101618 OBJECTIVE YOU WILL CONDUCT INQUIRY 2 1

10/16/18 OBJECTIVE: YOU WILL CONDUCT INQUIRY 2. 1, “MEASURING THE MASS AND VOLUME OF WATER” 1. COPY HW: WRITE A LOL (LINE OF LEARNING) ABOUT FINDING THE DENSITY OF WATER 2. PICK UP YOUR STC BOOK AND READ PROCEDURES 1 -3 P. 21 WITH YOUR LAB TEAM BE PREPARED TO DISCUSS

MATERIALS NEEDED FOR INQUIRY 2. 1 1 100 ML PLASTIC GRADUATED CYLINDER 1 PLASTIC FUNNEL 1 PLASTIC PIPETTE 1 ELECTRONIC BALANCE 1 200 ML GLASS BEAKER (FILL THE BEAKER WITH 100 ML OF TAP WATER. ) LAB PROCEDURE SHEETS (GLUED IN SNB)
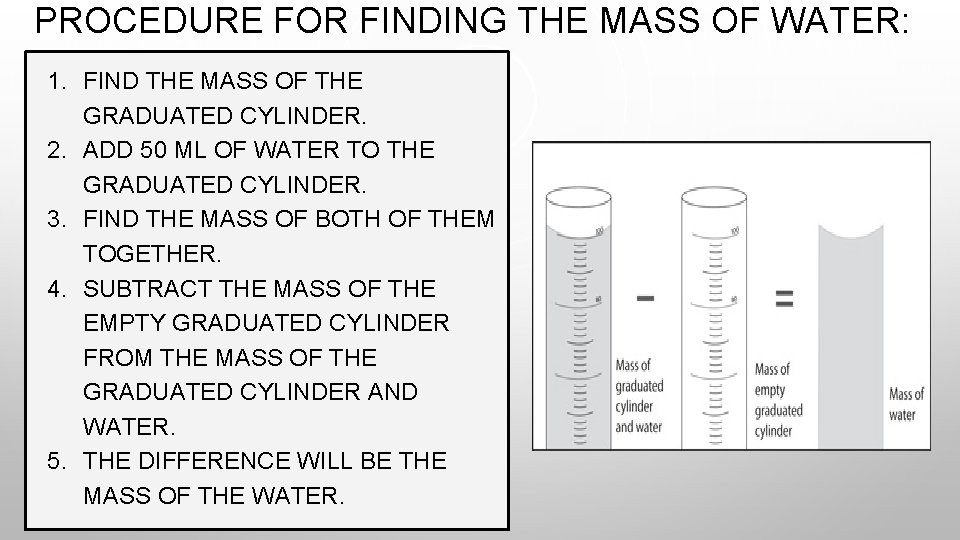
PROCEDURE FOR FINDING THE MASS OF WATER: 1. FIND THE MASS OF THE GRADUATED CYLINDER. 2. ADD 50 ML OF WATER TO THE GRADUATED CYLINDER. 3. FIND THE MASS OF BOTH OF THEM TOGETHER. 4. SUBTRACT THE MASS OF THE EMPTY GRADUATED CYLINDER FROM THE MASS OF THE GRADUATED CYLINDER AND WATER. 5. THE DIFFERENCE WILL BE THE MASS OF THE WATER.

- = MASS OF WATER

COPY ONTO #1 OF YOUR 2. 1 RECORDING SHEET: STEPS FOR FINDING THE MASS OF WATER A. SET THE SCALE TO ZERO & MAKE SURE THE UNIT IS GRAMS B. MEASURE THE MASS OF THE GRADUATED CYLINDER C. PUT EXACTLY 25/50 ML OF WATER INTO THE GRADUATED CYLINDER D. FIND THE MASS OF THE GRADUATED CYLINDER PLUS WATER

DO NOW: • COPY YOUR HW AND UPDATE YOUR TOC YOU WILL GRAPH THE RESULTS OF INQUIRY 2. 1 TO VISUALLY REPRESENT THE DENSITY OF WATER HW: DENSITY PROBLEMS IN SNB • GLUE THE TWO SHEETS FROM THE DO NOW BASKET ONTO YOUR INPUT PAGE FACING YESTERDAY’S INQUIRY • GLUE THE HW ON THE NEXT OUTPUT PAGE • MAKE SURE EVERYONE ON YOUR TEAM HAS ALL OF THE DATA COMPLETED FROM INQUIRY 2. 1 DISCUSS: WHAT IS THE DENSITY OF WATER IF YOU ROUND YOUR ANSWER TO THE NEAREST WHOLE NUMBER?

