1 Welcome Thanks for joining us ITRCs Internetbased

1 Welcome – Thanks for joining us. ITRC’s Internet-based Training Program Protocols for Use of Five Passive Samplers ITRC Protocols for Use of Five Passive Samplers to Sample for a Variety of Contaminants in Groundwater (DSP-5, 2007) Sponsored by: Interstate Technology and Regulatory Council (www. itrcweb. org) Hosted by: US EPA Clean Up Information Network (www. cluin. org)
2 Housekeeping u u Course time is 2¼ hours Phone line participants • Do NOT put this call on hold • *6 to mute; #6 to unmute Question & Answer breaks • Phone - unmute #6 to ask question out loud • Simulcast - ? icon at top to type in a question Turn off any pop-up blockers u u Move through slides • Arrow icons at top of screen • List of slides on left Feedback form available from last slide – please complete before leaving This event is being recorded Archives accessed for free http: //cluin. org/live/archive/ Go to slide 1 Move back 1 slide Move forward 1 slide Go to last slide Download slides as PPT or PDF Go to seminar homepage Submit comment or question Report technical problems

3 ITRC Disclaimer and Copyright Although the information in this ITRC training is believed to be reliable and accurate, the training and all material set forth within are provided without warranties of any kind, either express or implied, including but not limited to warranties of the accuracy, currency, or completeness of information contained in the training or the suitability of the information contained in the training for any particular purpose. ITRC recommends consulting applicable standards, laws, regulations, suppliers of materials, and material safety data sheets for information concerning safety and health risks and precautions and compliance with then-applicable laws and regulations. ECOS, ERIS, and ITRC shall not be liable for any direct, incidental, special, consequential, or punitive damages arising out of the use of any information, apparatus, method, or process discussed in ITRC training, including claims for damages arising out of any conflict between this the training and any laws, regulations, and/or ordinances. ECOS, ERIS, and ITRC do not endorse or recommend the use of, nor do they attempt to determine the merits of, any specific technology or technology provider through ITRC training or publication of guidance documents or any other ITRC document. Copyright 2010 Interstate Technology & Regulatory Council, 444 North Capitol Street, NW, Suite 445, Washington, DC 20001
4 ITRC (www. itrcweb. org) – Shaping the Future of Regulatory Acceptance u u Host organization Network u • • • State regulators § All 50 states, PR, DC • Federal partners DOE DOD EPA • ITRC Industry Affiliates Program • Academia • Community stakeholders Wide variety of topics u Technologies Approaches Contaminants Sites Products • Technical and regulatory guidance documents • Internet-based and classroom training

5 ITRC Course Topics Planned for 2010 – More information at www. itrcweb. org Popular courses from 2009 u u u New in 2010 u Decision Framework for Decontamination and Decommissioning of Applying Attenuation Radiologically-Contaminated Facilities Processes to Metals Enhanced Attenuation of Chlorinated Organics and Radionuclides In Situ Bioremediation of Chlorinated Ethene u LNAPL Part 3: DNAPL Source Zones Evaluating LNAPL Part 1: An Improved Understanding of Remedial Technologies for Achieving Project LNAPL Behavior in the Subsurface Goals LNAPL Part 2: LNAPL Characterization and u Mining Waste Recoverability u Remediation Risk Perchlorate Remediation Technologies Management: An Performance-based Environmental Management Approach to Effective Phytotechnologies Remedial Decisions Protocol for Use of Five Passive Samplers and More Protective Quality Consideration for Munitions Response Cleanups Determination/Application of Risk-Based Values Use of Risk Assessment in ITRC 2 -day Classroom Training: Management of Contaminated Sites Vapor Intrusion Pathway

6 Meet the ITRC Instructors Kimberly Mc. Evoy New Jersey Department of Environmental Protection Trenton, New Jersey 609 -530 -8705 Kim. Mc. Evoy@dep. state. nj. us Hugh Rieck US Army Corps of Engineers Omaha, Nebraska 402 -697 -2660 Hugh. J. Rieck@usace. army. mil Louise Parker U. S. Army Engineer Research and Development Center Hanover, New Hampshire 603 -646 -4393 Louise. V. Parker@usace. army. mil

7 What you will learn… u u u What is passive sampling? What passive samplers offer • Quantitative data • Cost savings (40 -70%) How passive samplers reflect aquifer conditions Technical and regulatory guidance Acceptance of passive sampling Classes and types of passive samplers

8 Passive Sampler Team u u Diffusion Sampler Team formed in 2000 Initial goal • Develop guidance on polyethylene diffusion bags (PDBs) for collection of volatile organic compounds (VOCs) in groundwater § 1 st passive sampling device - diffusion type sampler (DSP-3) § Limited in analyte capabilities § Increased interest and development of passive devices u Transition to “Passive Sampler Team” • • What technologies are being developed and what they can do? Disseminate guidance on passive sampling technologies Be premier resource on the use of passive sampling technologies Promote adoption of regulatory guidance (i. e. , acceptance)

9 What Does a Purge Sample Represent? u u u Active transport of water induced either by pumping or hand-purging Often draws water from above and below as well as adjacent to the screened interval/open borehole Flow-weighted average • Based on indicator parameter stabilization or evacuation of the sampling system (i. e. , volume purge) • Gas exchange and mixing u May elevate turbidity • Mobilization of colloids and sediment • Mobilization of normally immobile NAPL microglobules u Compliance with drinking water standards

10 What Does a Passive Sample Represent? No active transport of water induced by pumping or purging u Samples are collected from a specific depth u Rely on sampling device and well water being in ambient equilibrium with the formation water during deployment period u Reduce disturbance to the well and aquifer typically caused by bailing or over-pumping u Reduce turbidity u • Represent “natural conditions”

