10 4 Carboxylic Acids and Esters Carboxylic Acids

10. 4 Carboxylic Acids and Esters

Carboxylic Acids • These are compounds with a carboxyl group, which consists of a carbonyl group and a hydroxyl group (an OH group like in alcohols) • The general structural formula is: O II R - C - OH • this can be written: R-COOH

Carboxylic Acids • They are weak acids due to their slight ionization in water to release a hydrogen ion. O II R - C - OH(aq) O II R - C - O-(aq) + H+(aq)

Naming Carboxylic Acids • In order to name these compounds: • First, identify the longest parent chain with the carboxyl group. • Then drop the -e ending and add the ending oic and the word acid.

The first three Carboxylic Acids • methanoic acid O II H - C - OH O II CH 3 - C - OH propanoic acid O II CH 3 – CH 2 - C - OH

Polycarboxylic Acids • You do not have to memorize these names and condensed formulas, all you must recognize is that these are carboxylic acids.

Properties of Carboxylic Acids • Like other organic derivatives many of these compounds have common names, derived from the origin of the acid. • Carboxylic acids are widely distributed in nature. • They are present in all cells and are a component of fats. • Fatty acids are carboxylic acids in fact.

Properties of Carboxylic Acids • The common name for methanoic acid is formic acid since it is found in ants (the Latin for ant is formica). • For ethanoic acid the common name is acetic acid, from acetum (Latin for sour wine, as in vinegar).

Properties of Carboxylic Acids • Short chain carboxylic acids are volatile liquids with sharp, unpleasant odours such as sweaty feet, vomit and rancid butter. • Tracker dogs follow the trail of carboxylic acid smell from the body odour of the subject. • The polar nature of the carboxyl group allows these organic acids to be misable with water. • Larger chain organic acids are waxy solids that are insoluble in water, but will dissolve in solvents such as ethanol or propanone.

Esters • A compound called an ester is composed of an alcohol and a carboxylic acid. • The process to create an ester is simply called esterfication.
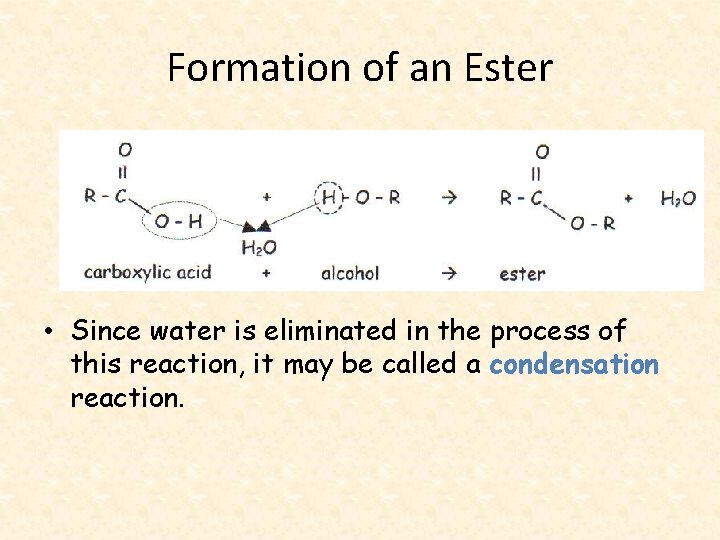
Formation of an Ester • Since water is eliminated in the process of this reaction, it may be called a condensation reaction.

