1 Welcome Thanks for joining us ITRCs InternetBased

1 Welcome – Thanks for joining us. ITRC’s Internet-Based Training Program Permeable Reactive Barriers: Lessons Learned & New Directions ITRC Technical and Regulatory Guidance on Permeable Reactive Barriers: Lessons Learned / New Directions This training is co-sponsored by the EPA Office of Superfund Remediation and Technology Innovation
2 ITRC (www. itrcweb. org) – Shaping the Future of Regulatory Acceptance u u u Network • State regulators • Federal government • Industry • Consultants • Academia • Community stakeholders Documents • Technical and regulatory guidance documents • Technology overviews • Case studies Training • Internet-based • Classroom Host Organization ITRC State Members ITRC Member State Federal Partners DOE DOD EPA

3 ITRC Course Topics Planned for 2006 Popular courses from 2005 u u u u New in 2006 Alternative Landfill Covers u Characterization, Design, Construction and Monitoring of Constructed Treatment Wetlands Bioreactor Landfills Environmental Management at u Direct-Push Wells for Long-term Operational Outdoor Small Arms Monitoring Ranges u Ending Post Closure Care at DNAPL Performance Assessment Landfills Mitigation Wetlands u Planning and Promoting of Perchlorate Overview Ecological Re-use of Permeable Reactive Barriers: Remediated Sites Lessons Learn and New Direction u Rads Real-time Data Collection Radiation Risk Assessment u Remediation Process Radiation Site Cleanup Optimization Advanced Training Remediation Process Optimization u More in development……. Site Investigation and Remediation for Munitions Response Projects Triad Approach What’s New With In Training dates/details at www. itrcweb. org Situ Chemical Training archives at http: //cluin. org/live/archive. cfm Oxidation

4 Permeable Reactive Barriers: Lessons Learned & New Directions Logistical Reminders • Phone line audience ü Keep phone on mute ü *6 to mute, *7 to un-mute to ask question during designated periods ü Do NOT put call on hold • Simulcast audience ü Use at the top of each slide to submit questions • Course time = 2¼ hours Presentation Overview • • • Hydraulic issues Performance issues Iron and zeolite case studies Questions and answers Bio-barrier case study Compost wall case study Links to additional resources Your feedback Questions and answers

5 Meet the ITRC Instructors Mike Duchene Enviro. Metal Technologies Inc. Waterloo, Ontario 519. 746. 2204 mduchene@eti. ca Scott Warner Geomatrix Consultants Oakland, California 510. 663. 4269 swarner@geomatrix. com

6 Meet the ITRC Instructors Alec Naugle California Water Quality Regional Control Board Oakland, California 510. 622. 2510 anaugle@waterboards. ca. gov Dave Smyth Department of Earth Sciences, University of Waterloo, Ontario 519. 888. 4567 dsmyth@sciborg. uwaterloo. ca

7 What You Will Learn… Update on the general performance of PRBs from over the last 10 years u Specific details on the design, operation, monitoring, and assessments of PRBs presented as four case studies u • • Iron PRB for treatment of VOCs Sorption barrier for treatment of radionuclides Bio barrier for treatment of VOCs Solid organic carbon barrier for treatment of acid mine drainage

8 History of PRBs and the ITRC PRB Team u PRB technology (iron-based) • 10 years of data • Has evolved from innovative to accepted standard practice u ITRC PRB Team • Created in 1996 • Produced 4 ITRC guidance documents • Collaborated on 2 additional guidance documents with Do. D, DOE, and EPA • Delivered 14 classroom training sessions • Created 3 Internet-based training courses delivering more than 20 classes
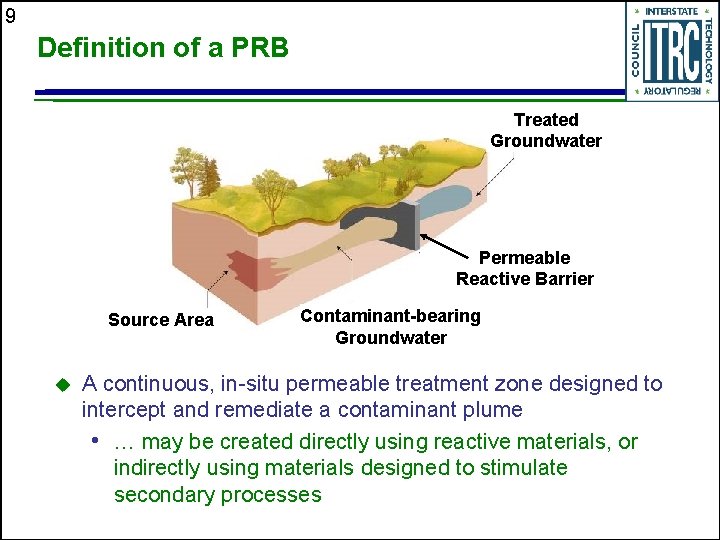
9 Definition of a PRB Treated Groundwater Permeable Reactive Barrier Source Area u Contaminant-bearing Groundwater A continuous, in-situ permeable treatment zone designed to intercept and remediate a contaminant plume • … may be created directly using reactive materials, or indirectly using materials designed to stimulate secondary processes
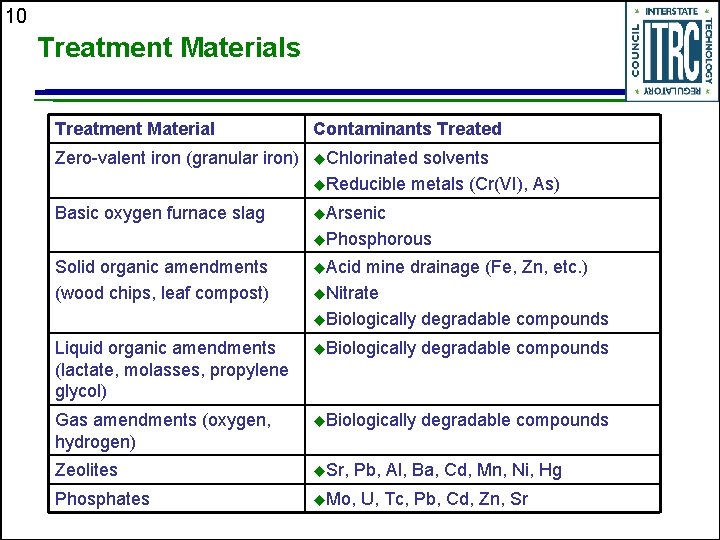
10 Treatment Materials Treatment Material Contaminants Treated Zero-valent iron (granular iron) u. Chlorinated Basic oxygen furnace slag u. Arsenic solvents u. Reducible metals (Cr(VI), As) u. Phosphorous Solid organic amendments (wood chips, leaf compost) u. Acid mine drainage (Fe, Zn, etc. ) u. Nitrate u. Biologically degradable compounds Liquid organic amendments (lactate, molasses, propylene glycol) u. Biologically degradable compounds Gas amendments (oxygen, hydrogen) u. Biologically degradable compounds Zeolites u. Sr, Phosphates u. Mo, Pb, Al, Ba, Cd, Mn, Ni, Hg U, Tc, Pb, Cd, Zn, Sr
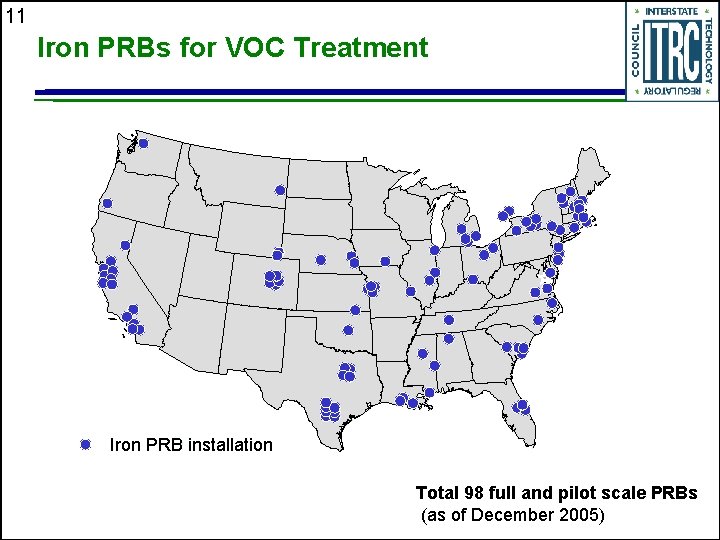
11 Iron PRBs for VOC Treatment Iron PRB installation Total 98 full and pilot scale PRBs (as of December 2005)

12 Performance Assessment u Four types of assessment • • Chemical (contaminants of concern) Hydraulic Geochemical Microbial Assessments are interdependent u Section focuses on granular iron PRBs for VOC treatment u
13 Monitoring Programs u u Compliance monitoring ( wells) • Driven by regulatory requirements • Typically only contaminants are regulated Performance monitoring ( wells) • Identify any changes in system that may affect treatment effectiveness and longevity

14 Downgradient Concentrations u u Compliance wells installed in contaminated aquifer downgradient of PRB Time required for ‘flushing’ of contaminants in downgradient aquifer Varies at each site Challenge for assessment

15 Hydraulic Performance u Deviation from hydraulic design performance caused by one or more of the following • Sewer lines influencing flow • Construction artifacts altering flow u Result can be • Reduced residence time leading to insufficient treatment • Plume bypass