SNB SET UP

EXTENDING OUR FINDINGS
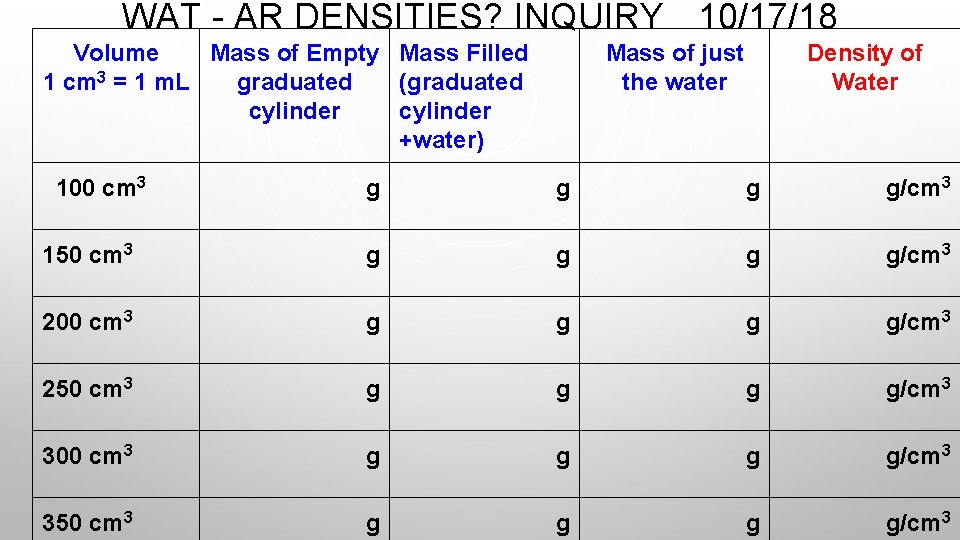
WAT - AR DENSITIES? INQUIRY 10/17/18 Volume Mass of Empty Mass Filled 1 cm 3 = 1 m. L graduated (graduated cylinder +water) 100 cm 3 Mass of just the water Density of Water g g/cm 3 150 cm 3 g g/cm 3 200 cm 3 g g/cm 3 250 cm 3 g g/cm 3 300 cm 3 g g/cm 3 350 cm 3 g g/cm 3

EVIDENCE OF LEARNING: INQUIRY 2. 1 ● SOLIDS AND LIQUIDS HAVE MASS AND VOLUME. ● VOLUME IS THE SPACE MATTER TAKES UP AND IS MEASURED IN MILLILITERS (ML) OR CUBIC CENTIMETERS (CM 3). ● MASS IS THE AMOUNT OF MATTER IN AN OBJECT AND IS MEASURED IN GRAMS OR KILOGRAMS USING A BALANCE. ● DENSITY THE RATIO OF MASS TO VOLUME (MASS: VOLUME OR MASS/VOLUME) IS 1: 1 OR 1 TO 1. ○ THE DENSITY OF WATER IS 1. 0 G/CM 3.