11 Advantages of Passive Samplers u u Highly reproducible data Provides low turbidity samples Disposable/dedicated - no decontamination between wells Decrease costs • Field labor § Rapid field deployment and collection § Leave in quarterly u u • Little or no disposal cost (no purge-water) Samples discrete intervals • Vertical contaminant profiling • Monitor zone of highest contaminant influx Easy to use – minimal equipment needs No depth limit “Green” sampling method

12 Limitations of Passive Samplers May have volume/analyte limitations u Contaminant stratification requires consideration before deploying u Well must restabilize before sample collection u

13 Passive Sampler Team Publications u User’s Guide for Polyethylene-Based Passive Diffusion Bag Samplers to Obtain VOC Concentrations in Wells (March 2001, DSP-1) • Jointly developed with USGS • Basic principles for deployment u Technical and Regulatory Guidance for Using Polyethylene Diffusion Bag Samplers to Monitor VOCs in Groundwater (February 2004, DSP-3) • Easy to use for groundwater and surface water • Quantify savings (40 -70%) u Technology Overview of Passive Sampler Technologies (March 2006, DSP-4) u ITRC Protocols for Use of Five Passive Samplers to Sample for a Variety of Contaminants in Groundwater (February 2007, DSP-5 ) • Main application was groundwater sampling • Summarized 12 passive sampling technologies • Details on “mature” passive sampling technologies from Overview Document (DSP-4) http: //www. itrcweb. org/teamresources_1. asp

14 Classes of Passive Samplers u Diffusion Samplers: analytes reach and maintain equilibrium via diffusion through membrane • Regenerated-Cellulose Dialysis Membrane (Dialysis) Sampler • Rigid Porous Polyethylene (RPP) Sampler u Equilibrated Grab Samplers: collect a whole-water sample instantaneously • Snap Sampler™ • Hydra. Sleeve™ Sampler u Accumulation Sampler: rely on diffusion and sorption to accumulate analytes in sampler • GORE™ Module

15 Ambient Flow Through a Well u Relies on flow through in the well screen • Screened zone is in active exchange with formation water u u Water above screen may be “stagnant” References • ASTM, 2002 • Powell R. M. , and R. W. Puls, 1993 • Robin, M. J. L. and R. W. Gillham, 1987

16 General Deployment Device suitable for analytes of interest u Sample volume u • i. e. , QA/QC and duplicates • Appendix A: Minimum Volumes u Deployment period • Device and site specific § Well restabilization § Sampler equilibration u Deployment depth • Should not be arbitrary § Depends on well or site specific data quality objectives (DQOs) • Sampler represents a depth interval

17 Contaminant Stratification u Stratification is well-specific u Majority of wells are not stratified u Contaminant stratification in an aquifer vs. in the well
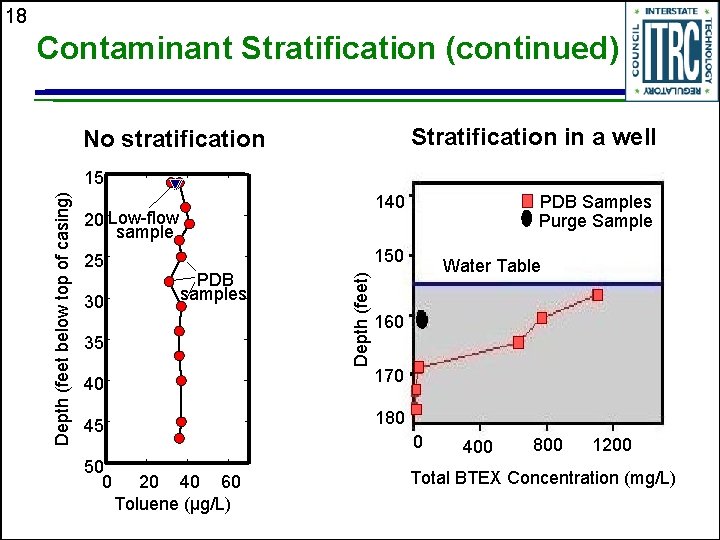
18 Contaminant Stratification (continued) Stratification in a well No stratification 140 20 Low-flow sample 25 30 PDB samples 35 Water Table 160 40 170 45 180 50 0 PDB Samples Purge Sample 150 Depth (feet) Depth (feet below top of casing) 15 0 20 40 60 Toluene (µg/L) 400 800 1200 Total BTEX Concentration (mg/L)

19 Contaminant Distribution u Multiple samplers deployed through screened or open interval • Can represent contaminant concentrations over water column u Vertical flow profiling, depending data quality objectives (DQOs), determines primary input/exit of groundwater flow • Borehole flowmeter • Interval packer/pump tests u Profiling techniques can aid in • Refining site conceptual model • Remedial process optimization (RPO) u Profiling techniques • Target a specific depth interval • Can monitor interval with highest concentration § Conservative approach for long-term monitoring

20 Data Quality Objectives (DQOs) Prior to implementation, all parties should agree on DQOs u For instance u • Vertical contaminant distribution may be a DQO so multiple samplers deployed in a well may be advised (vertical profiling) • Long-term monitoring projects, a single sampler may be appropriate for the DQO Is your sampling method meeting the DQOs? u Do all parties agree? u

21 Data Quality Objectives (DQOs) (continued) u DQOs define • Sampling goal • Target analytes • Hydrologic concerns Pumping moves water toward intake from the induced flow field in proportion to hydraulic conductivity Every groundwater sampling technique characterizes contamination differently! u u Pumping methods • Draw groundwater into the well screen from an undefined area • Example: 3 -volume purge and low flow Passive methods • Sample depth-specific intervals in well • Groundwater moves through the well screen under ambient flow conditions Pump Intake