16 Hydraulic Issues - Characterization Thorough site characterization required as PRBs are not easily modified u Past characterization issues include u • • Incomplete plume capture in 3 dimensions Variation in seasonal flow direction Variation in hydraulic gradient and permeability Incorrect groundwater flow velocity / variation along PRB alignment • Influence of sewer lines
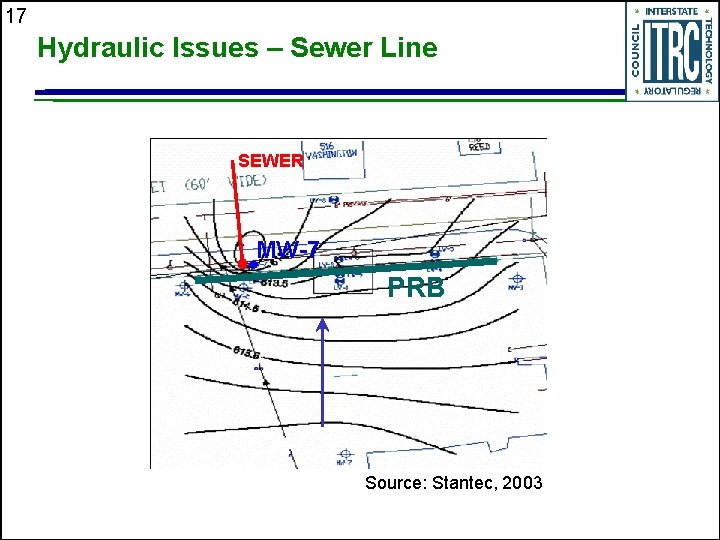
17 Hydraulic Issues – Sewer Line SEWER MW-7 PRB Source: Stantec, 2003

18 Hydraulic Issues – Construction Related u u u Aquifer sediments mixing with reactive media Reduced permeability zone at interface Short-term effect on hydraulics

19 Hydraulic Issues – Funnel and Gate PRBs u u Inadequate funnel length Flow over reactive material in gate Reduced hydraulic conductivity zone at gate entrance Flow beneath funnel sections

20 Geochemical Performance u Reactions within PRB result in change in geochemistry • Provides evidence treatment process is working § Increase in p. H, decrease in redox potential (Eh) § Reduction in carbonate concentration u Provides data to assess longevity of PRBs

21 Geochemical Performance u u u Carbonate precipitates may drive long-term performance (sulphides in some cases) Precipitate build-up begins at upgradient interface Long-term lab simulations show some permeability loss and significant reactivity loss in precipitate zones

22 Geochemical Performance No indication to date that precipitates causing sufficient loss of reactivity requiring rejuvenation and/or iron replacement u Flow through PRBs at field sites to date much less than simulated flow through laboratory columns u Estimate of >10 -15 years before refurbishing at most sites appears reasonable u • Dependent on mass flux of carbonate

23 Microbial Assessment Microbial activity does occur in iron PRBs u No indication of significant biomass buildup u • Exception is Denver Federal Center – attributed to low-flow conditions and high sulfate Shift in populations to sulphate reducers and anaerobic metal reducers u Potential beneficial effects u

24 PRB Cost Comparison u u 20’ 60’ 1 ft / day Confining Unit Economic analysis by Du. Pont for EPA/RTDF training Evaluated • PRB with natural attenuation • Pump and treat • Monitored natural attenuation

25 Cost Comparison Details u Plume • TCE=10, 000 ppb, c. DCE=1, 000 ppb, VC=100 ppb Treatment to federal MCLs u Capital costs u • Design and construction of PRB or pump and treat system • Monitoring wells u Operating costs • Sampling and analysis • Operations for pump and treat system
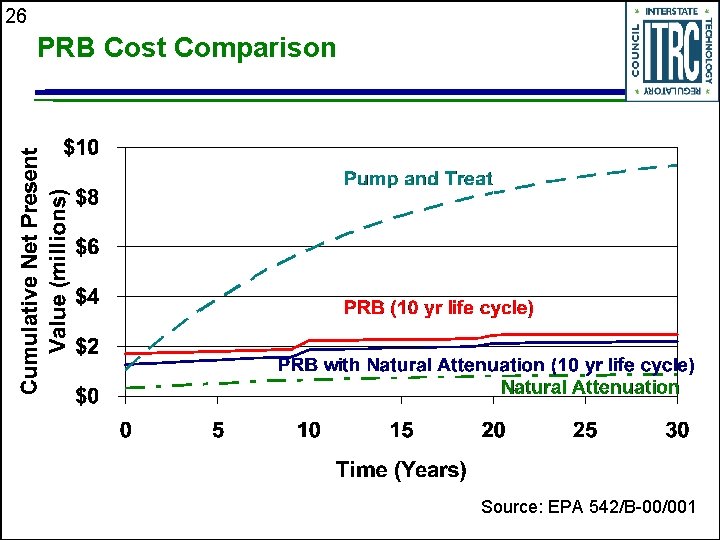
26 PRB Cost Comparison Source: EPA 542/B-00/001

27 Summary Of the few systems with inadequate performance, system hydraulics are the main cause u Ongoing refinement/improvement of construction methods is minimizing adverse impacts due to construction u Estimate of >10 -15 years before refurbishing at most sites appears reasonable u
28 First Commercial Granular-Iron PRB 10 Year Update Site N Santa Clara County, California

29 Site History 1983 -1987 Site characterization, source remediation, pump and treat implementation 1991 -1993 PRB concept, design work, regulatory process Nov 1994 Feb 1995 PRB construction 1999 Five-year effectiveness evaluation 2004 Ten-year effectiveness evaluation Historical notables • Hydrogen gas monitoring, passive diffusion bag sampling • Slurry wall breach and repair, system alarm installation