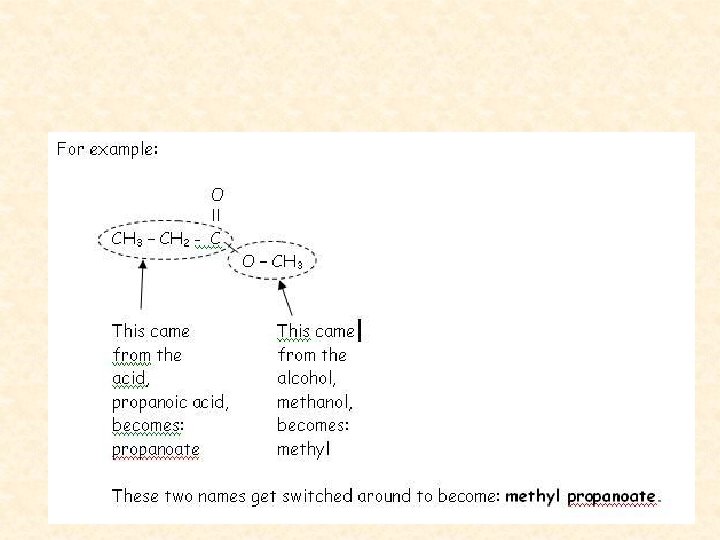
Naming Esters • We name esters by identifying the alcohol chain, dropping the -ol, and adding –yl. • Then we name the acid by dropping the ic acid and adding -ate.
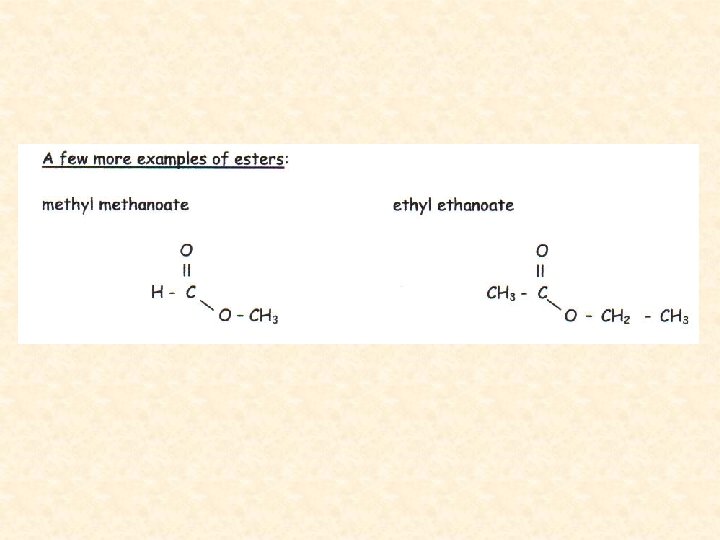

Properties and Occurrence of Esters • Lower molecular weight esters are colourless, volatile liquids that are soluble in water. • Higher molecular weight examples are colourless, waxy solids that are insoluble in water. • All esters have characteristic strong “fruity” smells and are soluble in non-polar organic solvents. • Esters are themselves good organic solvents.

Properties and Occurrence of Esters • Esters break down into their respective organic acid and alcohol from which they are formed. • This process is called hydrolysis. • When sodium hydroxide is added to an ester, for example to ethyl ethanoate, a salt, sodium ethanoate is formed along with ethanol.

Properties and Occurrence of Esters • The reaction is shown below: • Hydrolysis of an ester with an alkaline solution like sodium hydroxide is known as saponification. • This reaction is used in the preparation of soaps from fats, which are esters of glycerol and fatty acids.

Properties and Occurrence of Esters • Esters are found widely in nature as the flavour and odors of flowers, fruits and nuts. • They are manufactured as artificial perfumes or scents and artificial food flavours that are added in many edible items like ice creams, soft drinks, sweets, etc. • In industry, esters are used as solvents for making cellulose, fats, paints and varnishes and in the pharmaceutical industries. • Esters are also used as softeners in plastic and molding industries.