22 Regulatory Perspective Does your state have any Statutes, Regulations, or Guidance that prohibit or impede the use of passive sampling technologies for the collection of groundwater samples? (16 state responses: Appendix B) u u No regulatory or statutory prohibitions to using passives samplers “De facto” acceptance of passive samplers in 50 states and worldwide New Jersey Department of Environmental Protection guidance on polyethylene diffusion bags (PDBs) (2005) Regulatory agencies use ITRC Polyethylene Diffusion Bag (PDB) guidance for state guidance Passive samplers have been used

23 In Summary u u Passive samples collect analytes that come in contact with the sampler under ambient flow Value of passive samplers • Inexpensive • Broad analyte capabilities • Reduced sampler error Assist in site characterization identifying • Stratification • Target zones for remediation • Migration pathways 1: 1 correlation may not occur • Discrete concentration vs. flow weighted concentration • May reflect nature of sampling method § i. e. , dilution during purging, pumping versus passive

24 Questions and Answers Covered so far u Introduction to passive (nopurge) sampling u Advantages/limitations u General considerations when using passive samplers u Regulatory perspectives Next – technical aspects for five passive samplers u Diffusion Samplers: analytes reach and maintain equilibrium via diffusion through membrane 1. Regenerated-Cellulose Dialysis Membrane (Dialysis) Sampler 2. Rigid Porous Polyethylene (RPP) Sampler u Now u Questions and answers Equilibrated Grab Samplers: collect a whole-water sample instantaneously 3. Snap Sampler™ 4. Hydra. Sleeve™ Sampler u Accumulation Sampler: rely on diffusion and sorption to accumulate analytes in sampler 5. GORE™ Module

25 Diffusion Samplers u Diffusion Samplers: analytes reach and maintain equilibrium via diffusion through membrane 1. Regenerated-Cellulose Dialysis Membrane (Dialysis) Sampler 2. Rigid Porous Polyethylene (RPP) Sampler u Equilibrated Grab Samplers: collect a whole-water sample instantaneously 3. Snap Sampler™ 4. Hydra. Sleeve™ Sampler u Accumulation Sampler: rely on diffusion and sorption to accumulate analytes in sampler 5. GORE™ Module
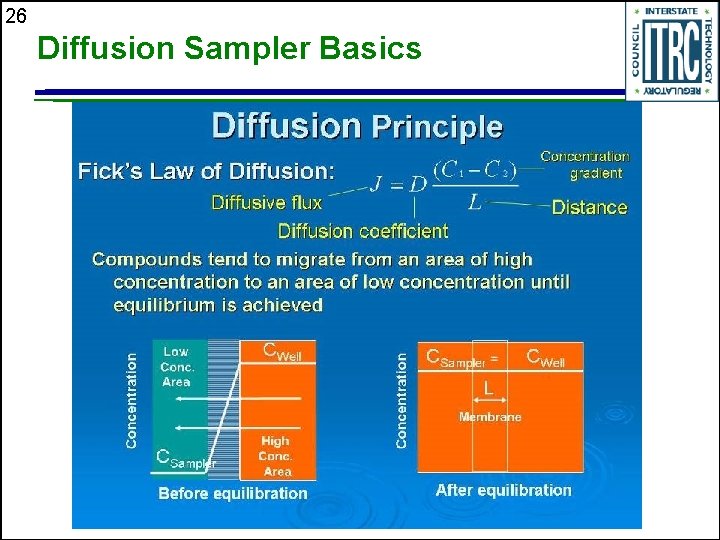
26 Diffusion Sampler Basics

27 Diffusion Sampler Advantages u u Groundwater sampling time in the field is decreased – no pumping needed Eliminates purge water and disposal costs Excludes turbidity from groundwater samples – no filtering needed Disposable – no cleaning or cross-contamination Regenerated Cellulose Dialysis Membrane (Dialysis) Rigid Porous Polyethylene (RPP)

28 Regenerated-Cellulose Dialysis Membrane Sampler Basics u u u u Referred to as the “Dialysis Sampler” Regenerated-cellulose dialysis membrane • Filled with deionized water • Hydrophilic membrane Currently must be constructed Membrane sizes • 2. 5 -inch diameter for 4 -inch wells • 1. 25 -inch diameter for 2 -inch wells Sample volumes • 2. 5 -inch x 2 ft long contains 2 liters • 1. 25 -inch x 2 ft long contains 500 mls Pore size is 18 Angstroms Developed by U. S. Geological Survey (USGS) Fully assembled Dialysis sampler ready for deployment

29 Dialysis Equilibration Times Determined in laboratory in bench-scale tests u 95% or greater equilibrium reached in dialysis samplers within u • 1 -7 days for most cations and trace metals • 1 -3 days for all VOCs on 8260 B list (including MTBE) • 1 -3 days for anions, silica, DOC, CH 4, sulfide • 7 -14 days for explosives compounds • 28 days or more for Hg, Ag, Sn

30 Dialysis Sampler Advantages Collects inorganic and organic chemical constituents u Quick equilibration and deployment times – generally 1 -2 weeks u Relatively inexpensive to construct u Excludes turbidity from groundwater samples – no filtering needed u Sample volume can be up to 2 L u