30 Focus of Case Study Presentation Layout u Hydraulics u Geochemical conditions u • p. H • Redox • Inorganic character Dissolved gas measurements u Economic performance u
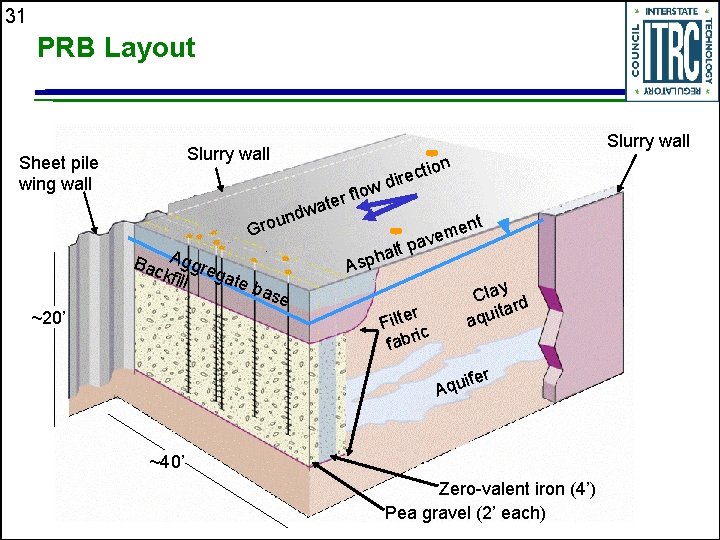
31 PRB Layout Slurry wall Sheet pile wing wall ow l f r e wat d n u Gro Bac Aggre gate kfill b ase ~20’ ion t c e dir ent m e av alt p h p As r Filte ic fabr Clay d itar u q a ifer u q A ~40’ Zero-valent iron (4’) Pea gravel (2’ each)
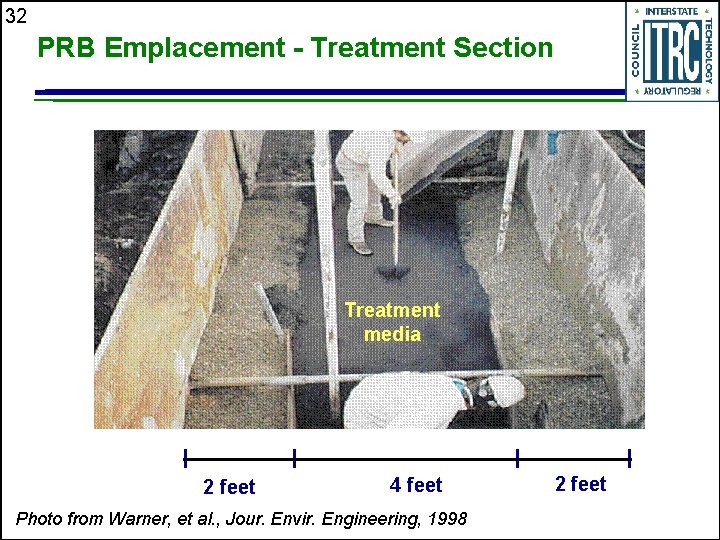
32 PRB Emplacement - Treatment Section Treatment media 2 feet 4 feet Photo from Warner, et al. , Jour. Envir. Engineering, 1998 2 feet
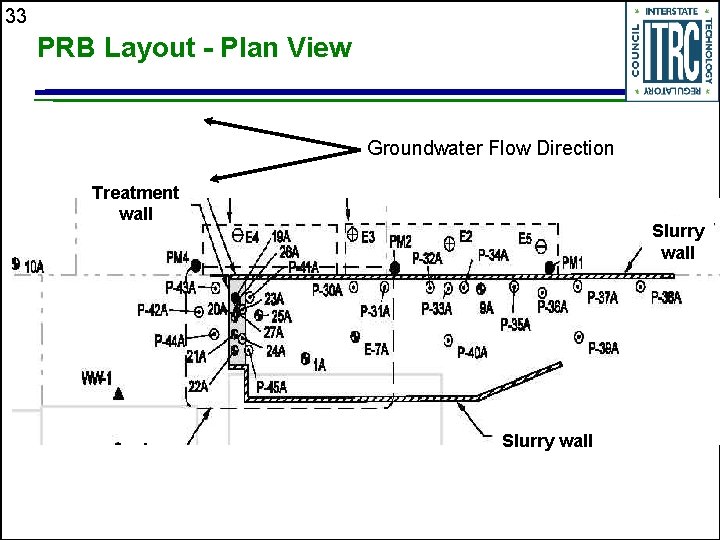
33 PRB Layout - Plan View Groundwater Flow Direction Treatment wall Slurry wall
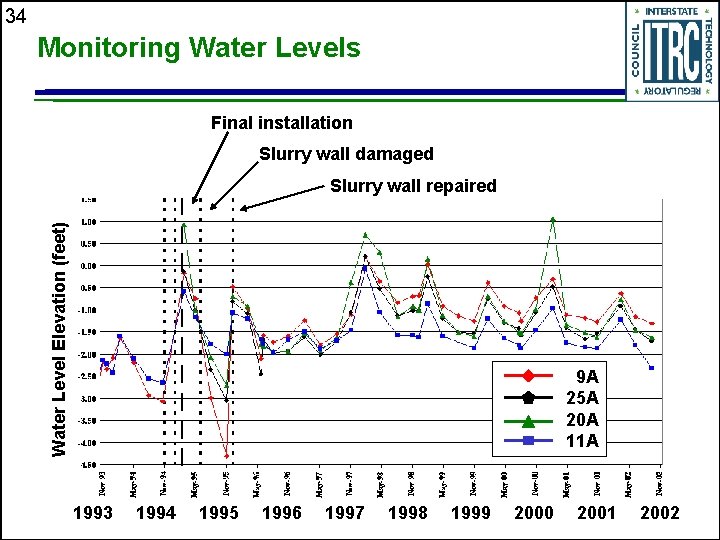
34 Monitoring Water Levels Final installation Slurry wall damaged Water Level Elevation (feet) Slurry wall repaired 9 A 25 A 20 A 11 A 1993 1994 1995 1996 1997 1998 1999 2000 2001 2002
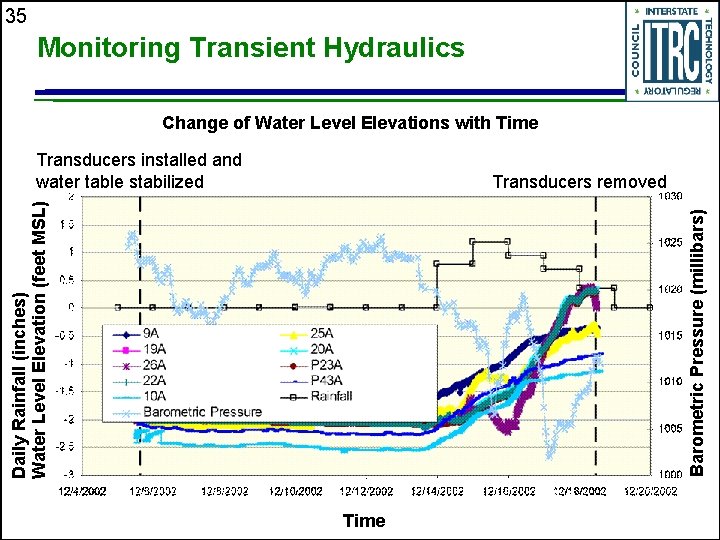
35 Monitoring Transient Hydraulics Change of Water Level Elevations with Time Transducers installed and water table stabilized Barometric Pressure (millibars) Daily Rainfall (inches) Water Level Elevation (feet MSL) Transducers removed Time
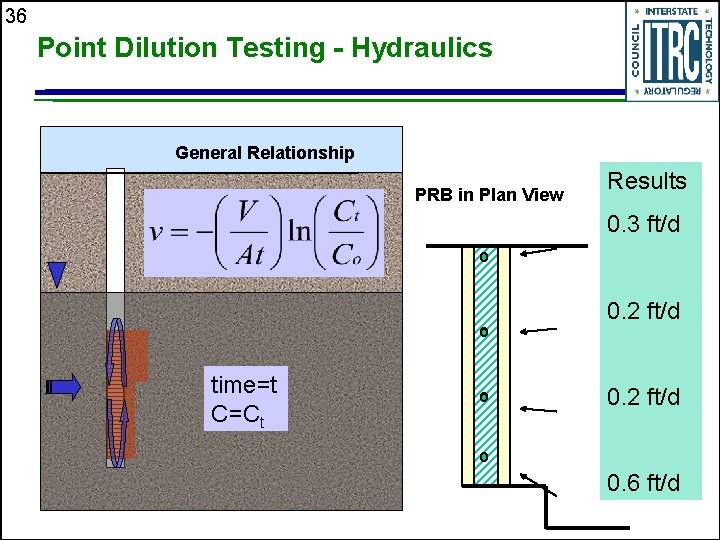
36 Point Dilution Testing - Hydraulics General Relationship PRB in Plan View Results 0. 3 ft/d 0. 2 ft/d time=t C=Ct 0. 2 ft/d 0. 6 ft/d
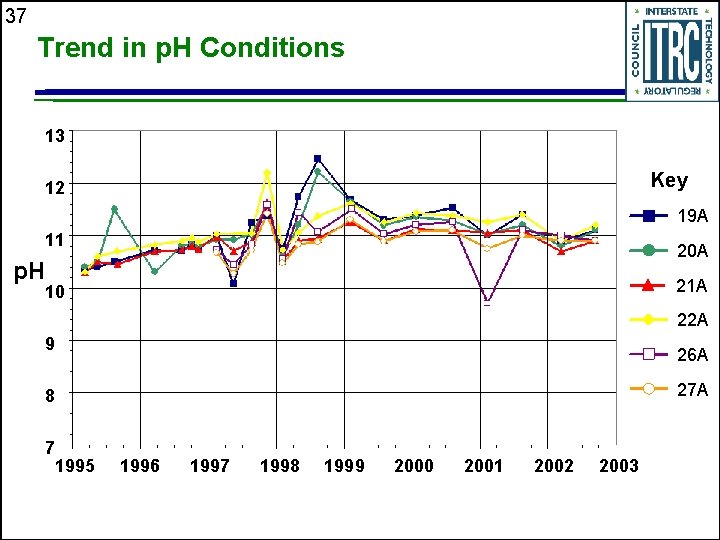
37 Trend in p. H Conditions 13 Key 12 19 A 11 20 A p. H 21 A 10 22 A 9 26 A 27 A 8 7 1995 1996 1997 1998 1999 2000 2001 2002 2003
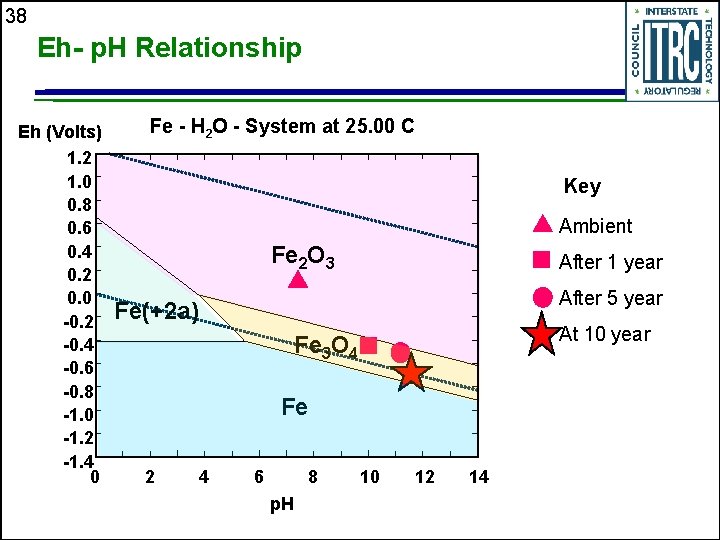
38 Eh- p. H Relationship Eh (Volts) 1. 2 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 -0. 2 -0. 4 -0. 6 -0. 8 -1. 0 -1. 2 -1. 4 0 Fe - H 2 O - System at 25. 00 C Key Ambient Fe 2 O 3 After 1 year After 5 year Fe(+2 a) At 10 year Fe 3 O 4 Fe 2 4 6 8 p. H 10 12 14
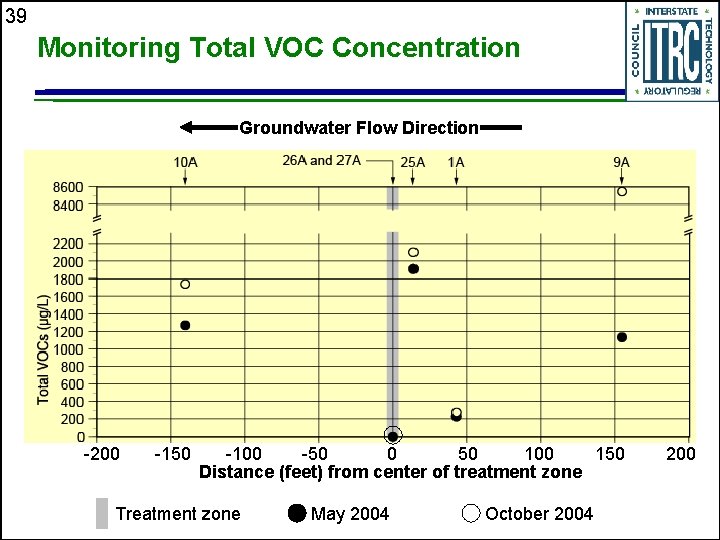
39 Monitoring Total VOC Concentration Groundwater Flow Direction -200 -150 -100 -50 0 50 100 150 Distance (feet) from center of treatment zone Treatment zone May 2004 October 2004 200
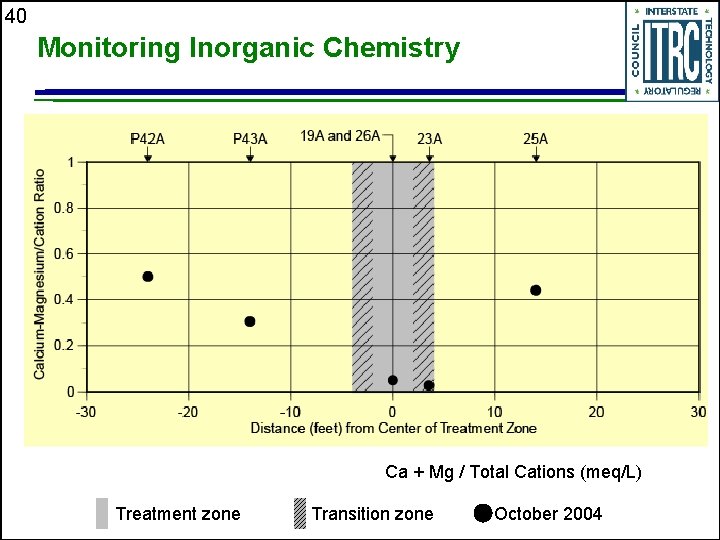
40 Monitoring Inorganic Chemistry Ca + Mg / Total Cations (meq/L) Treatment zone Transition zone October 2004
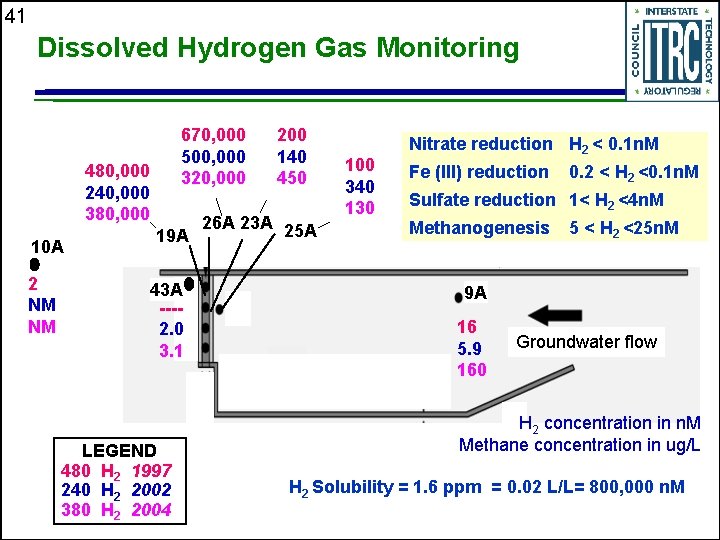
41 Dissolved Hydrogen Gas Monitoring 670, 000 500, 000 320, 000 480, 000 240, 000 380, 000 10 A 2 NM NM 19 A 43 A ---2. 0 3. 1 LEGEND 480 H 2 1997 240 H 2 2002 380 H 2 2004 200 140 450 26 A 23 A 25 A 100 340 130 Nitrate reduction H 2 < 0. 1 n. M Fe (III) reduction 0. 2 < H 2 <0. 1 n. M Sulfate reduction 1< H 2 <4 n. M Methanogenesis 5 < H 2 <25 n. M 9 A 16 5. 9 160 Groundwater flow H 2 concentration in n. M Methane concentration in ug/L H 2 Solubility = 1. 6 ppm = 0. 02 L/L= 800, 000 n. M
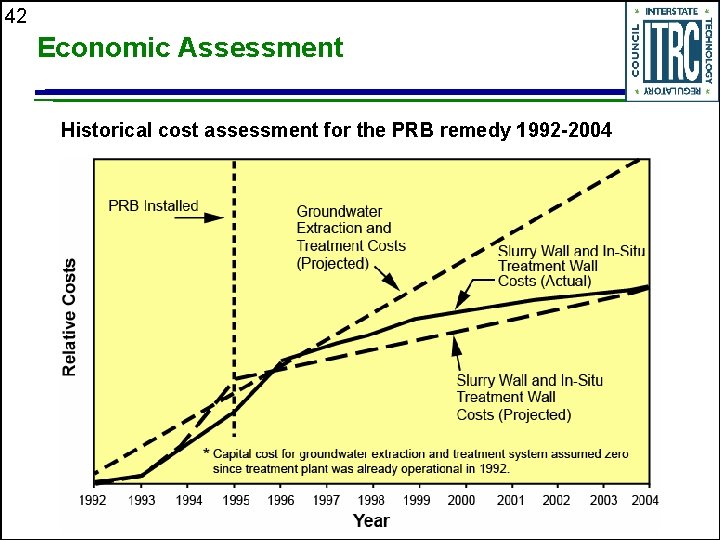
42 Economic Assessment Historical cost assessment for the PRB remedy 1992 -2004

