10 1 Introduction A Isotopes atomic number Z

10. 1 Introduction A. Isotopes atomic number (Z) mass number (A) atomic number (Z) = the number of protons + the number of neutrons 12 6 C number of protons number of neutrons 6 12 – 6 = 6 1
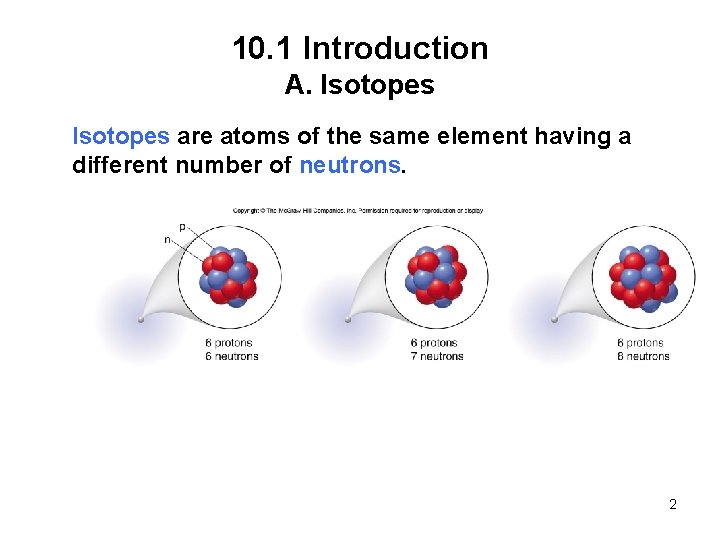
10. 1 Introduction A. Isotopes are atoms of the same element having a different number of neutrons. 2

10. 1 Introduction A. Isotopes • A radioactive isotope, called a radioisotope, is unstable and spontaneously emits energy to form a more stable nucleus. • Radioactivity is the nuclear radiation emitted by a radioactive isotope. • Of the known isotopes of all elements, 264 are stable and 300 are naturally occurring but unstable. • An even larger number of radioactive isotopes called artificial isotopes, have been produced in the laboratory. 3

10. 1 Introduction B. Types of Radiation • Types of radiation: alpha particles, beta particles, positrons, and gamma radiation. • An alpha (α) particle is a high-energy particle that contains 2 protons and 2 neutrons. • It has a +2 charge and a mass number of 4. alpha particle: 4

10. 1 Introduction B. Types of Radiation • A beta (β) particle is a high-energy electron. • It has a − 1 charge and a negligible mass compared to a proton. beta particle: • A β particle is formed when a neutron (n) is converted to a proton (p) and an electron (e). 5

10. 1 Introduction B. Types of Radiation • A positron is called an antiparticle of a β particle. • Their charges are opposite, but their masses are the same (i. e. , effectively zero). • A positron has a +1 charge and is called a “positive electron. ” positron: • A positron is formed when a proton is converted to a neutron. 6

10. 1 Introduction B. Types of Radiation • Gamma rays are high-energy radiation released from a radioactive nucleus. • They are a form of energy, so they have no mass and no charge. gamma ray: 7

10. 1 Introduction B. Types of Radiation 8

What is the difference between an α particle and a helium atom? Identify Q in each of the following symbols: a) 0 b) 4 c) 0 -1 Q 2 Q +1 Q 9

10. 2 Nuclear Reactions Radioactive decay is the process by which an unstable radioactive nucleus emits radiation. A nuclear equation can be written for this process: original nucleus new nucleus + radiation emitted The following must be equal on both sides of a nuclear equation : • The sum of the mass numbers (A) • The sum of the atomic numbers (Z) 10

10. 2 Nuclear Reactions A. Alpha Emission Alpha emission is the decay of a nucleus by emitting an a particle. 11

10. 2 Nuclear Reactions A. Alpha Emission HOW TO Balance an Equation for a Nuclear Reaction Example Write a balanced nuclear equation showing how americium-241 decays to form an a particle. Step [1] Write an incomplete equation with the original nucleus on the left and the particle emitted on the right. 12

10. 2 Nuclear Reactions A. Alpha Emission HOW TO Balance an Equation for a Nuclear Reaction Step [2] Calculate the mass number and atomic number of the newly formed nucleus on the right. Step [3] Use the atomic number to identify the new nucleus and complete the equation. 13