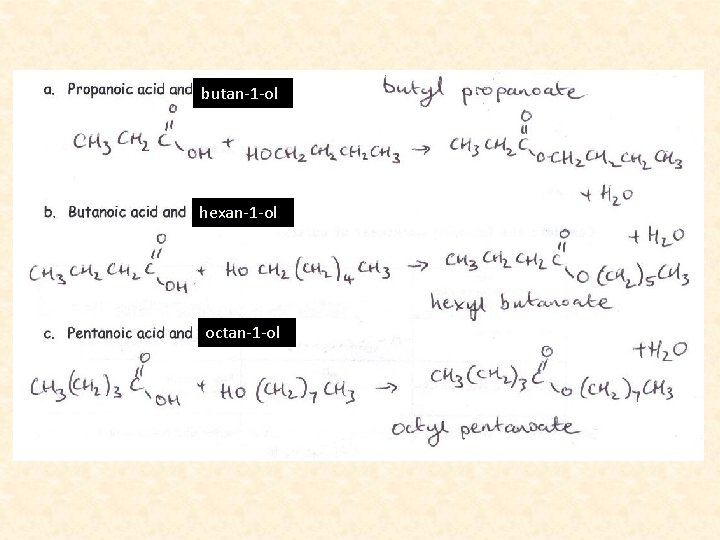
butan-1 -ol hexan-1 -ol octan-1 -ol
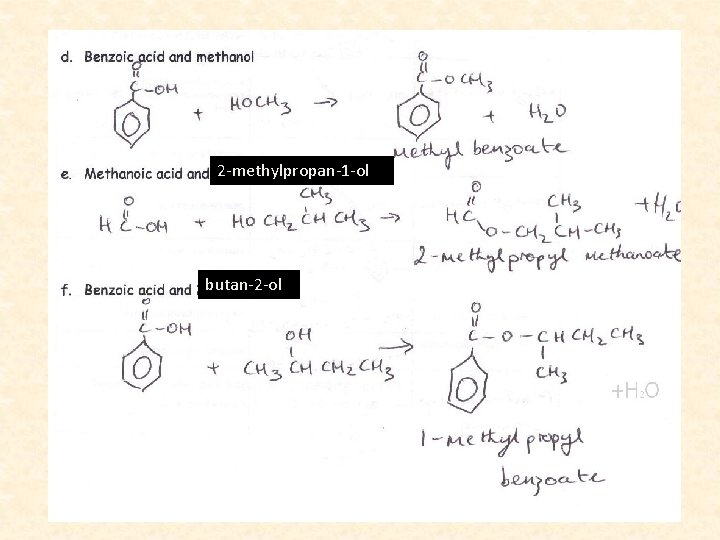
2 -methylpropan-1 -ol butan-2 -ol
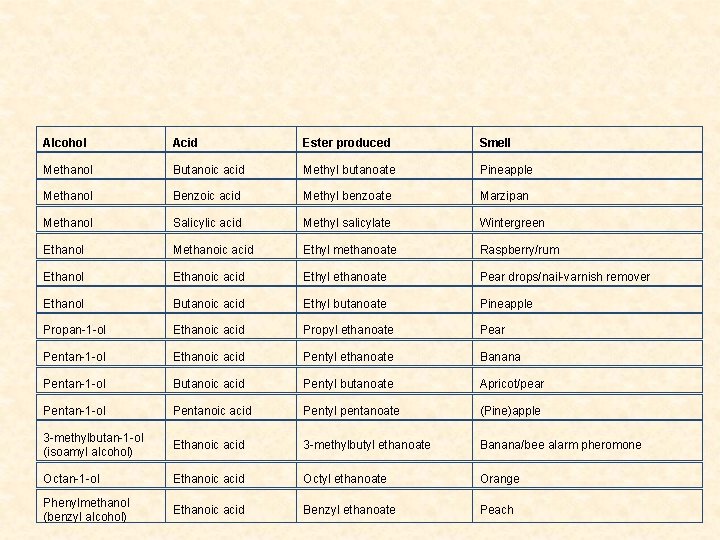
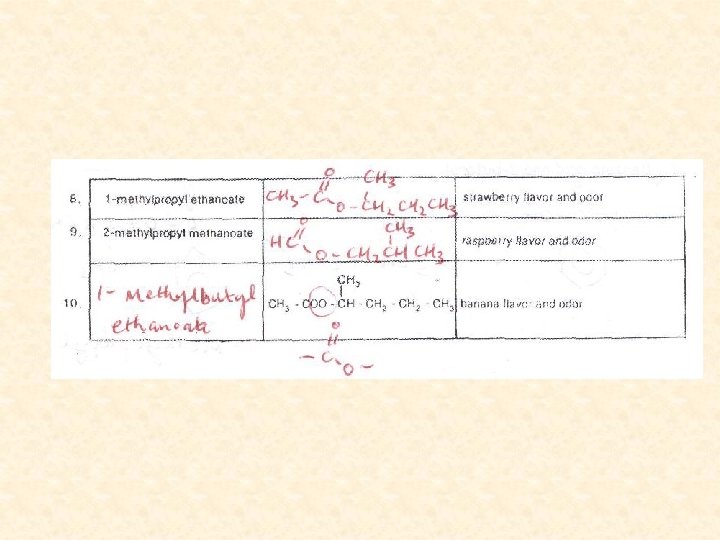
Alcohol Acid Ester produced Smell Methanol Butanoic acid Methyl butanoate Pineapple Methanol Benzoic acid Methyl benzoate Marzipan Methanol Salicylic acid Methyl salicylate Wintergreen Ethanol Methanoic acid Ethyl methanoate Raspberry/rum Ethanol Ethanoic acid Ethyl ethanoate Pear drops/nail-varnish remover Ethanol Butanoic acid Ethyl butanoate Pineapple Propan-1 -ol Ethanoic acid Propyl ethanoate Pear Pentan-1 -ol Ethanoic acid Pentyl ethanoate Banana Pentan-1 -ol Butanoic acid Pentyl butanoate Apricot/pear Pentan-1 -ol Pentanoic acid Pentyl pentanoate (Pine)apple 3 -methylbutan-1 -ol (isoamyl alcohol) Ethanoic acid 3 -methylbutyl ethanoate Banana/bee alarm pheromone Octan-1 -ol Ethanoic acid Octyl ethanoate Orange Phenylmethanol (benzyl alcohol) Ethanoic acid Benzyl ethanoate Peach
- Slides: 23