11 5 Dissociation of Water The equilibrium reached
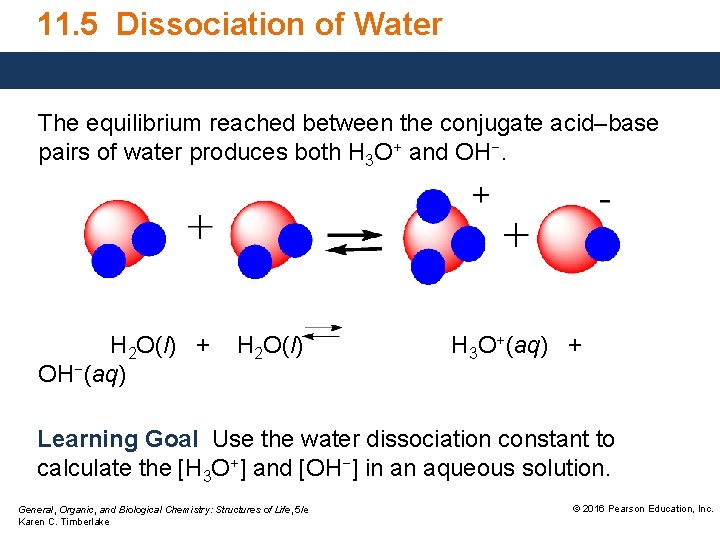
11. 5 Dissociation of Water The equilibrium reached between the conjugate acid–base pairs of water produces both H 3 O+ and OH−. H 2 O(l) + OH−(aq) H 2 O(l) H 3 O+(aq) + Learning Goal Use the water dissociation constant to calculate the [H 3 O+] and [OH−] in an aqueous solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
Dissociation Constant of Water, Kw Water is amphoteric—it can act as an acid or a base. In water, • H+ is transferred from one H 2 O molecule to another. • one water molecule acts as an acid, while another acts as a base. • equilibrium is reached between the conjugate acid–base pairs. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Writing the Dissociation Constant, Kw In the equation for the dissociation of water, there is both a forward and a reverse reaction. H 2 O(l) + H 2 O(l) Base Acid H 3 O+(aq) + OH−(aq) Conjugate acid Conjugate base • In pure water, the concentrations of H 3 O+ and OH− at 25 °C are each 1. 0 10− 7 M. Kw [H 3 O+] = [OH−] = 1. 0 10– 7 M Kw = [H 3 O+] [OH−] = (1. 0 10− 7 M) = 1. 0 10– 14 at 25 °C General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Dissociation Constant, Kw The ion product constant for water, Kw, is defined as • the product of the concentrations of H 3 O+ and OH−. • equal to 1. 0 10− 14 at 25 °C (the concentration units are omitted). When • [H 3 O+] and [OH−] are equal, the solution is neutral. • [H 3 O+] is greater than the [OH−], the solution is acidic. • [OH−] is greater than the [H 3 O+], the solution is basic. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Using Kw to Calculate [H 3 O+] and [OH−] • If we know the Using Kw to Calculate [H 3 O+] and [OH−] • If we know the](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-5.jpg)
Using Kw to Calculate [H 3 O+] and [OH−] • If we know the [H 3 O+] of a solution, we can use the Kw to calculate the [OH−]. • If we know the [OH−] of a solution, we can use the Kw to calculate the [H 3 O+]. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
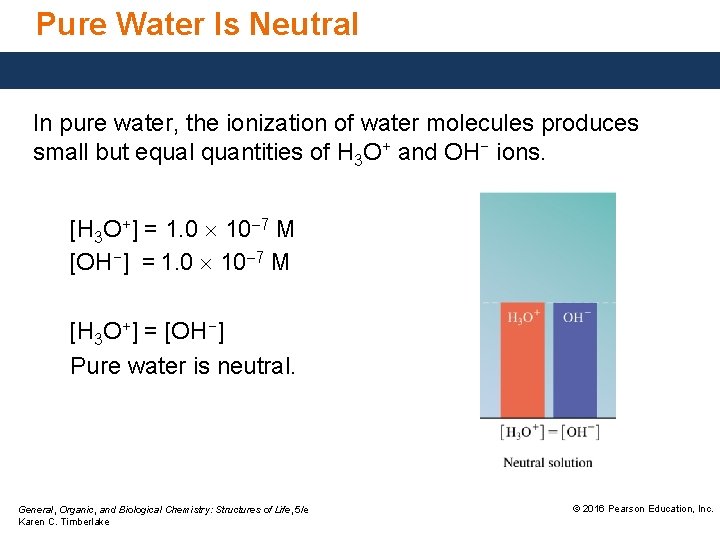
Pure Water Is Neutral In pure water, the ionization of water molecules produces small but equal quantities of H 3 O+ and OH− ions. [H 3 O+] = 1. 0 10 7 M [OH−] = 1. 0 10 7 M [H 3 O+] = [OH−] Pure water is neutral. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Acidic Solutions Adding an acid to pure water • increases the [H 3 O+]. Acidic Solutions Adding an acid to pure water • increases the [H 3 O+].](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-7.jpg)
Acidic Solutions Adding an acid to pure water • increases the [H 3 O+]. • causes the [H 3 O+] to exceed 1. 0 10− 7 M. • decreases the [OH−]. [H 3 O+] > [OH−] The solution is acidic. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Basic Solutions Adding a base to pure water • increases the [OH−] • causes Basic Solutions Adding a base to pure water • increases the [OH−] • causes](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-8.jpg)
Basic Solutions Adding a base to pure water • increases the [OH−] • causes the [OH−] to exceed 1. 0 10− 7 M • decreases the [H 3 O+] < [OH−] The solution is basic. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Comparison of [H 3 O+] and [OH−] General, Organic, and Biological Chemistry: Structures of Comparison of [H 3 O+] and [OH−] General, Organic, and Biological Chemistry: Structures of](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-9.jpg)