Write a balanced equation showing how each nucleus decays to form a new element after releasing an α particle a. Polonium-218 b. Thorium-230 c. 252 Es 14
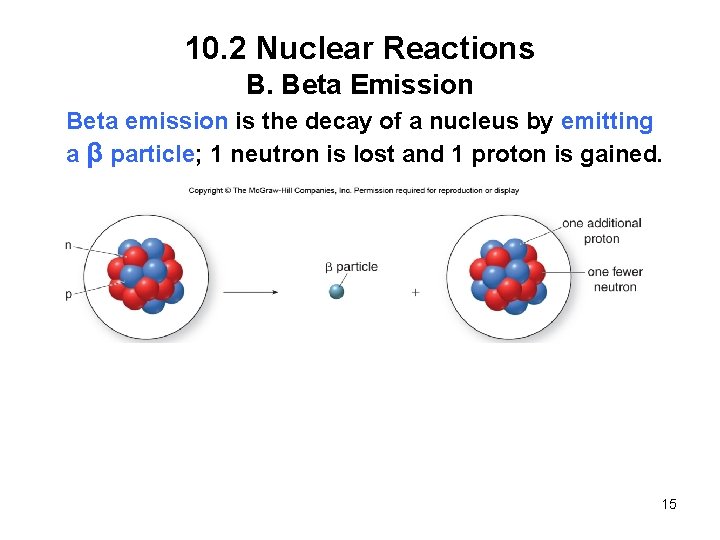
10. 2 Nuclear Reactions B. Beta Emission Beta emission is the decay of a nucleus by emitting a β particle; 1 neutron is lost and 1 proton is gained. 15

Write a balanced nuclear equation for the β emission of phosphorus-32, a radioisotope used to treat leukemia and other blood disorders 16

Write a balanced nuclear equation for the β emission of each of the following isotopes: a. 20 9 F b. 92 38 Sr c. Chromium-55 17

10. 2 Nuclear Reactions C. Positron Emission Positron emission is the decay of a nucleus by emitting a positron, β+; 1 proton is lost and 1 neutron is gained. 18

Write a balanced nuclear equation for the positron emission of fluorine-18, a radioisotope used for imaging in PET scans. 19

Write a balanced nuclear equation for the positron emission of each of the following nuclei: a. Arsenic-74 b. Oxygen-15 20

10. 2 Nuclear Reactions D. Gamma Emission Gamma emission is the decay of a nucleus by g radiation. The g rays are a form of energy only. emitting • • Their emission causes no change in the atomic number or the mass number. 99 m 43 Tc 99 43 Tc + g • Technetium-99 m is a metastable isotope; it decays by gamma emission to the more stable (but still radioactive) technetium-99. 21
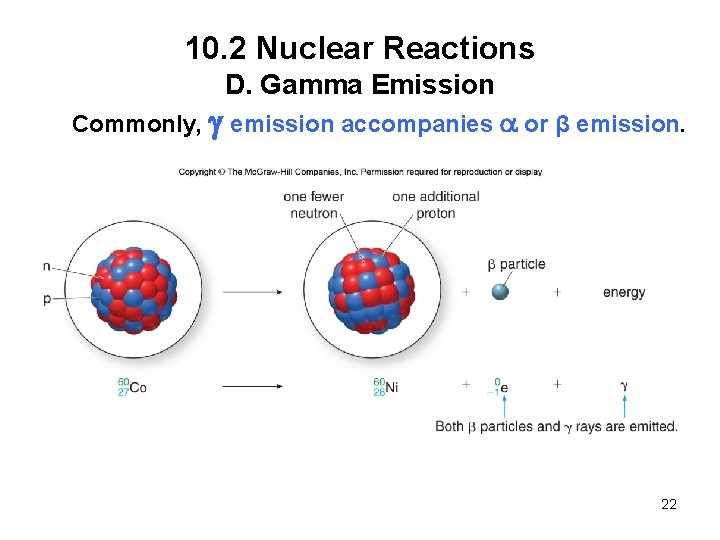
10. 2 Nuclear Reactions D. Gamma Emission Commonly, g emission accompanies a or β emission. 22

