100 100 200 200 300 300 400 400
































![What does the following symbol stand for? [H 3 + O ] What does the following symbol stand for? [H 3 + O ]](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-34.jpg)
![[H 3 + O ] Hydronium Ion Concentration [H 3 + O ] Hydronium Ion Concentration](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-35.jpg)
![What does the following symbol stand for? [OH ] What does the following symbol stand for? [OH ]](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-36.jpg)
![[OH ] Hydroxide Ion Concentration [OH ] Hydroxide Ion Concentration](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-37.jpg)


































- Slides: 72









$100 $100 $200 $200 $300 $300 $400 $400 $500 $500

What makes water an effective solvent?

Its Polar Nature

What characteristic of water remains the same no matter what is dissolved in it?

The ratio of hydrogen to oxygen

Hard water has a p. H higher than 7 and an abundance of calcium and magnesium salts. What would be the best cleaning solution for removing hard-water residue from drinking glasses?

A mild acid such as vinegar

Explain in detail what makes a water molecule polar. Double the score if correct!

Oxygen has greater electronegativity so it doesn’t share electrons equally, oxygen keeps the electrons most of the time

The picture shows the results of pouring a blue liquid into a clear liquid and allowing the mixture to settle for 25 minutes. Why did the solution end up looking like this?

2 Liquids are immiscible and the blue one has a higher density

What is an electrolyte?

A solution that conducts electricity.

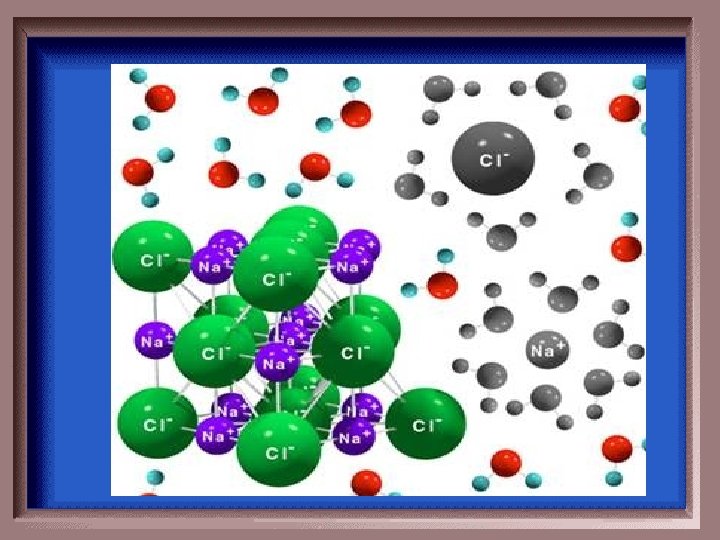
What process is depicted in the following picture?

Dissociation

Why does dissolving salt in water increase the conductivity of the solution?

concentration of ions in the solution increases

Why will bathwater normally have electrolytic behaviors even though distilled water does not?

Bathwater generally contains dissolved minerals

Why doesn’t pure water conduct electricity?

No Ions or minerals are dissolved in it

A true solution is a homogeneous mixture and it has to also be _________

Colorless or transparent

What will be the p. H of a solution having an equal number of H+ ions and OH– ions?

p. H = 7
![What does the following symbol stand for H 3 O What does the following symbol stand for? [H 3 + O ]](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-34.jpg)
What does the following symbol stand for? [H 3 + O ]
![H 3 O Hydronium Ion Concentration [H 3 + O ] Hydronium Ion Concentration](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-35.jpg)
[H 3 + O ] Hydronium Ion Concentration
![What does the following symbol stand for OH What does the following symbol stand for? [OH ]](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-36.jpg)
What does the following symbol stand for? [OH ]
![OH Hydroxide Ion Concentration [OH ] Hydroxide Ion Concentration](https://slidetodoc.com/presentation_image_h/fa6110a26dbbdc546c692933117f28ba/image-37.jpg)
[OH ] Hydroxide Ion Concentration

Write down the 3 factors that influence the RATE of dissolving?

1. Temperature 2. Agitation / Stirring 3. Surface Area of Particles

Which choice has the most surface area? A or B

Choice B

Which of the following choices will dissolve more salt? 50˚ 100˚

For solids, as temperature increases solubility increases.

What can you do to liquid soda to get it to dissolve more carbonated gas (CO 2)?

Lower the Temperature

What does it mean to be miscible?

That substances are able to mix together

What are the 3 factors affecting solubility?

1. Nature of the Solvent 2. Pressure (gases only) 3. Temperature

Why would swimmers say that they can float more easily in the ocean than in a freshwater pond?

density of ocean water is higher than that of pond water

A block of maple wood with a volume of 405 cubic centimeters and a density of 0. 67 g/cm 3 is sawed in half. What is the density of one of the pieces?

The same: 0. 67 g/cm 3 Remember water always has a density of 1 g/cm 3 regardless whether you have a cup of water or a swimming pool full of water

Water normally boils at 100°C at sea level and at 97°C on a North American mountaintop. Why?

Difference in air pressure

Students in a chemistry lab measure the time it takes four different 100 m. L solutions to pass through a hole in the bottom of a cup. Which of the following properties of the solutions is most likely being measured? A Buoyancy B Mass C Viscosity D Volume

C. Viscosity


Why are Bronsted. Lowry acids called proton donors?

+ H is the same thing as a proton.

What is a solvent?

Part of a solution that does the dissolving

What is a solute?

Part of a solution that gets dissolved in a solution by the solvent

Two clear solutions are placed in separate beakers. The first solution has a p. H of 4, and the p. H of the second solution is unknown. If the two solutions are mixed and the resulting p. H is 5, what can be said about the second solution?

Second solution had a higher concentration of OH – ions

Water molecules generally have which effect on a soluble ionic compound mixed into water?

They break the bonds between the ions.

A student added a small amount of sodium carbonate solution (Na 2 CO 3) to a test tube containing calcium chloride (Ca. Cl 2) solution. A white solid immediately appeared. What conclusion can be made from this experiment?

At least one product of the reaction is insoluble.

How can you create your own Solubility curve for Sugar in water?

Find the amount of solute required for a solution to become saturated at various different temperatures