43 The First Zero-valent Iron PRB at 10 Years Remedial intent is being achieved u Economic property use has been restored u Geochemical conditions confirm remedial process u Mineralization likely; no significant affect - yet u Hydrogen shows effect beyond PRB boundary u Hydraulic conditions are transient u • Flow conditions appear to be maintained

44 Performance of a Zeolitic PRB West Valley, New York Zeolite (clinoptilolite) (Na, K, Ca)2 -3 Al 3(Al, Si)2 Si 13 O 36 -12 H 2 O Specific Gravity = 2 to 2. 5 Cation Exchange Capacity = 1. 7 meq/g Sorption capacity 2350 m. L/g Bulk Density 0. 8 g/m. L material testing by University at Buffalo
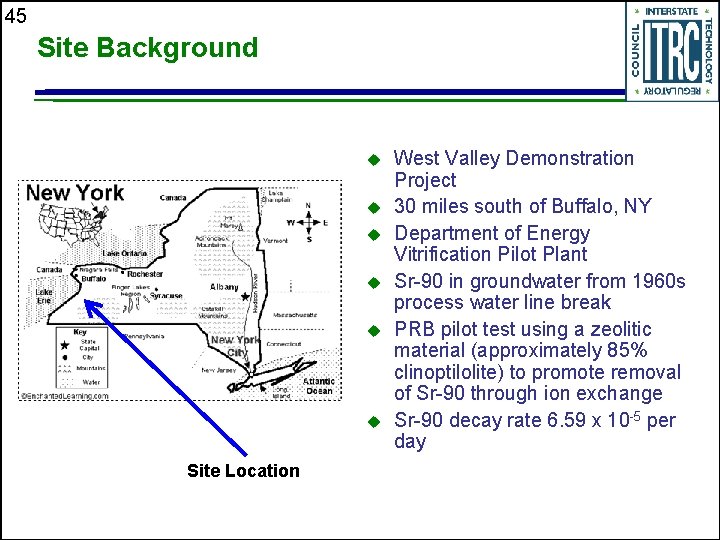
45 Site Background u u u Site Location West Valley Demonstration Project 30 miles south of Buffalo, NY Department of Energy Vitrification Pilot Plant Sr-90 in groundwater from 1960 s process water line break PRB pilot test using a zeolitic material (approximately 85% clinoptilolite) to promote removal of Sr-90 through ion exchange Sr-90 decay rate 6. 59 x 10 -5 per day

46 Case Study Focus Areas Hydraulic performance anomalies u Post construction evaluation u Monitoring of inorganic performance parameters u Consideration of hydraulic improvements – if needed u Reassessment of performance – long-term u Evaluating success of the pilot program with respect to developing a potential full-scale design u
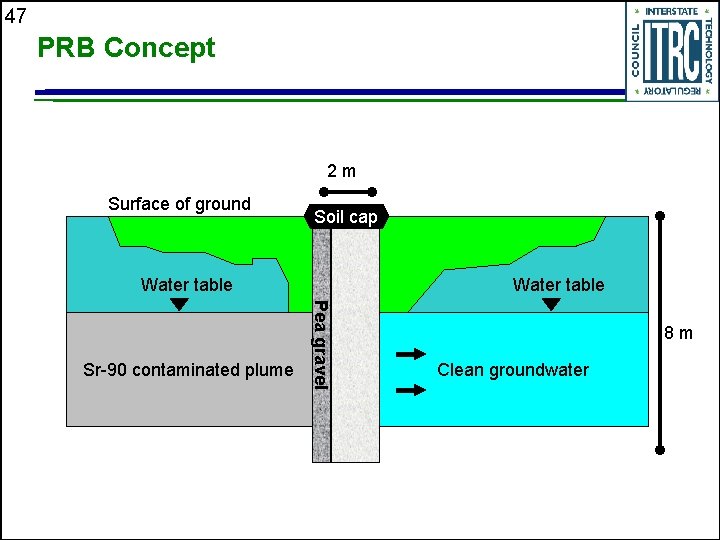
47 PRB Concept 2 m Surface of ground Soil cap Water table Pea gravel Sr-90 contaminated plume Water table 8 m Clean groundwater