1. Complete Density problems 1 -3 on your input page 2. Remember to enter the value for mass first in your calculator.

“I DENSITY”

DO NOW, CONTINUED • GLUE STUDENT SHEET 2. 2 UNDER YOUR OBJECTIVE • OPEN YOUR STC BOOK TO PAGE 23 • READ & DISCUSS PROCEDURES 1 & 2 WITH YOUR TABLE GROUP. • BE PREPARED TO SHARE YOUR

WHAT IS GRAVITY? HTTPS: //WWW. YOUTUBE. COM/WATCH? LIST=PL 908547 EAA 7 E 4 AE 74&V=P_O 4 AY 7 XKXG

EVIDENCE OF LEARNING: INQUIRY 2. 1 CREATING A LINE GRAPH • LABEL THE X AXIS (HORIZONTAL) • LABEL THE Y AXIS (VERTICAL) • PLOT THE DATA • CONNECT THE POINTS TO FORM THE LINE

HOMEWORK FOR INQUIRY 2. 3 1. QUIZ SIGNED 2. REVIEW NOTES ON FINDING DENSITY OF WATER AND OF REGULAR & IRREGULAR OBJECTS- PRACTICAL LAB MOVED TO MONDAY, 10/22/18


Do NOW: 1 - Copy HW and Objective 2 - List the steps to find the density of an irregular object under your objective. Hint: think about Archimedes! Find the mass of the object. Measure a specific volume of water. Add the object to the water & record the amount of water displaced- this is the volume of the object. Divide the mass by the volume to find the


INQUIRY 2. 3 Use the boxes on student sheet 2. 3 to create a set of labeled diagrams to visually represent how to find the densities of the irregular objects in your bin.

Do NOW: 1 - Copy HW: Review notes on finding the density of a regular object, irregular object, and water- practical lab Monday 2 - Update TOC- Inquiry 2. 3 Finding Density of irregular objects 3 - Glue the blank table on the INPUT page facing your visual procedures.
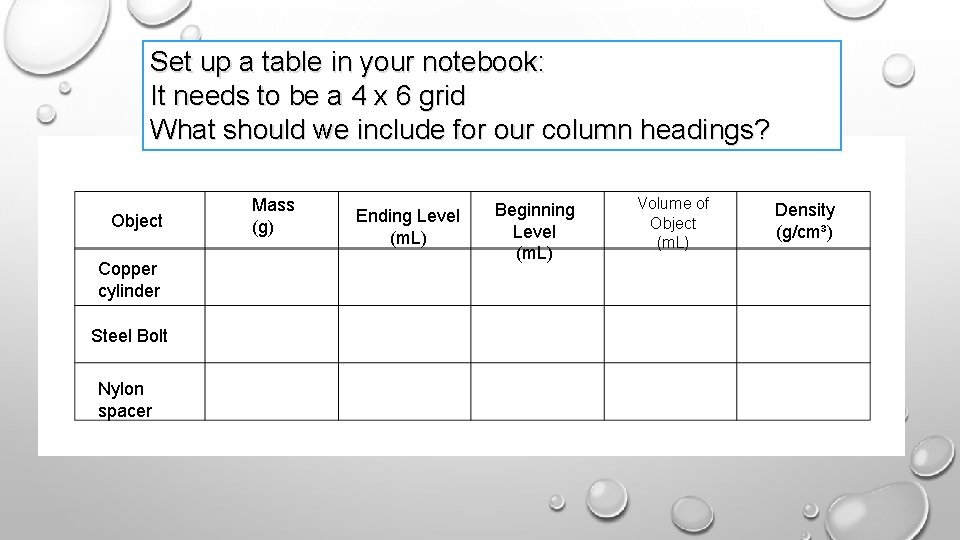
Set up a table in your notebook: It needs to be a 4 x 6 grid What should we include for our column headings? Object Copper cylinder Steel Bolt Nylon spacer Mass (g) Ending Level (m. L) Beginning Level (m. L) Volume of Object (m. L) Density (g/cm³)

HOW TO ADD AN OBJECT WITHOUT SPLASHING OR BREAKING THE GRADUATED CYLINDER

CLOSURE: MEASURING DENSITIES OF IRREGULAR OBJECTS USE YOUR DATA TABLE TO ANSWER QUESTIONS 3 & 4 ON THE EXIT TICKET USE THE DATA FROM YOUR INQUIRY TO SUPPORT YOUR
- Slides: 24