H 3 O 2 and 1. 0 M C 2 H 3 O 2−? HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2−(aq) + H 3 O+(aq) STEP 3 Substitute [HA] and [A−] into the Ka expression. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution The Ka for acetic acid, HC 2 H 3 O 2, is 1. 8 10– 5. What is the p. H of a buffer prepared with 1. 0 M HC 2 H 3 O 2 and 1. 0 M C 2 H 3 O 2−? HC 2 H 3 O 2(aq) + H 2 O(l) C 2 H 3 O 2−(aq) + H 3 O+(aq) STEP 4 Use [H 3 O+] to calculate p. H. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating the p. H of a Buffer Because Ka is a constant at a given temperature, • the [H 3 O+] is determined by the [HC 2 H 3 O 2]/[C 2 H 3 O 2−] ratio. • the addition of small amounts of either acid or base changes the ratio of [HC 2 H 3 O 2]/[C 2 H 3 O 2−] only slightly. • the changes in [H 3 O+] will be small and the p. H will be maintained. • the addition of a large amount of acid or base may exceed the buffering capacity of the system. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Buffers and p. H Changes Buffers can be prepared from conjugate acid–base pairs such as H 2 PO 4−/HPO 42− and HPO 42−/PO 43−, HCO 3−/CO 32−, or NH 4+/NH 3. The p. H of the buffer solution will depend on the conjugate acid–base pair chosen. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Buffers and p. H Changes Using a common phosphate buffer for biological specimens, we can look at the effect of using different ratios of [H 2 PO 4−/HPO 42−] on the [H 3 O+] and p. H. The Ka of H 2 PO 4− is 6. 2 × 10− 8. The equation and the [H 3 O+] are written as follows: H 2 PO 4−(aq) + H 2 O(l) H 3 O+(aq) + HPO 42−(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Buffers and p. H Changes General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chemistry Link to Health: Buffers in Blood Plasma The arterial blood plasma has a normal p. H of 7. 35 to 7. 45. If changes in H 3 O+ lower the p. H below 6. 8 or raise it above 8. 0, cells cannot function properly and death may result. In our cells, CO 2 • is continually produced as an end product of cellular metabolism. • is carried to the lungs for elimination, and the rest dissolves in body fluids such as plasma and saliva, forming carbonic acid, H 2 CO 3. As a weak acid, carbonic acid dissociates to give bicarbonate, HCO 3−, and H 3 O+. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson. Education, Inc. © 2016 Pearson Inc.

Chemistry Link to Health: Buffers in Blood Plasma Kidneys also supply more of the bicarbonate anion, HCO 3−, setting up an important buffer system in the body fluid: CO 2(g) + H 2 O(l) H 2 CO 3(aq) HCO 3−(aq) + H 3 O+(aq) Excess H 3 O+ entering the body fluids reacts with the HCO 3−, and excess OH− reacts with the carbonic acid. H 2 CO 3(aq) + H 2 O(l) HCO 3−(aq) + H 3 O+(aq) Equilibrium shifts in the direction of the reactants. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson. Education, Inc. © 2016 Pearson Inc.

Chemistry Link to Health: Buffers in Blood Plasma Kidneys also supply more of the bicarbonate anion, HCO 3−, setting up an important buffer system in the body fluid: CO 2(g) + H 2 O(l) H 2 CO 3(aq) HCO 3−(aq) + H 3 O+(aq) • Excess H 3 O+ entering the body fluids reacts with the HCO 3−: H 2 CO 3(aq) + H 2 O(l) HCO 3−(aq) + H 3 O+(aq) Equilibrium shifts in the direction of the reactants. • Excess OH– entering the body fluids reacts with the H 2 CO 3: H 2 CO 3(aq) + OH−(aq) H 2 O(l) + HCO 3−(aq) Equilibrium shifts in the direction of the products. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson. Education, Inc. © 2016 Pearson Inc.

Chemistry Link to Health: Buffers in Blood Plasma To maintain the normal blood plasma p. H (7. 35 to 7. 45), • the ratio of [H 2 CO 3]/[HCO 3−] needs to be about 1 to 10. • concentrations of 0. 0024 M H 2 CO 3 and 0. 024 M HCO 3− work to maintain that p. H. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson. Education, Inc. © 2016 Pearson Inc.

Chemistry Link to Health: Buffers in Blood Plasma In the body, the concentration of carbonic acid is closely associated with the partial pressure of CO 2, PCO 2. • If the CO 2 level rises, increasing H 2 CO 3, the equilibrium shifts to produce more H 3 O+, which lowers the p. H. This condition is called acidosis. • A lowering of the CO 2 level leads to a high blood p. H, a condition called alkalosis. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson. Education, Inc. © 2016 Pearson Inc.
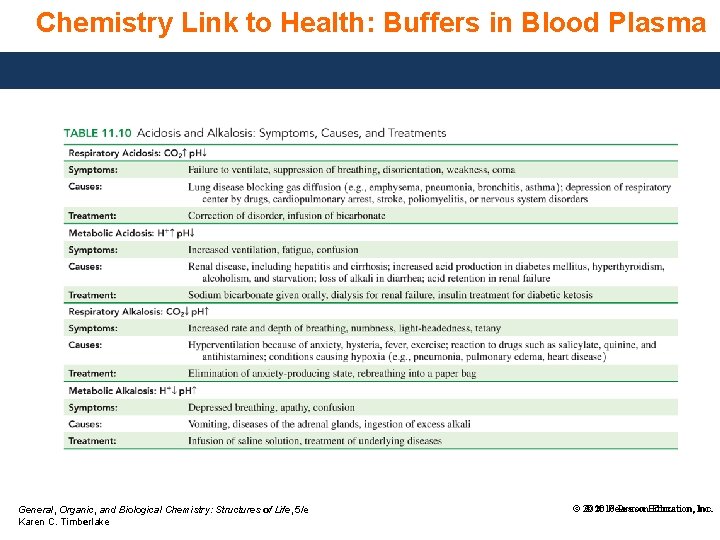
Chemistry Link to Health: Buffers in Blood Plasma General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson. Education, Inc. © 2016 Pearson Inc.
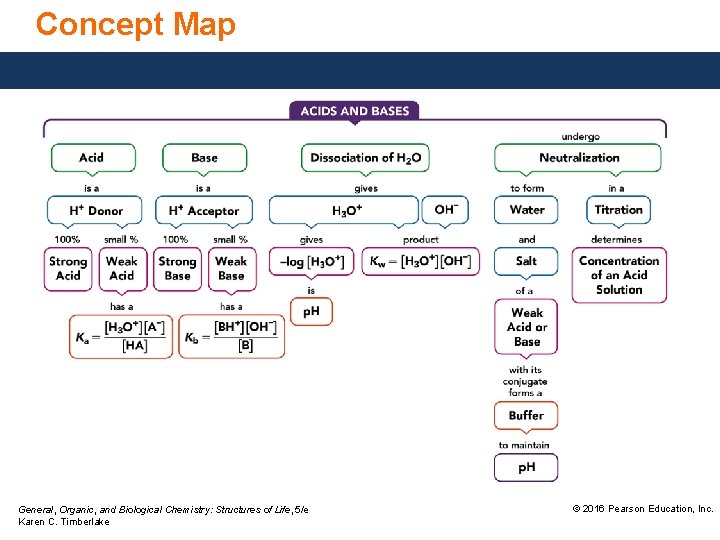
Concept Map General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 25