48 Construction

49 Construction Excavation Gravel and Zeolite Placement
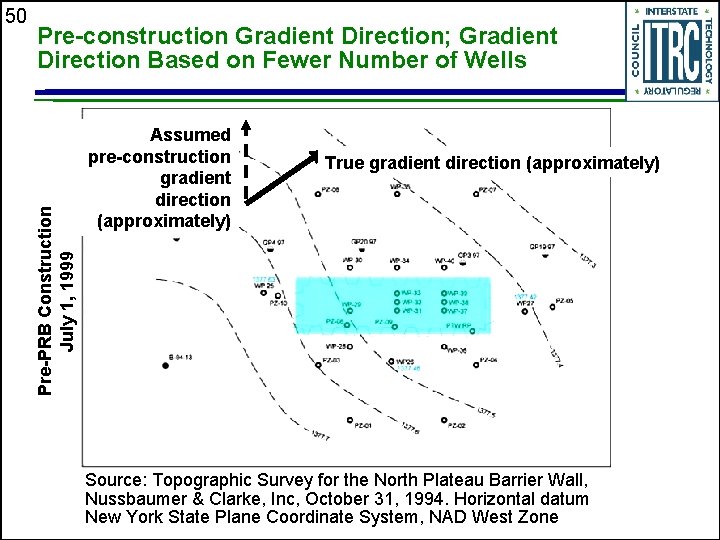
Pre-construction Gradient Direction; Gradient Direction Based on Fewer Number of Wells Pre-PRB Construction July 1, 1999 50 Assumed pre-construction gradient direction (approximately) True gradient direction (approximately) Source: Topographic Survey for the North Plateau Barrier Wall, Nussbaumer & Clarke, Inc, October 31, 1994. Horizontal datum New York State Plane Coordinate System, NAD West Zone
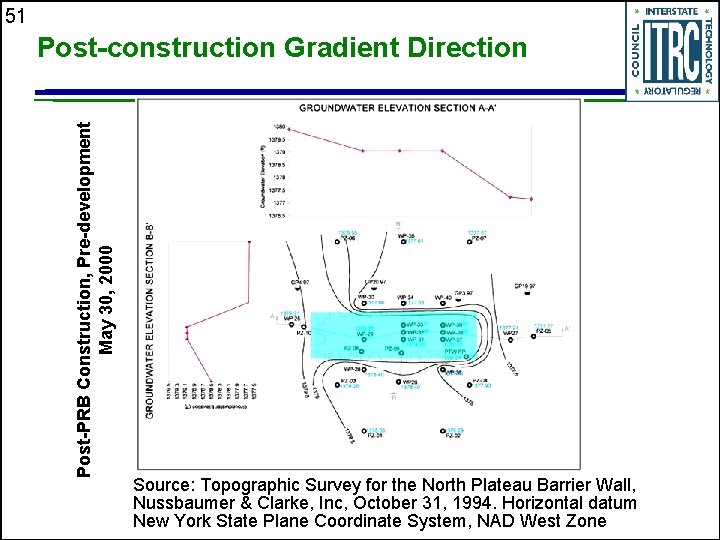
51 Post-PRB Construction, Pre-development May 30, 2000 Post-construction Gradient Direction Source: Topographic Survey for the North Plateau Barrier Wall, Nussbaumer & Clarke, Inc, October 31, 1994. Horizontal datum New York State Plane Coordinate System, NAD West Zone
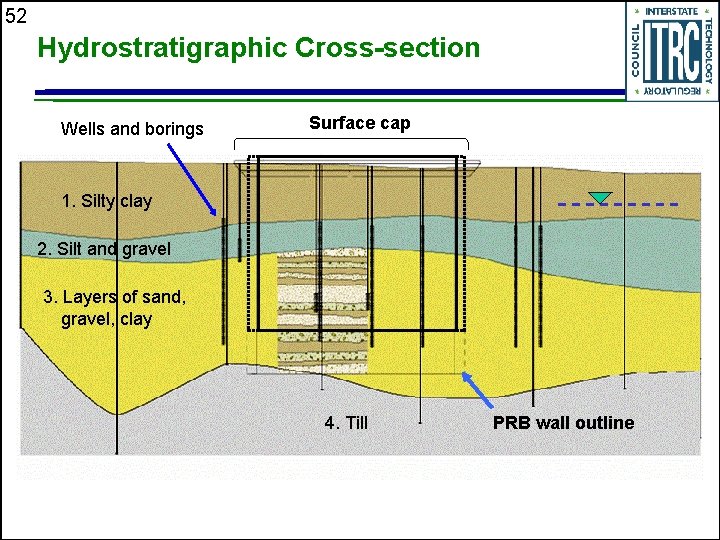
52 Hydrostratigraphic Cross-section Wells and borings Surface cap 1. Silty clay 2. Silt and gravel 3. Layers of sand, gravel, clay 4. Till PRB wall outline
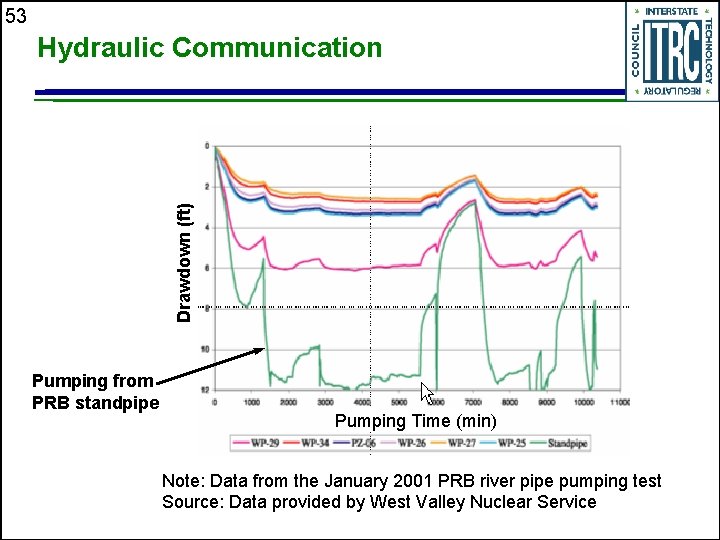
53 Drawdown (ft) Hydraulic Communication Pumping from PRB standpipe Pumping Time (min) Note: Data from the January 2001 PRB river pipe pumping test Source: Data provided by West Valley Nuclear Service
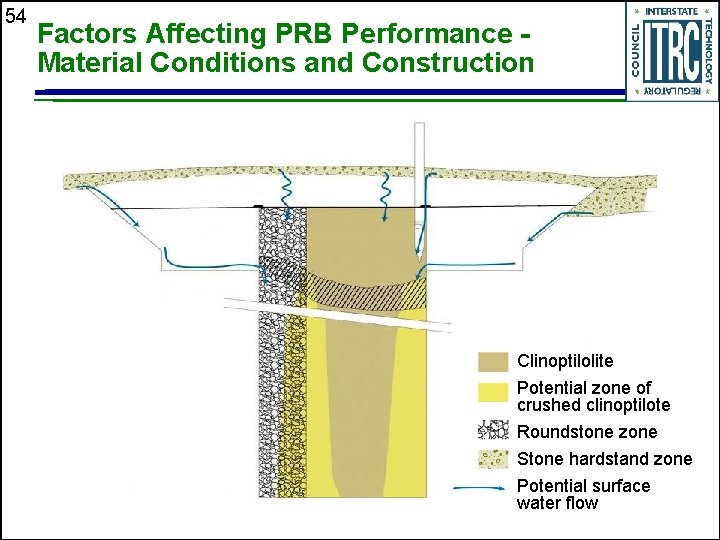
54 Factors Affecting PRB Performance Material Conditions and Construction Clinoptilolite Potential zone of crushed clinoptilote Roundstone zone Stone hardstand zone Potential surface water flow
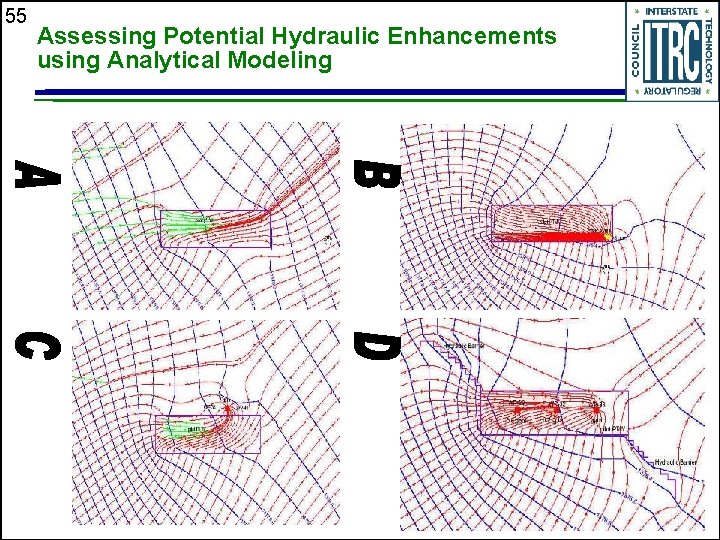
55 Assessing Potential Hydraulic Enhancements using Analytical Modeling
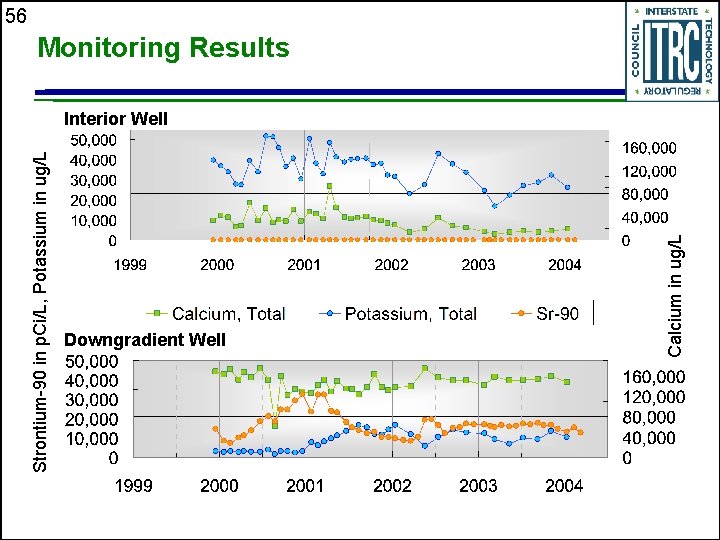
Strontium-90 in p. Ci/L, Potassium in ug/L Downgradient Well Calcium in ug/L 56 Monitoring Results Interior Well

