10 4 NOTES Boyles and Charles Law Complete

10. 4 – NOTES Boyle’s and Charles Law

• Complete the questions at the beginning of the notes with your partner!

• Explain the following observations: • Even if they have ample supplies of oxygen gas, airplane passengers experience discomfort when the cabin undergoes a drop in air pressure. • When air pressure decreases, gases in the lungs, stomach and sinuses expand, creating discomfort. • New tennis balls are sold in pressurized containers. • Tennis ball have an internal pressure greater than atmospheric pressure to give bounce. Cans create the same pressure so gas doesn’t leak out. • After descending from a high mountain, the capped, half-filled plastic water bottle from which your drank while standing at the summit now appears dented or slightly crushed. • Pressure outside the bottle increases as you descend, crushing the bottle.

• Pressure-Volume Behavior of Gases • If you increase pressure, the volume decreases • • Boyle’s Law • • P 1 V 1=P 2 V 2 • • Inversely proportional
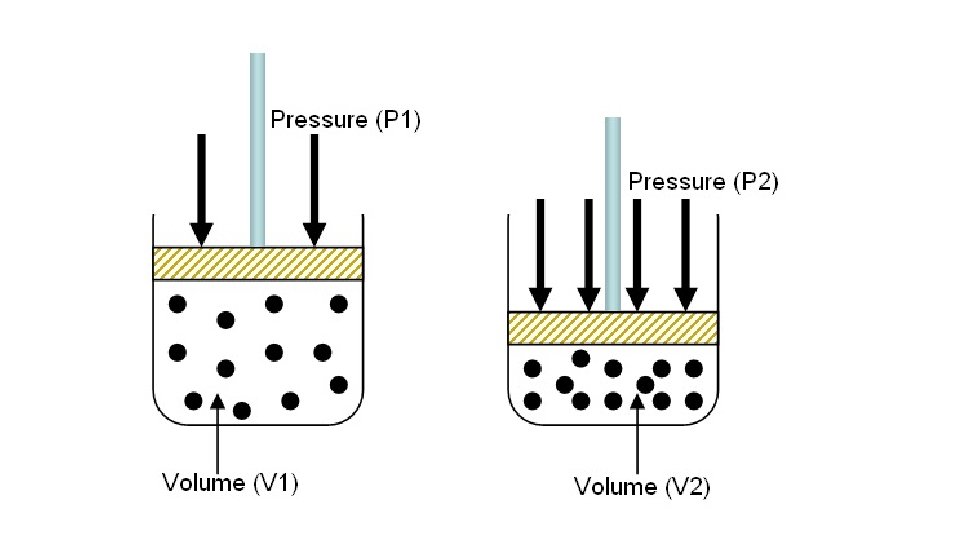

• Example: A sample of oxygen gas occupies a volume of 250 ml at 1 atm of pressure. If the pressure increases to 1. 2 atm, what is the new volume?

• Exploring Temperature-Volume Relationships • Matter expands when heated • • V 1/T 1 = V 2/T 2 or V 1 T 2 = V 2 T 1 •

• Kelvin temperature scale • SI unit of temperature • • The advantage of the Kelvin scale is that it has no value for 0 degrees, and there are no negative numbers. • • Must be used when doing any calculation with temperature • • Absolute zero • Lowest temperature theoretically possible • 0 K = -273°C • K = °C +273 • Molecular Motion Stops here

• STP – standard temperature and pressure • • 1 atm and 0°C • You will need to convert the temperature from Celsius to Kelvin if these conditions are used in the a problem. • • 273 K = standard temperature in equations • • 1 atm = 101. 3 k. Pa = 760 mm. Hg •

• Temperature-Volume Behavior of Gases • As temperature increases, the volume will increase when the pressure is constant • Linear relationship/directly proportional • • Charles’ law • V 1/T 1 = V 2/T 2 or V 1 T 2 = V 2 T 1 • • Volume and Temperature change when pressure is constant • • THE TEMPERATURE MUST BE IN KELVINS
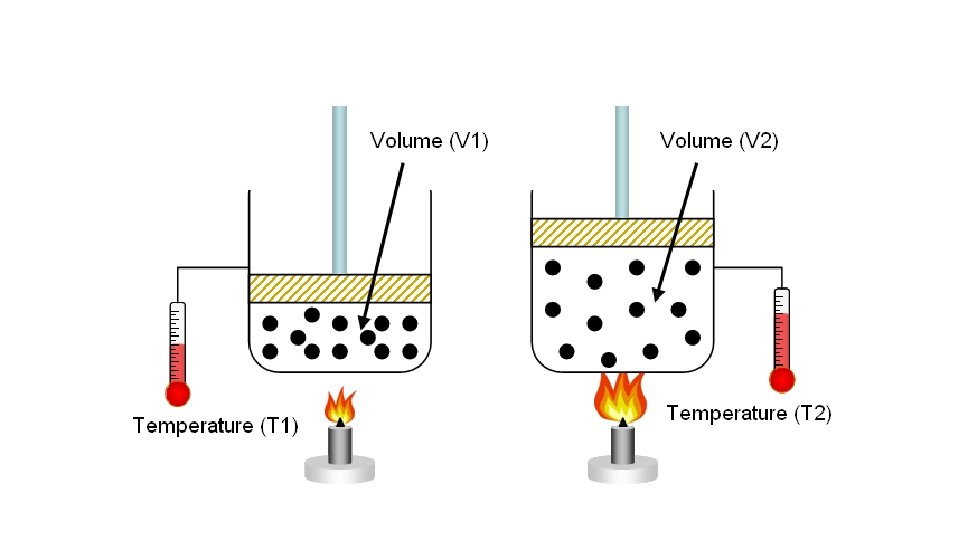

• Example: If helium occupies a volume of 3. 8 L at 228 K, what volume will it occupy at 318 K? •

• Example: A sample of chlorine occupies a volume of 2 L at 25 C. What will the temperature be if the volume expands to 4 L?
- Slides: 13