10. 1 Introduction 23

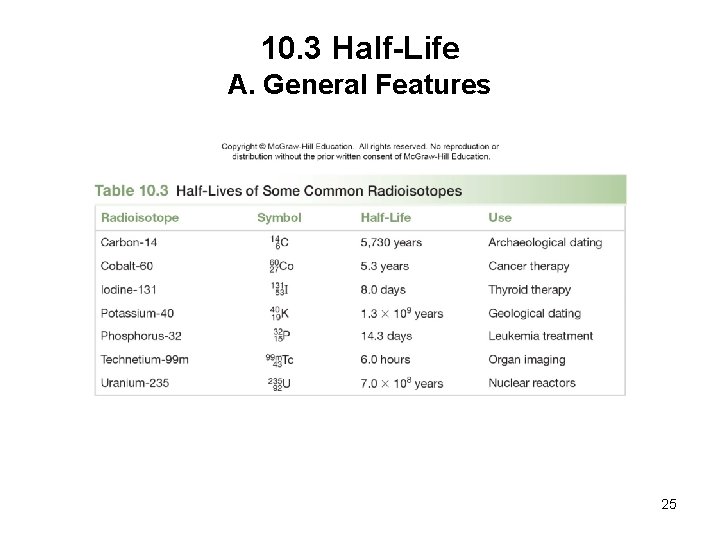
10. 3 Half-Life A. General Features The half-life (t 1/2) of a radioactive isotope is the time it takes for one-half of the sample to decay. The half-life of a radioactive isotope is a property of a given isotope and is independent of the amount of sample, temperature, and pressure. 24
10. 3 Half-Life A. General Features 25

10. 3 Half-Life A. General Features HOW TO Use a Half-Life to Determine the Amount of Radioisotope Present Example If the half-life of iodine-131 is 8. 0 days, how much of a 100. mg sample remains after 32 days? Step [1] Determine how many half-lives occur in the given amount of time. 26

10. 3 Half-Life A. General Features HOW TO Use a Half-Life to Determine the Amount of Radioisotope Present Step [2] For each half-life, multiply the initial mass by one-half to obtain the final mass. 27

If Tc-99 m has a half life of 6 hours how much of a 160 mg sample remains after the following times: a. 6 hours b. 18 hours c. 24 hours d. 2 days 28

10. 6 Nuclear Fission and Nuclear Fusion A. Nuclear Fission Nuclear fission is the splitting apart of a heavy nucleus into lighter nuclei and neutrons. It can begin when a neutron bombards a uranium-235 nucleus: • The bombarded U-235 nucleus splits apart into krypton-91, barium-142, and three high-energy neutrons, while releasing a great deal of energy. 29

10. 6 Nuclear Fission and Nuclear Fusion A. Nuclear Fission Nuclear fission is the splitting apart of a heavy nucleus into lighter nuclei and neutrons. It can begin when a neutron bombards a uranium-235 nucleus: • The released neutrons can then bombard other uranium nuclei, creating a chain reaction. • Critical mass: The minimum amount of U-235 needed to sustain a chain reaction. 30

10. 6 Nuclear Fission and Nuclear Fusion A. Nuclear Fission • A nuclear power plant uses the large amount of energy released in fission. • This energy is used to boil water and create steam, which turns a turbine and generates electricity. • The dangers of generating nuclear power are possible radiation leaks and the disposal of nuclear waste. • Radiation leaks can be minimized by containment facilities within the power plant itself. • Nuclear waste is currently buried, but it is unclear whether this is the best method. 31

Write a nuclear equation for the fission of uranium-235 by neutron bombardment to form strontium-90, an isotope of xenon, and three neutrons 32

Write a nuclear equation for the fission of uranium-235 by neutron bombardment to form antimony-133, three neutrons, and one other isotope. 33

10. 6 Nuclear Fission and Nuclear Fusion B. Nuclear Fission Nuclear fusion is the joining together of two light nuclei to form a larger nucleus. • Hydrogen-2 (deuterium) and hydrogen-3 (tritium) undergo fusion to create a helium nucleus: 2 H 1 + 3 H 1 4 He 2 + 1 n 0 • A neutron and a large amount of energy are also produced. Fusion is not currently useable as an energy source because it can only occur at extremely high temperatures and pressures. 34
- Slides: 34