31 Dialysis Sampler Limitations u u u Must construct sampler from raw materials Samplers must be kept wet between construction and deployment Membrane can biodegrade within 4 -6 weeks • Not a problem for shorter deployments • Can maintain integrity for longer periods in very cold water u Samplers lose water volume slowly (<3% per week) • Not a problem for short deployments • Internal support for high ionic strength waters is available
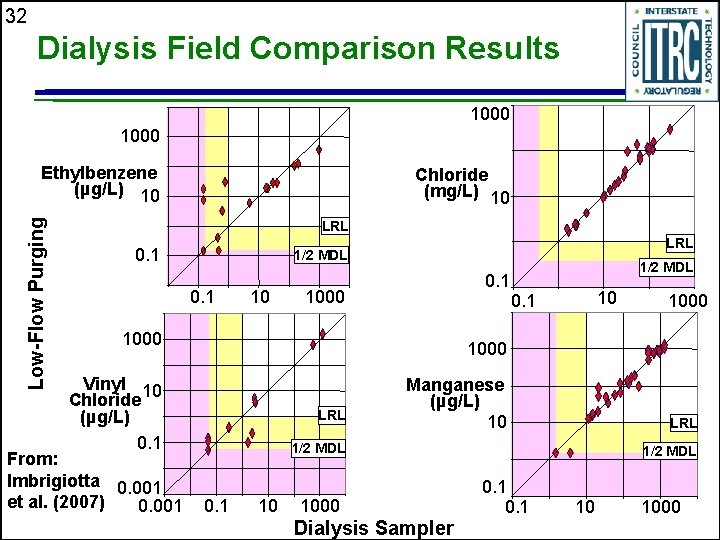
32 Dialysis Field Comparison Results 1000 Low-Flow Purging Ethylbenzene (µg/L) 10 Chloride (mg/L) 10 LRL 0. 1 LRL 1/2 MDL 0. 1 10 1000 10 0. 1 1000 Vinyl 10 Chloride (µg/L) 0. 1 From: Imbrigiotta 0. 001 et al. (2007) 0. 001 1/2 MDL 0. 1 LRL Manganese (µg/L) 10 LRL 1/2 MDL 0. 1 10 1000 Dialysis Sampler 1/2 MDL 0. 1 10 1000

33 Dialysis Field Comparison Results (Dialysis Samplers vs Purging Methods) u Parameters with favorable results • VOCs • Cations and anions • Most trace metals • Explosive compounds • Others (silica, ethene, CO 2, CH 4, TDS, SC, DOC) u Parameters with questionable results • p-Isopropyltoluene • n-Butylbenzene • s-Butylbenzene • Nickel • Sulfide See Table 5 -3 in ITRC Protocols Document (DSP-5)

34 Dialysis Sampler Summary u u u Collects both organic and inorganic chemical constituents Do not require filtration of samples Equilibrate within 1 -2 weeks for most constituents Deployment times 1 -2 weeks in most wells Dialysis samplers recover comparable concentrations of • VOCs vs. PDB samplers • VOCs and most inorganics vs. low-flow and purging and sampling u Dialysis samplers should not be used when • Sampling for mercury, silver, or tin • Equilibration will take longer than 4 weeks • Total concentrations are needed u Dialysis samplers should be used with caution when • Sampling for nickel and sulfide

35 Rigid Porous Polyethylene (RPP) Samplers Cap Made of rigid, porous polyethylene u Pore sizes 6 -15 microns u 5 inches long u 1. 5 inches in diameter u Filled with deionized water u Standard size In protective mesh ready for holds 90 -100 m. L deployment and packaged in u Delrin plug disposable water-filled sleeve for shipping
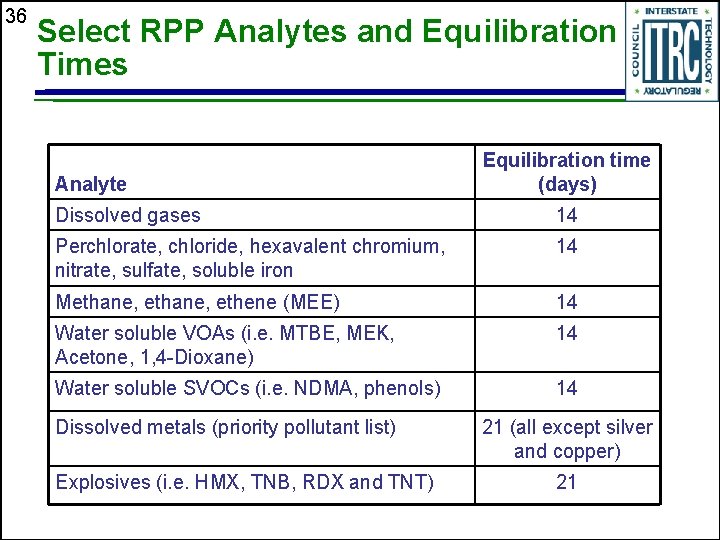
36 Select RPP Analytes and Equilibration Times Analyte Equilibration time (days) Dissolved gases 14 Perchlorate, chloride, hexavalent chromium, nitrate, sulfate, soluble iron 14 Methane, ethene (MEE) 14 Water soluble VOAs (i. e. MTBE, MEK, Acetone, 1, 4 -Dioxane) 14 Water soluble SVOCs (i. e. NDMA, phenols) 14 Dissolved metals (priority pollutant list) Explosives (i. e. HMX, TNB, RDX and TNT) 21 (all except silver and copper) 21

37 RPP Advantages Can be used to collect most inorganic and limited organic analytes u Are commercially available and field-ready u Can be stacked when additional volume needed u Excludes particles larger than the pore space of the sampler u