57 Lessons Learned u Site characterization • Hydraulic conditions • Vertical and lateral heterogeneity Pilot testing objectives u Construction methods u Details for potential full-scale installation u

58 Questions and Answers

59 Bio-Barriers: In-Situ Bioremediation Using a Permeable Reactive Barrier Design Presentation Overview: u What is a bio-barrier? u Typical “amendments” for in-situ bioremediation u Case example
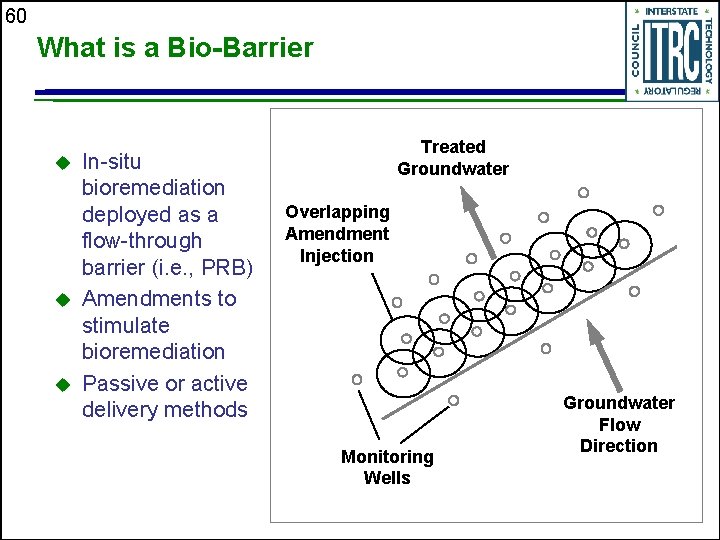
60 What is a Bio-Barrier u u u In-situ bioremediation deployed as a flow-through barrier (i. e. , PRB) Amendments to stimulate bioremediation Passive or active delivery methods Treated Groundwater Overlapping Amendment Injection Monitoring Wells Groundwater Flow Direction

61 Example of Bio-Barrier Using Oxygen/Air Injection Groundwater Flow Monitoring Wells Air/O 2 injection Wells

62 Typical Bio-Barrier Amendments u Solid organic amendments u Liquid organic amendments • Wood chips • Leaf compost • • • Lactate Molasses Cheese whey Propylene glycol Edible vegetable oils u Gas amendments u Microbial cultures (bioaugmentation) • Oxygen / air • Hydrogen • Oxygen / hydrogen release compound (ORC / HRC)
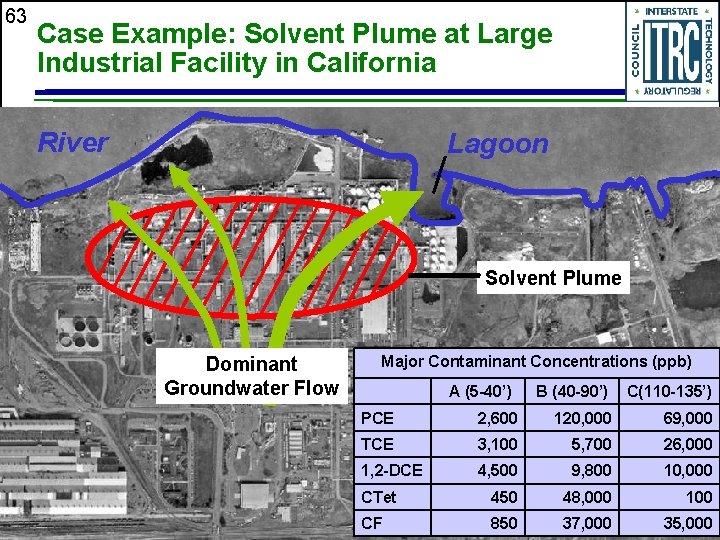
63 Case Example: Solvent Plume at Large Industrial Facility in California River Lagoon Solvent Plume Dominant Groundwater Flow Major Contaminant Concentrations (ppb) A (5 -40’) B (40 -90’) C(110 -135’) PCE 2, 600 120, 000 69, 000 TCE 3, 100 5, 700 26, 000 1, 2 -DCE 4, 500 9, 800 10, 000 CTet 450 48, 000 100 CF 850 37, 000 35, 000
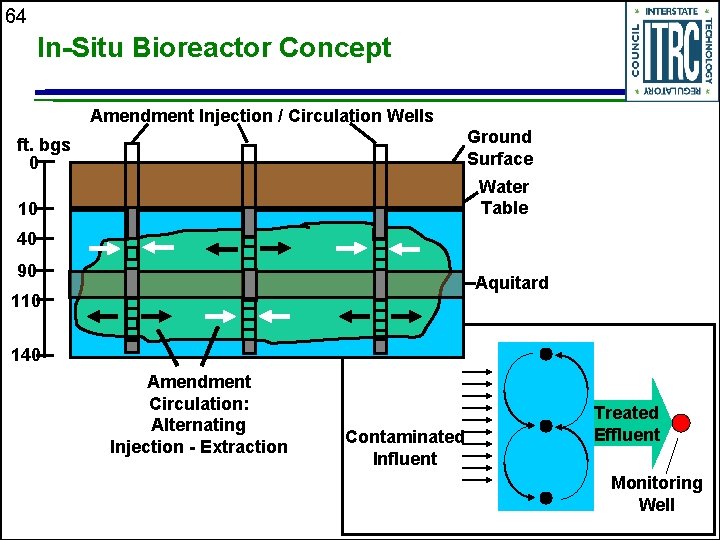
64 In-Situ Bioreactor Concept Amendment Injection / Circulation Wells Ground Surface ft. bgs 0 Water Table 10 40 90 Aquitard 110 140 Amendment Circulation: Alternating Injection - Extraction Contaminated Influent Treated Effluent Monitoring Well
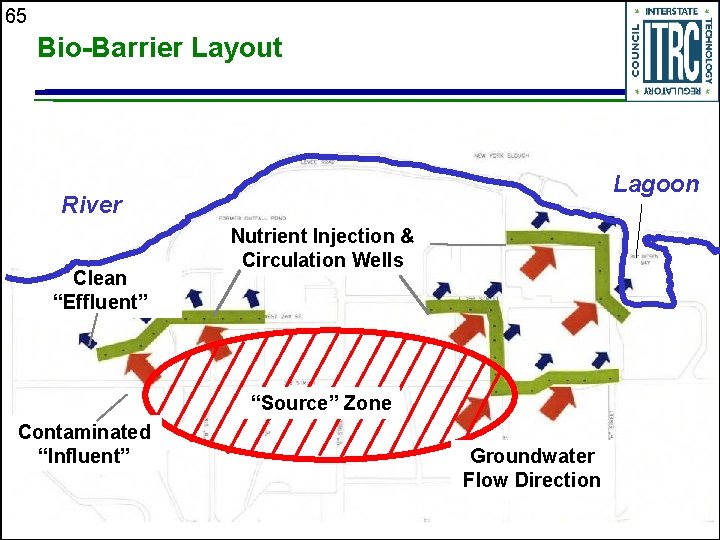
65 Bio-Barrier Layout Lagoon River Clean “Effluent” Nutrient Injection & Circulation Wells “Source” Zone Contaminated “Influent” Groundwater Flow Direction
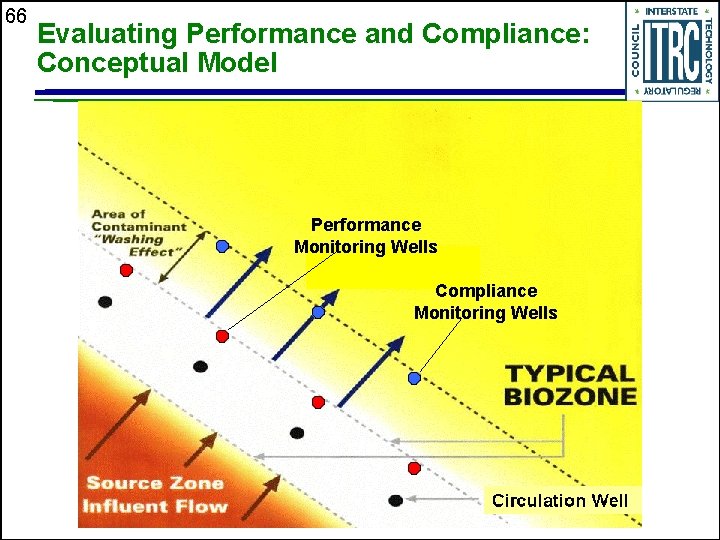
66 Evaluating Performance and Compliance: Conceptual Model Performance Monitoring Wells Compliance Monitoring Wells
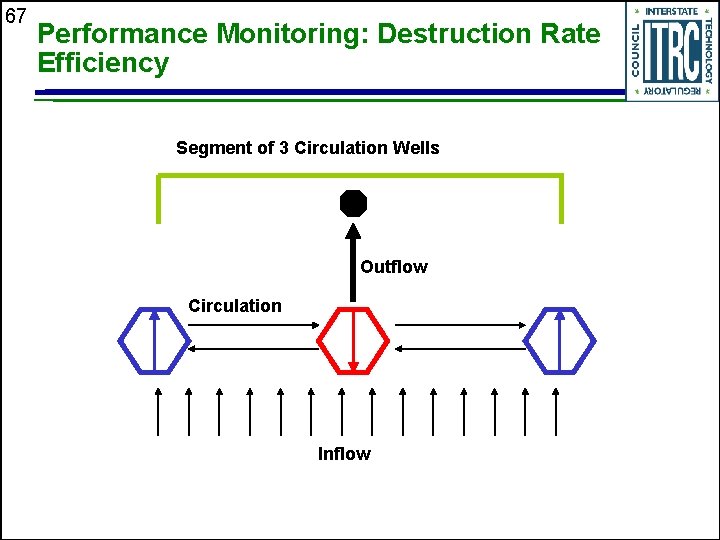
67 Performance Monitoring: Destruction Rate Efficiency Segment of 3 Circulation Wells Outflow Circulation Inflow
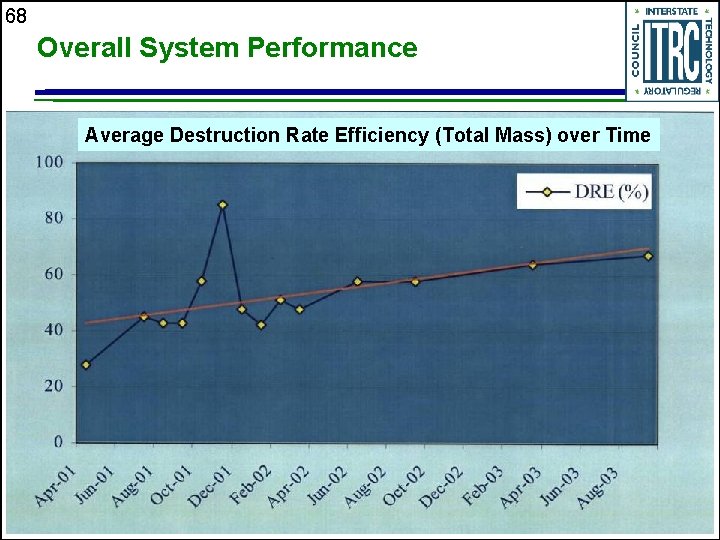
68 Overall System Performance Average Destruction Rate Efficiency (Total Mass) over Time
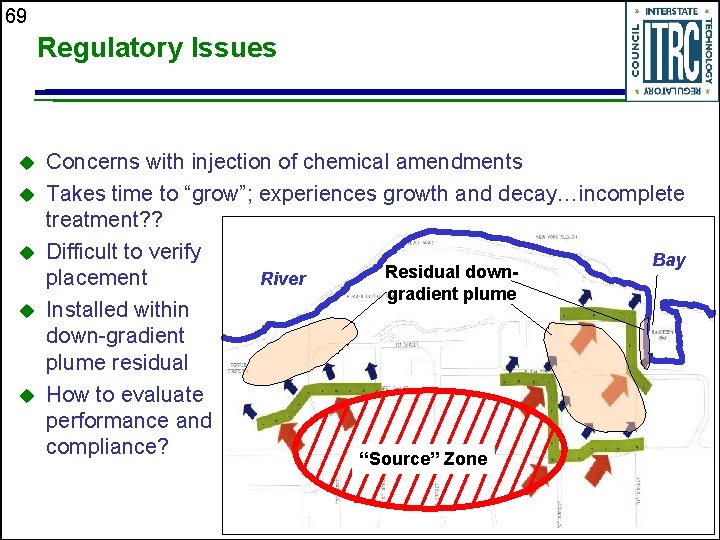
69 Regulatory Issues u u u Concerns with injection of chemical amendments Takes time to “grow”; experiences growth and decay…incomplete treatment? ? Difficult to verify Bay Residual down. River placement gradient plume Installed within down-gradient plume residual How to evaluate performance and compliance? “Source” Zone