Comparison of [H 3 O+] and [OH−] General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Neutral, Basic, and Acidic Solutions Core Chemistry Skill Calculating [H 3 O+] and [OH−] Neutral, Basic, and Acidic Solutions Core Chemistry Skill Calculating [H 3 O+] and [OH−]](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-10.jpg)
Neutral, Basic, and Acidic Solutions Core Chemistry Skill Calculating [H 3 O+] and [OH−] in Solutions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Guide to Calculating [H 3 O+] and [O–] in Aqueous Solutions General, Organic, and Guide to Calculating [H 3 O+] and [O–] in Aqueous Solutions General, Organic, and](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-11.jpg)
Guide to Calculating [H 3 O+] and [O–] in Aqueous Solutions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Calculating [H 3 O+] What is the [H 3 O+] of a solution if Calculating [H 3 O+] What is the [H 3 O+] of a solution if](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-12.jpg)
Calculating [H 3 O+] What is the [H 3 O+] of a solution if [OH−] is 5. 0 × 10− 8 M? STEP 1 State the given and needed quantities. ANALYZE Given Need THE [OH−] = 5. 0 × 10− 8 M [H 3 O+] PROBLEM Know Kw = [H 3 O+][OH−] = 1. 0 × 10− 14 STEP 2 Write the Kw for water and solve for the unknown [H 3 O+]. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Calculating [H 3 O+] What is the [H 3 O+] of a solution if Calculating [H 3 O+] What is the [H 3 O+] of a solution if](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-13.jpg)
Calculating [H 3 O+] What is the [H 3 O+] of a solution if [OH−] is 5. 0 × 10− 8 M? STEP 3 Substitute in the known [H 3 O+] or [OH−] and calculate. Because the [H 3 O+] of 2. 0 × 10– 7 M is larger than the [OH−] of 5. 0 × 10– 8 M, the solution is acidic. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Study Check If lemon juice has [H 3 O+] of 2. 0 × 10– Study Check If lemon juice has [H 3 O+] of 2. 0 × 10–](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-14.jpg)
Study Check If lemon juice has [H 3 O+] of 2. 0 × 10– 3 M, what is the [OH−] of the solution? A. 2. 0 × 10− 11 M B. 5. 0 × 10− 11 M C. 5. 0 × 10− 12 M General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Solution If lemon juice has [H 3 O+] of 2. 0 × 10− 3 Solution If lemon juice has [H 3 O+] of 2. 0 × 10− 3](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-15.jpg)
Solution If lemon juice has [H 3 O+] of 2. 0 × 10− 3 M, what is the [OH−] of the solution? STEP 1 State the given and needed quantities. ANALYZE Given Need Know THE [H 3 O+] = 2. 0 × 10− 3 M [OH−] Kw = [H 3 O+][OH−] PROBLEM = 1. 0 × 10− 14 STEP 2 Write the Kw for water and solve for the unknown [H 3 O+] or [OH−]. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Solution If lemon juice has [H 3 O+] of 2. 0 × 10− 3 Solution If lemon juice has [H 3 O+] of 2. 0 × 10− 3](http://slidetodoc.com/presentation_image_h2/0e7986fd671b25c676e889ce831af353/image-16.jpg)
Solution If lemon juice has [H 3 O+] of 2. 0 × 10− 3 M, what is the [OH−] of the solution? STEP 3 Substitute in the known [H 3 O+] or [OH−] and calculate. Because the [H 3 O+] concentration of 2. 0 × 10− 3 M is greater than the [OH−] of 5. 0 × 10− 12 M, the solution is acidic. The answer is C. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 16