38 RPP Limitations Must be stored and shipped fully immersed in deionized water u Have not been tested for all analytes u Multiple samplers are needed to obtain sufficient volume for multiple Analyte types and/or QA/QC u Requires advanced analytical techniques to analyze for SVOCs u Equilibrium times for less water soluble VOCs and SVOCs are not currently known u
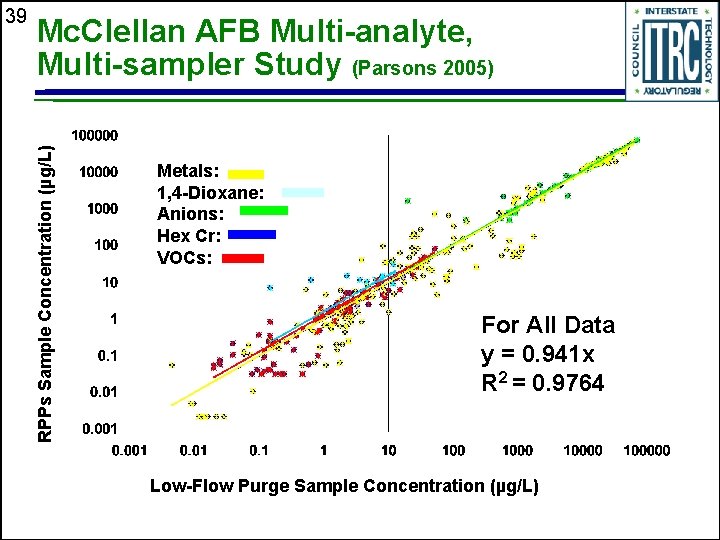
Mc. Clellan AFB Multi-analyte, Multi-sampler Study (Parsons 2005) RPPs Sample Concentration (µg/L) 39 Metals: 1, 4 -Dioxane: Anions: Hex Cr: VOCs: For All Data y = 0. 941 x R 2 = 0. 9764 Low-Flow Purge Sample Concentration (µg/L)
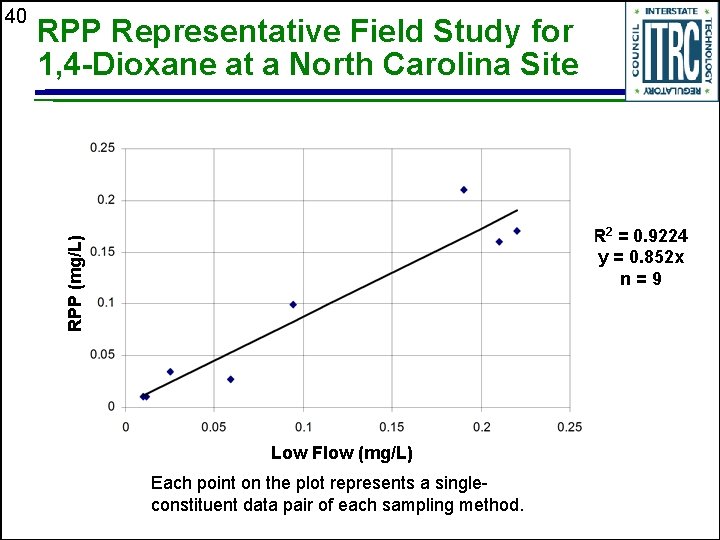
RPP Representative Field Study for 1, 4 -Dioxane at a North Carolina Site R 2 = 0. 9224 y = 0. 852 x n=9 RPP (mg/L) 40 Low Flow (mg/L) Each point on the plot represents a singleconstituent data pair of each sampling method.
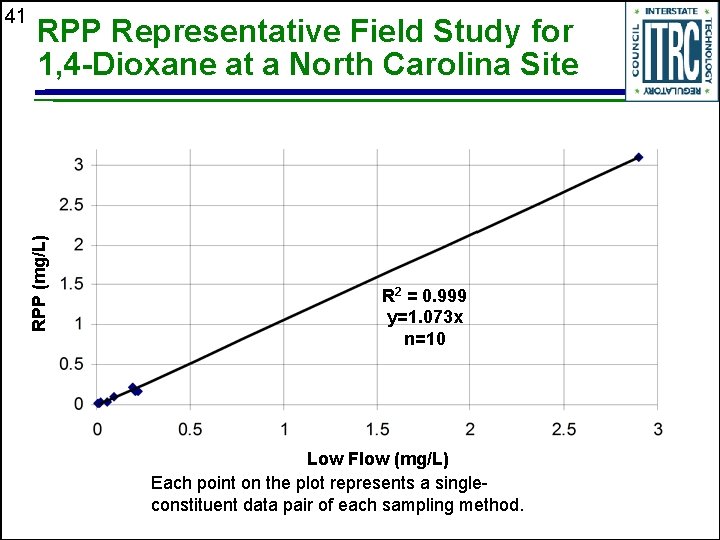
RPP Representative Field Study for 1, 4 -Dioxane at a North Carolina Site RPP (mg/L) 41 R 2 = 0. 999 y=1. 073 x n=10 Low Flow (mg/L) Each point on the plot represents a singleconstituent data pair of each sampling method.

42 RPP Summary u Can be used to sample for • Most inorganics • Water soluble VOCs and SVOCs It’s not currently known if they can be used for water-insoluble VOCs and SVOCs u Can be used in deep wells u Can be used in conjunction with PDBs u Disposable sampler u • No decontamination required

43 Diffusion Sampler Summary Regenerated Cellulose Dialysis Membrane u u u u Rigid Porous Polyethylene (RPP) RPP and Dialysis Membrane samplers can be used for VOCs, SVOCs, metals, anions, and cations Minimum deployment time for RPP and Dialysis sampler is ~2 weeks Compare well with conventional methods Collect samples at a discrete interval in well screen RPP sampler can be used for quarterly or longer deployments Major limitation of RPP sampler is sample volume Major limitation of Dialysis sampler is that it undergoes biodegradation

44 Equilibrated Grab Samplers u Diffusion Samplers: analytes reach and maintain equilibrium via diffusion through membrane 1. Regenerated-Cellulose Dialysis Membrane (Dialysis) Sampler 2. Rigid Porous Polyethylene (RPP) Sampler u Equilibrated Grab Samplers: collect a whole-water sample instantaneously 3. Snap Sampler™ 4. Hydra. Sleeve™ Sampler u Accumulation Sampler: rely on diffusion and sorption to accumulate analytes in sampler 5. GORE™ Module