70 Operation and Maintenance Challenges Daily/Weekly Injection of Amendments On-Site Storage of Amendments Bio-fouling of Wells

71 Bio-Barriers Summary and Conclusions Bio-barriers function similar to PRBs u Significant regulatory concerns and O&M challenges exist u Monitoring program is key to demonstrate success: u • Different performance vs. compliance well locations • Down-gradient residual can obscure results

72 Sulfate-Reduction PRBs u Metals in sulfate-rich groundwater • Acid-mine drainage • Industrial and waste management sites Organic carbon in reactive mixtures u Field-scale PRB installed at the Nickel Rim Mine, Sudbury, Canada in 1995 u
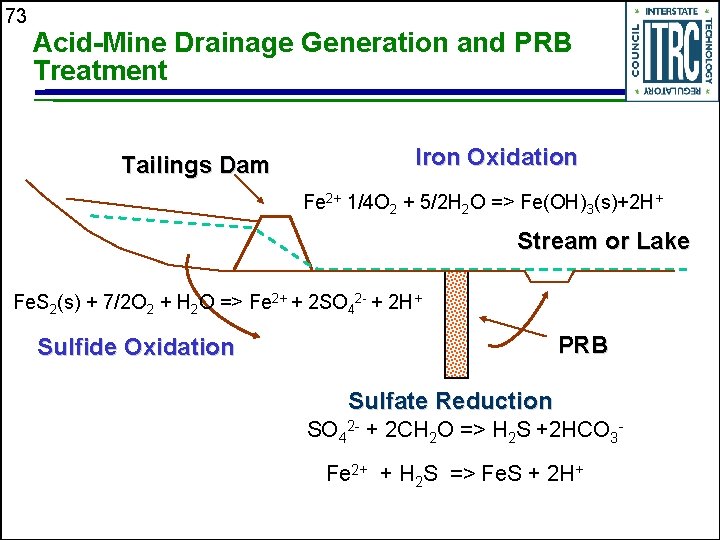
73 Acid-Mine Drainage Generation and PRB Treatment Tailings Dam Iron Oxidation Fe 2+ 1/4 O 2 + 5/2 H 2 O => Fe(OH)3(s)+2 H+ Stream or Lake Fe. S 2(s) + 7/2 O 2 + H 2 O => Fe 2+ + 2 SO 42 - + 2 H+ PRB Sulfide Oxidation Sulfate Reduction SO 42 - + 2 CH 2 O => H 2 S +2 HCO 3 Fe 2+ + H 2 S => Fe. S + 2 H+

74 Remedial Objectives for Acid-Mine Drainage in Groundwater u Removal of sulfate and iron • Acid-generating potential of ferrous iron if discharged to surface water Removal of metals and metalloids u Generation of alkalinity; acid consuming characteristics u

75 Sulfate-Reduction Process Carbon provides energy for sulfate-reducing bacteria (SRBs) u Reduction of sulfate to H 2 S u H 2 S combines with metals u Precipitation of metal sulfide minerals u Decreases concentrations of sulfate and dissolved metals u Increased alkalinity and p. H u

76 Nickel Rim Mine, Sudbury, Ontario u u u u Field installation (1995) following laboratory batch and column studies Acid-Mine Drainage (AMD)-impacted plume from tailings Shallow aquifer in bedrock-bounded valley p. H ~ 6; 1, 000 -7, 000 mg/L SO 4; 500 -2, 000 mg/L Fe Plume advancing so sulfate and iron concentrations increasing with time Water table very close to ground surface Groundwater temperature fluctuations (winter and summer influences) Tailings-impacted material in surficial sediments
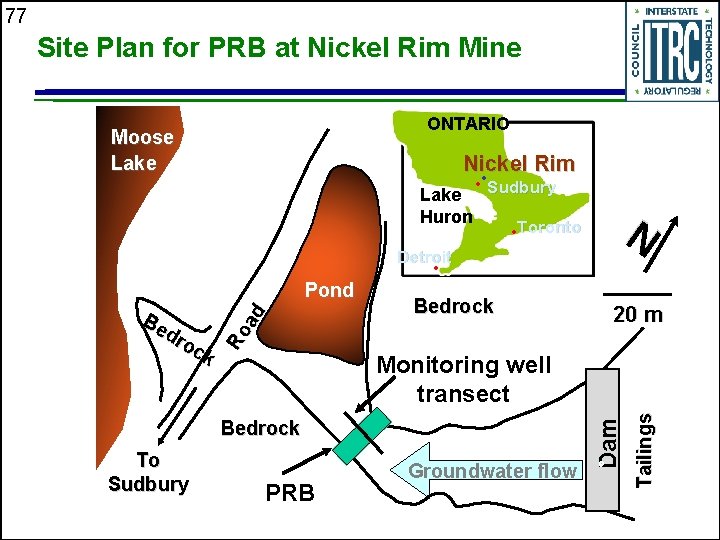
77 Site Plan for PRB at Nickel Rim Mine ONTARIO Moose Lake Nickel Rim • Toronto N Lake Huron • • Sudbury Detroit • Bedrock PRB Groundwater flow Tailings Monitoring well transect Bedrock To Sudbury 20 m Dam Be dr oc k Ro ad Pond

78 PRB Construction Backfilled trench; unsupported excavation u Reactive materials (40 % municipal plant compost, 40 % leaf mulch, 19 % wood chips, 1% limestone) mixed 1: 1 with gravel u 15 m long, 4 m deep and 8 m thick including sand zones u Cost for materials and installation approximately $35 K (US) in 1995 u Benner et al. , 1997; 1999; 2002 u

79 Installation of Nickel Rim PRB

80 Nickel Rim PRB

81 Nickel Rim PRB Clay Cap © University of Waterloo
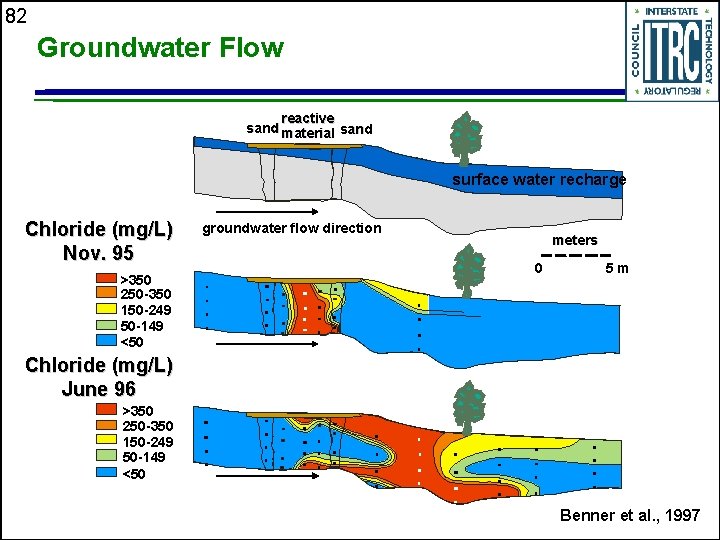
82 Groundwater Flow reactive sand material sand surface water recharge Chloride (mg/L) Nov. 95 >350 250 -350 150 -249 50 -149 <50 groundwater flow direction meters 0 5 m Chloride (mg/L) June 96 >350 250 -350 150 -249 50 -149 <50 Benner et al. , 1997
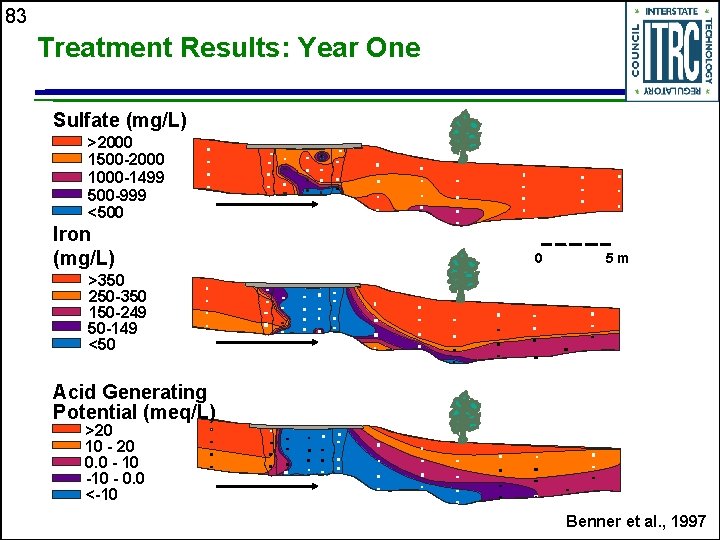
83 Treatment Results: Year One Sulfate (mg/L) >2000 1500 -2000 1000 -1499 500 -999 <500 Iron (mg/L) >350 250 -350 150 -249 50 -149 <50 meters sand reactive material sand 0 5 m groundwater flow direction Acid Generating Potential (meq/L) >20 10 - 20 0. 0 - 10 - 0. 0 <-10 Benner et al. , 1997
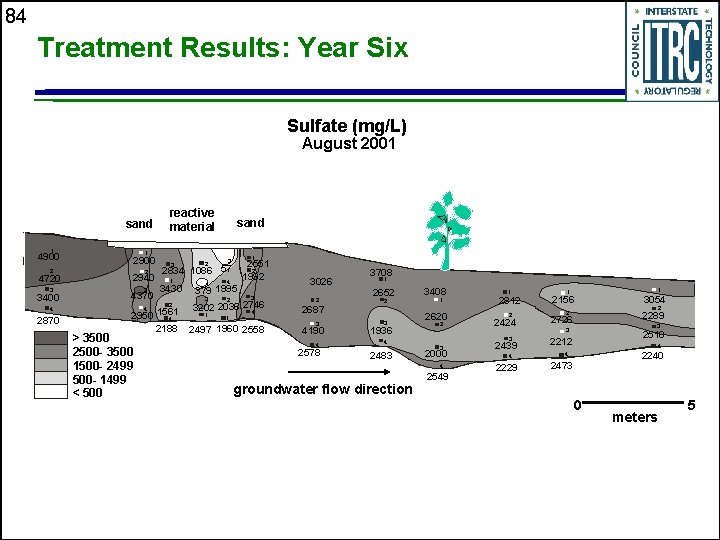
84 Treatment Results: Year Six Sulfate (mg/L) August 2001 sand 1 4900 2 2 2940 3 3 4 2870 sand 1 2900 4720 3400 reactive material 4370 1 3430 2 4 2950 1561 4 2188 > 3500 2500 - 3500 1500 - 2499 500 - 1499 < 500 2 3 4 4 3 2 1 2551 2 1992 3 2834 1086 57 379 1995 3 3026 2 3202 2036 2746 4 2687 2497 1960 2558 4190 1 1 3 4 2578 3708 1 2652 2 3 1936 4 2483 3408 1 2620 2 2 2424 3 3 2000 4 groundwater flow direction 1 2812 2549 2439 4 2229 1 1 2156 3054 2 2 2289 3 2510 2726 3 2212 4 2240 4 2473 0 meters 5
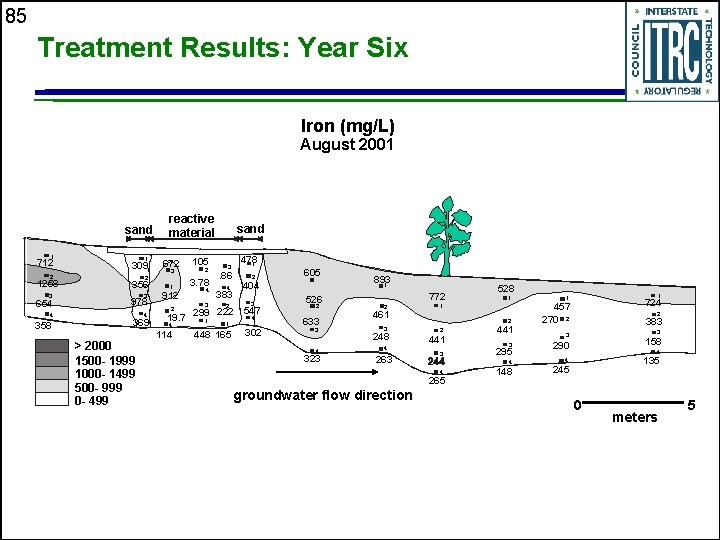
85 Treatment Results: Year Six Iron (mg/L) August 2001 sand 1 712 2 1258 3 654 4 358 1 309 2 356 3 978 4 369 > 2000 1500 - 1999 1000 - 1499 500 - 999 0 - 499 reactive material 672 105 3 2 1 3. 78 912 4 3 sand 3 478 1 2 605 4 404 2 3 526 . 86 383 222 547 4 19. 7 299 1 2 4 114 1 448 165 302 2 633 3 4 323 893 1 772 2 461 3 248 4 263 1 2 2 441 3 4 265 groundwater flow direction 528 1 441 290 4 4 148 724 2 383 3 295 1 1 457 270 2 158 4 135 245 0 meters 5

86 Evidence for Sulfate Reduction in PRB u Decreasing sulfate concentrations • >1, 000 mg/L • >15 mg/L per day u u u Enumeration of sulfate-reducing bacteria Dissolved sulfide present in groundwater Isotopic enrichment of 34 S in remnant sulfate Iron monosulfides identified in cores Continued accumulation of sulfides reflects sustained reactivity of the PRB over time • 1995 -1997: 100 mmol/g per year • 1997 -2001: ~100 mmol/g per year

87 Summary of Nickel Rim PRB u u u u The reactive wall has removed significant portion of the dissolved iron from the plume; full treatment would have required thicker PRB with longer residence time PRB has reduced flux of contaminants in groundwater; reduced acid-generating potential of groundwater in receiving surface water Significant contaminant removal continues Heterogeneities: high groundwater velocity zone with shorter residence time Influence on carbon release from reactive materials Reactivity of other carbon sources Influence of temperature

88 Summary and Conclusions u u u Complete and accurate site characterization is necessary for successful deployment of a PRB Performance assessment should include hydraulic, geochemical, and microbiological evaluation PRBs have been deployed at over 200 sites Other reactive materials such as zeolite, limestone, carbon sources, and phosphates have been deployed for use in PRBs and show promising results PRBs have been shown to be cost effective in relation to conventional technologies

89 Thank You for Participating u Links to additional resources • http: //www. clu-in. org/conf/itrc/prbll/resource. cfm u 2 nd question and answer session © University of Waterloo
- Slides: 89