45 Equilibrated Grab Samplers u u u Collects sample from discrete interval in well screen Collect “whole water” samples that can be tested for any analyte Collects samples in “real time” Equilibration period allows • Well to recover from sampler placement • Materials to equilibrate with analytes in well water Technologies Hydra. Sleeve™ • Snap Sampler™ Sampler • Hydra. Sleeve™ Sampler Snap Sampler™

46 Snap Sampler. TM Components u u Sampler body with trigger mechanism Bottles • Have two openings & spring -activated caps • 40 -m. L VOA glass vials § Fits in 2 -inch wells • 125 -m. L HDPE bottles § Fits in 2 -inch wells • 350 -m. L HDPE bottles § Fits in 4 -inch wells Trigger line • Mechanical • Electronic • Pneumatic Docking station 40 m. L 125 m. L

47 Snap Sampler. TM – Collecting a Sample u u u Sample bottles deployed & remain in open position Equilibration period • Minimum of 1 to 2 weeks • Can be used for quarterly, semi-annual, or annual sampling Pull handle on trigger line to close bottle (i. e. , collect sample) Samples sealed in situ No sample transfer required at the surface

48 Snap Sampler. TM Advantages u u No analyte restrictions Reduced sampling variability • Minimal agitation of well during sampling § Collect samples with ambient turbidity • Bottles remain sealed under in-situ conditions • No sample transfer § No exposure to weather, surface contamination, etc. § Some studies have shown better recovery of volatiles and gases

49 Snap Sampler. TM Limitations u Sample Volume • Multiple bottles are needed to obtain volume for multiple analyte types and/or QA/QC u Trigger lines are fixed length and thus cannot be readily moved to other wells
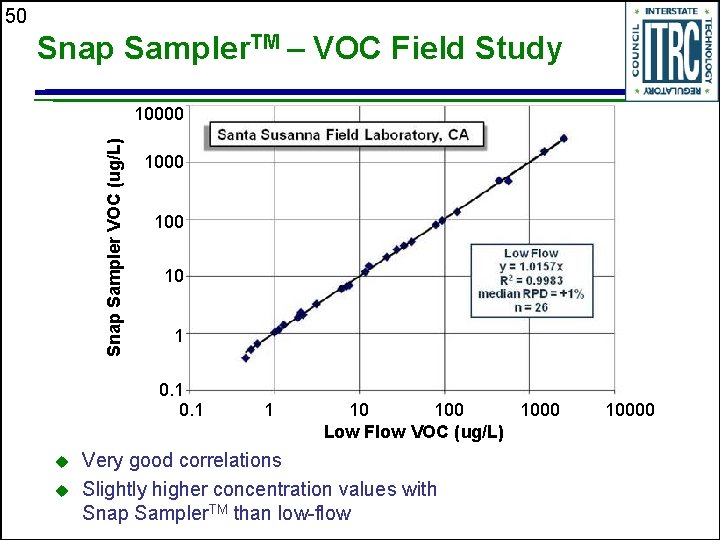
50 Snap Sampler. TM – VOC Field Study Snap Sampler VOC (ug/L) 10000 100 10 1 0. 1 u u 1 10 1000 Low Flow VOC (ug/L) Very good correlations Slightly higher concentration values with Snap Sampler. TM than low-flow 10000
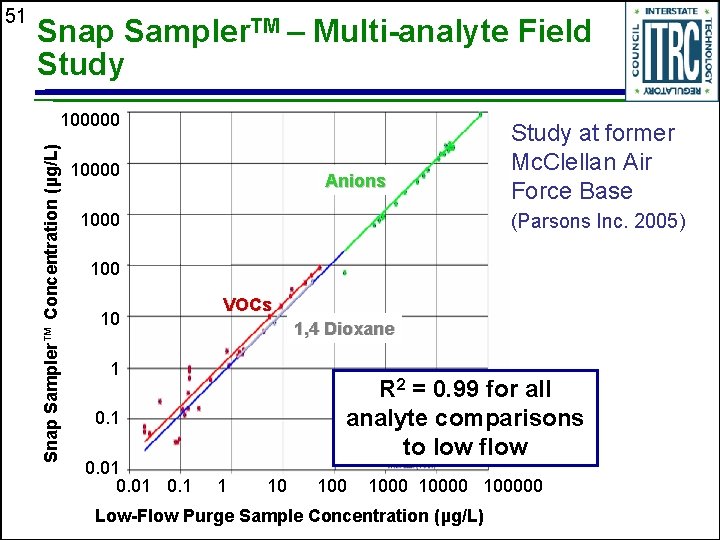
Snap Sampler. TM – Multi-analyte Field Study 100000 Snap Sampler™ Concentration (µg/L) 51 10000 Anions 1000 Study at former Mc. Clellan Air Force Base (Parsons Inc. 2005) 100 10 VOCs 1, 4 Dioxane 1 R 2 = 0. 99 for all analyte comparisons to low flow 0. 1 0. 01 0. 1 1 10 100000 Low-Flow Purge Sample Concentration (µg/L)

52 Snap Sampler™ Summary u u u Sample all analyte types Volume limited for long analyte list Samples are sealed at the point of collection No transfer of sample required Data correlates well with standard sampling methods

53 Hydra. Sleeve™ Components Sampler sleeve Reed valve Discharge tube Reusable sampler weight
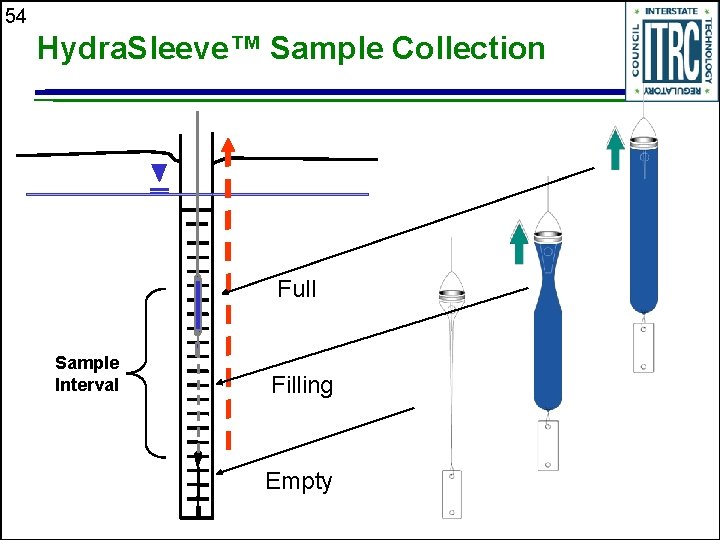
54 Hydra. Sleeve™ Sample Collection Full Sample Interval Filling Empty

55 Hydra. Sleeve™ Advantages u u u Fits in most diameter wells Can sample all types of analytes Sample Volume • 2 -inch HS collects 650 m. L to 1 L • 4 -inch HS collects 1250 m. L to 2 L u u Easy to use with minimal training Can sample • Very deep wells • Crooked wells u Can collect low turbidity samples

56 Hydra. Sleeve™ Limitations u Sample Volume • Custom samplers can be fabricated in a wider diameter and/or longer length to maximize sample volume for longer analyte lists • Work with lab regarding minimum sample volume § Using table in Appendix A of the ITRC Protocol Document, DSP-5
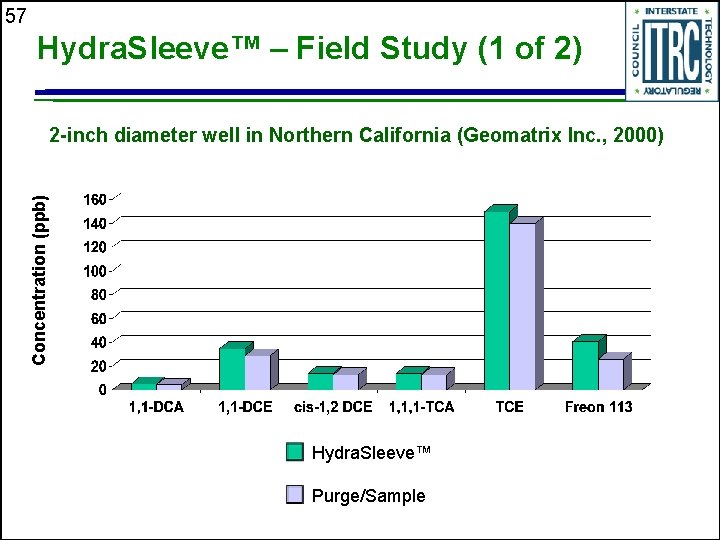
57 Hydra. Sleeve™ – Field Study (1 of 2) Concentration (ppb) 2 -inch diameter well in Northern California (Geomatrix Inc. , 2000) Hydra. Sleeve™ Purge/Sample

58 Hydra. Sleeve™ – Field Study (2 of 2) Former Mc. Clellan Air Force Base (Parsons Inc. , 2005) u u Comprehensive comparison of • Low-flow and 3 -well volume purged samples • Samples collected using 6 no-purge samplers Analytes included • VOCs, 1, 4 dioxane, anions, metals, and hexavalent chromium u Study Findings • “The Hydra. Sleeve and Snap Sampler. TM produced results most similar to the higher concentrations obtained by lowflow and 3 -well volume purging and sampling methods” • “Appears to be a technically viable method for monitoring all of the compounds in the demonstration”

59 Hydra. Sleeve™ Sampler Summary u u u Sample all analyte types Sample volumes up to 2 L Can be used in • Deep wells • Crooked wells Comparable results to conventional pumped methods Can be left in well for quarterly, semi-annual, or annual sampling Disposable sampler • No decontamination required

60 Equilibrated Grab Samplers Summary Snap Sampler™ Hydra. Sleeve™ Sampler u u u Samples can be analyzed for all analyte types • Providing there is adequate sample volume Collect whole water samples in real-time Can be used for quarterly, semi-annual, or annual sampling events Use an equilibration period to reduce sampling biases Collect samples at a discrete interval in well screen Compare well with conventional methods

61 Accumulation Samplers u Diffusion Samplers: analytes reach and maintain equilibrium via diffusion through membrane 1. Regenerated-Cellulose Dialysis Membrane (Dialysis) Sampler 2. Rigid Porous Polyethylene (RPP) Sampler u Equilibrated Grab Samplers: collect a whole-water sample instantaneously 3. Snap Sampler™ 4. Hydra. Sleeve™ Sampler u Accumulation Sampler: rely on diffusion and sorption to accumulate analytes in sampler 5. GORE™ Module

62 Accumulation Samplers u u Rely on diffusion and sorption Examples of accumulation samplers • • u Semi-permeable Membrane Devices (SPMD) Polar Organic Chemical Integrative Sampler (POCIS) Passive In-situ Concentration Extraction Sampler (PISCES) GORE™ Module More information on other accumulation samplers is available • Overview of Passive Sampler Technologies (March 2006, DSP-4) • http: //www. itrcweb. org/

63 GORE™ Module Components Knot (secure to wellhead) Attachment Line Loop to attach line u GORE™ Module Tag with unique serial number • Section 2 in ITRC Protocol Document (DSP-5) u u Attachment Line Stainless steel weights Adsorbent Weight

64 GORE™ Module Sample Collection u u Dissolved compounds partition to vapor (Henry’s Law) Diffusion through hydrophobic, vapor-permeable membrane Adsorption onto media GORE-TEX® Membrane Duplicate samples Vapors pass through Liquid water remains outside Adsorbents

65 GORE™ Module Analysis u u No adsorbent transfer in field Thermal desorption/GC/MS • VOCs and SVOCs • US EPA Method 8260/8270, modified for thermal desorption

66 GORE™ Module Advantages u Sample small diameter wells and multi-level systems • >0. 25 inches • Crooked wells u u No minimum sample volume limitation No need to refrigerate samples Minimal water disruption - ~10 mls displacement Short sampling period - 15 minutes to 4 hours • Longer-term deployment – sub ppb concentrations u US EPA ETV verified (Einfeld and Koglin, 2000) • http: //www. epa. gov/etv/pdfs/vrvs/01_vr_gore. pdf

67 GORE™ Module Limitations Sole source supplier and laboratory analysis u Organic compounds only u • Compound detection limited by vapor pressure u Data reporting • Measured mass (µg) • Concentrations are calculated by GORE based on § Measured mass, sampling rate, time, water temperature, and water pressure – Reference Section 2. 4. 5 of ITRC Protocol Document (DSP-5)
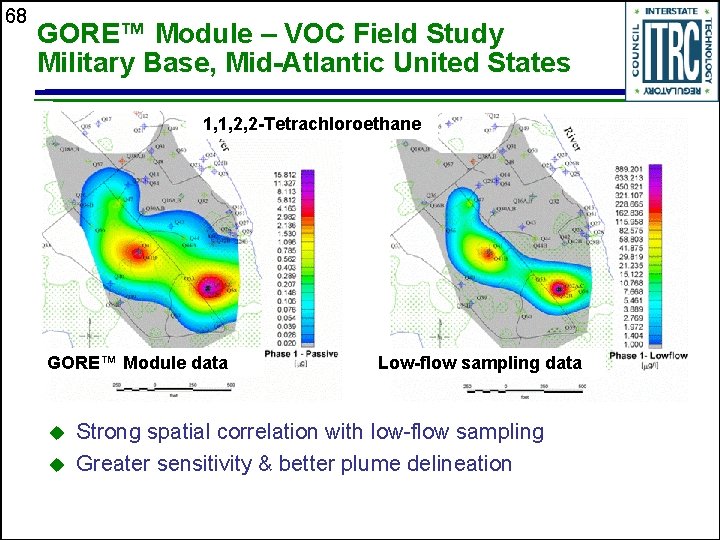
68 GORE™ Module – VOC Field Study Military Base, Mid-Atlantic United States 1, 1, 2, 2 -Tetrachloroethane GORE™ Module data u u Low-flow sampling data Strong spatial correlation with low-flow sampling Greater sensitivity & better plume delineation
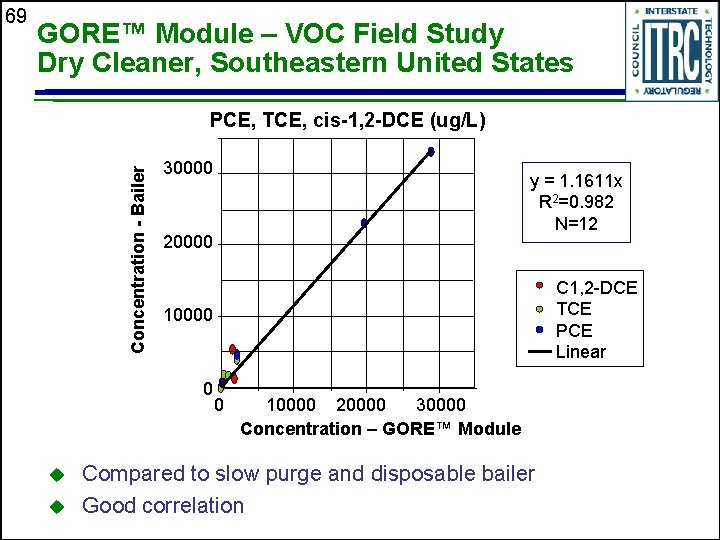
GORE™ Module – VOC Field Study Dry Cleaner, Southeastern United States PCE, TCE, cis-1, 2 -DCE (ug/L) Concentration - Bailer 69 30000 20000 u C 1, 2 -DCE TCE PCE Linear 10000 0 u y = 1. 1611 x R 2=0. 982 N=12 0 10000 20000 30000 Concentration – GORE™ Module Compared to slow purge and disposable bailer Good correlation

70 GORE™ Module Summary u Accumulation sampler u Easy field deployment u Able to detect very low analyte concentrations Collect samples at a discrete interval in well screen Data reported u u • Passive operation • Compounds partition to vapor, then diffuse to adsorbent • Small diameter wells • Short sampling period • Mass measured or as concentrations (calculated) Data comparable with conventional sampling Can only be used for organic compounds

71 Overall Summary for Protocols for Use of Five Passive Samplers u Passive Samplers offer • Quantitative data • Cost savings u u Use is dependent upon the DQOs Tech & Reg Guidance Acceptance Diffusion Samplers • RPP & Dialysis u Equilibrated Grab Samplers • Snap Sampler™ & Hydra. Sleeve™ u Accumulation Sampler • GORE™ Module

72 Thank You for Participating u 2 nd question and answer break u Links to additional resources • http: //www. clu-in. org/conf/itrc/passsamp/resource. cfm u Feedback form – please complete • http: //www. clu-in. org/conf/itrc/passsamp/feedback. cfm Need confirmation of your participation today? Fill out the feedback form and check box for confirmation email.
- Slides: 72